The growing complexity of platelet aggregation Review article
advertisement

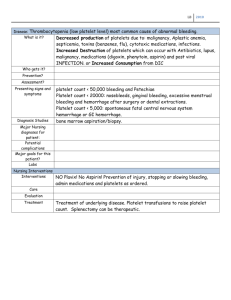
From www.bloodjournal.org by guest on September 7, 2015. For personal use only. Review article The growing complexity of platelet aggregation Shaun P. Jackson Australian Centre for Blood Diseases, Monash University, Melbourne, Australia Platelet aggregation, the process by which platelets adhere to each other at sites of vascular injury, has long been recognized as critical for hemostatic plug formation and thrombosis. Until relatively recently, platelet aggregation was considered a straightforward process involving the noncovalent bridging of integrin ␣IIb3 receptors on the platelet surface by the dimeric adhesive protein fibrinogen. However, with recent technical advances enabling real-time analysis of platelet aggre- gation in vivo, it has become apparent that this process is much more complex and dynamic than previously anticipated. Over the last decade, it has become clear that platelet aggregation represents a multistep adhesion process involving distinct receptors and adhesive ligands, with the contribution of individual receptorligand interactions to the aggregation process dependent on the prevailing blood flow conditions. It now appears that at least 3 distinct mechanisms can initiate platelet aggregation, with each of these mechanisms operating over a specific shear range in vivo. The identification of shear-dependent mechanisms of platelet aggregation has raised the possibility that vascular-bed–specific inhibitors of platelet aggregation may be developed in the future that are safer and more effective than existing antiplatelet agents. (Blood. 2007;109:5087-5095) © 2007 by The American Society of Hematology Introduction Historical aspects The propensity of platelets to clump together at sites of vascular injury was first recognized more than 100 years ago.1-4 This phenomenon, most accurately described as platelet cohesion although more commonly referred to as platelet aggregation, was quickly identified as important for hemostatic plug formation.5 It was also recognized at the time that platelets played a key role in the development of thrombosis,6 but, it was not until almost a century later that it became widely accepted that platelets played a pivotal role in development of cardiovascular disease.7 As a consequence, inhibitors of platelet aggregation have become increasingly important parts of the armamentarium for the prevention and treatment of many atherothrombotic disorders.8,9 For more than 3 decades, the factors mediating platelet aggregation appeared conceptually straightforward, requiring a platelet stimulus (agonist), a soluble adhesive protein (fibrinogen), and a membrane-bound platelet receptor (integrin ␣IIb3 or GPIIb-IIIa), leading to a simple unified model of platelet aggregation (Figure 1). Although these core elements remain fundamental, recent technical advances allowing real-time analysis of platelet aggregation in vivo have demonstrated a much more complex and dynamic process than previously anticipated. It is now widely accepted that one of the key elements influencing the platelet aggregation process is blood flow, with evidence that distinct aggregation mechanisms operate under different shear conditions.10-13 This has raised the interesting possibility that vascular bed-specific inhibitors of platelet aggregation may be developed in the future and has stimulated a reevaluation of the temporal sequence of events underlying the aggregation process. In this review, I discuss recent developments in the study of platelet aggregation with particular focus on the mechanisms operating under rapid blood flow. Platelet aggregation was first recognized in the late 1800s after a series of pioneering studies by Zahn (1872), Hayem (1878), Bizzozero (1881 and 1882), Osler (1886), and Eberth and Schimmelbusch (1885-1888).1-4,6 Using intravital imaging techniques, Bizzozero clearly established that small cell elements (in which he originated the term “Blut Plattchen” or blood platelets) clumped together at sites of vessel damage,1,14 a process subsequently described as viscous metamorphosis.2 Through a series of insightful observations, Bizzozero described the changes platelets undergo after exposure to foreign surfaces, including the formation of aggregates and subsequent “white thrombi.” He also clearly described the plugging of small vascular punctures by platelet thrombi, heralded as a major discovery at the time.5 It was soon appreciated that quantitative or qualitative defects in platelets lead to a bleeding disorder; and in 1918, Glanzmann15 described a familial bleeding diathesis (“hereditary hemorrhagic thrombasthenia”) that was later shown to be a primary defect in platelet aggregation. Progress in the understanding of platelet aggregation was slow to develop until the advent of the platelet aggregometer in 1962 by Born16 and independently by O’Brien.17 This relatively simple technique involved stirring a suspension of platelets in the presence of a platelet activating substance and, by monitoring changes in light transmission, the device could accurately monitor platelet clumping (aggregation) in suspension (Figure 1). Aggregometry revolutionized the study of platelet function, providing fundamental insights into the molecular mechanisms underlying plateletplatelet adhesive interactions. The identification and characterization of all major physiological agonists, antagonists, surface receptors, and intracellular signaling pathways in platelets has involved platelet aggregation studies, and in many situations there is a good correlation between defects in aggregation and bleeding disorders Submitted December 27, 2006; accepted February 9, 2007. Prepublished online as Blood First Edition Paper, February 20, 2007; DOI 10.1182/blood- 2006-12-027698. © 2007 by The American Society of Hematology BLOOD, 15 JUNE 2007 䡠 VOLUME 109, NUMBER 12 5087 From www.bloodjournal.org by guest on September 7, 2015. For personal use only. 5088 JACKSON BLOOD, 15 JUNE 2007 䡠 VOLUME 109, NUMBER 12 providing insight into the role of thrombin generation in regulating platelet aggregation at different flow rates. These assays have been instrumental in defining the adhesive steps mediating platelet adhesion to extracellular matrices, including a key role for the von Willebrand factor (VWF)–GPIb interaction in promoting initial platelet tethering to the vessel wall22,23 and a major role for collagen through engagement of GPVI and integrin ␣21 in promoting platelet arrest.21,24-26 These experimental systems have also confirmed an essential role for integrin ␣IIb3 (GPIIb-IIIa) in mediating platelet aggregation over the full range of shear conditions operating in vivo. Thus, flow-based studies were pivotal in the development of the concept that platelet adhesion (mediated by VWF and collagen) is distinct from platelet aggregation (mediated by fibrinogen). Conceptual advances in the understanding of platelet aggregation Figure 1. Traditional model of platelet aggregation. (A) Based on studies with the platelet aggregometer, 3 key elements have been identified as important for platelet aggregation: an activating stimulus (typically a soluble agonist), a plasma protein (predominantly fibrinogen), and a platelet surface receptor (integrin ␣IIb3 or GPIIbIIIa). Agonist-induced activation of integrin ␣IIb3 is essential for the binding of fluid-phase fibrinogen, which as a consequence of its dimeric structure, can physically bridge 2 adjacent platelets. (B) The platelet aggregometer is a relatively simple technique that involves stirring a suspension of platelets in the presence of a platelet activating substance and by monitoring changes in light transmission, the device can accurately monitor platelet clumping (aggregation) in suspension. in affected individuals. Similarly, antiplatelet drugs that increase bleeding risk in vivo can be detected using standard aggregation assays, further enhancing the clinical use of the assay. Thus, for many years, the aggregometer has provided an accurate readout of the fundamental processes regulating aggregation; as recognized in the early 1970s, however, this experimental system does not take into consideration the important influence of blood flow, a key variable increasing the complexity of the aggregation process. Importance of blood flow in the regulation of platelet aggregation An important consideration for platelet aggregation, and for platelet adhesion mechanisms more generally, is the rheological (blood flow) conditions operating at sites of vascular injury. Platelets are exposed to a broad range of hemodynamic conditions in vivo, ranging from relatively low flow situations in venules and large veins (typical wall shear rates ⬍ 500 s⫺1) to small arterioles (shear rates up to 5000 s⫺1) to stenosed arteries with shear rates as high as 40 000 s⫺1.18 Platelets have the unique capacity to form stable adhesion contacts over all shear conditions operating in vivo and are indispensable for hemostatic plug formation and thrombosis at elevated shear rates.19 In an aggregometer, platelet clumping occurs under low, nonlaminar shear conditions, experimental conditions that do not adequately simulate the flow-dependent recruitment (cohesion) of platelets onto thrombogenic surfaces. Experimental systems designed to study platelet adhesion and aggregation on thrombogenic substrates were initially developed in the 1970s.20,21 Such systems enable assessment of platelet aggregation in flowing whole blood (with or without anticoagulant) A number of studies in the 1980s suggested that the processes underlying platelet aggregation under flow conditions may be more complicated than those operating in the aggregometer. Thus, at high shear rates, VWF27,28 and fibronectin29 were demonstrated to not only promote primary adhesion, but also subsequent platelet aggregation. However, widespread recognition of the growing complexity of the platelet aggregation process was slow to develop. A number of technical advances in the late 1990s, including the development of genetically engineered mouse models and improved live cell imaging techniques enabling high-resolution visualization of the platelet aggregation process in vitro30 and in vivo,31,32 have profoundly impacted the study of platelet aggregation, heralding a renaissance in the field. In combination, these techniques have been instrumental in clarifying many aspects of the platelet aggregation process, including (a) the demonstration of a role for multiple adhesive ligands and receptors in the aggregation process; (b) identifying the importance of membrane tethers in initiating platelet aggregation; (c) improved insight into the role of individual adhesion and soluble agonist receptors in regulating the dynamics of platelet aggregation; and (d) the identification of surface ligands and receptors involved in stabilizing and sustaining formed aggregates. In the remainder of this review, I focus on recent developments in each of these areas. Involvement of multiple adhesion receptors and ligands in platelet aggregation One of the most dramatic conceptual changes in the understanding of platelet aggregation has been the demonstration that multiple adhesive ligands, such as VWF, fibrinogen, and fibronectin, regulate platelet-platelet interactions, with each ligand having distinct roles in the thrombotic process (Figure 2). In vitro perfusion studies on blood obtained from individuals with afibrinogenemia or type III von Willebrand’s disease,12,13,27,28,33,34 as well as in vivo studies on mice with a targeted deletion of VWF or fibrinogen,31,35 have demonstrated that VWF plays a major role in initiating aggregation under high shear with fibrinogen (and fibrin) playing a secondary role in stabilizing formed aggregates. Strikingly, mice lacking both VWF and fibrinogen still retained the capacity to form aggregates in vivo35 through an adhesion process dependent on plasma fibronectin36 and possibly other ligands. Why multiple integrin ␣IIb3 ligands are required for platelet aggregation and thrombus growth remains unclear. From www.bloodjournal.org by guest on September 7, 2015. For personal use only. BLOOD, 15 JUNE 2007 䡠 VOLUME 109, NUMBER 12 PLATELET AGGREGATION 5089 Figure 2. Involvement of multiple adhesion receptorligand interactions in platelet aggregation under high shear flow. Under conditions of rapid blood flow (typically wall shear rates ⬎ 1000 s⫺1), the initial tethering of platelets to the surface of immobilized platelets involves VWF-GPIb interaction. This adhesive interaction is rapidly reversible and at shear rates up to 10 000 s⫺1 does not readily support stable platelet-platelet adhesion, resulting in platelet movement (translocation) across the thrombus surface. Platelet stimulation by one or more soluble agonists during translocation promotes the binding of VWF and fibronectin to integrin ␣IIb3, leading to sustained platelet-platelet adhesion. At elevated shear rates, the principal role of the fibrin(ogen)-integrin ␣IIb3 interaction is to stabilize formed aggregates. Fibrinogen The importance of the fibrinogen-integrin ␣IIb3 interaction in supporting platelet aggregation has been reviewed in detail elsewhere.37 Fibrinogen, VWF, and fibronectin all have similar binding affinities to the activated form of integrin ␣IIb3; under low shear conditions, however, fibrinogen is thought to be the dominant ligand supporting platelet aggregation, principally attributable to its high molar ratio in plasma relative to other integrin ␣IIb3 ligands.38 Furthermore, with increasing shear (⬎ 1000 s⫺1), the initiation of aggregation becomes progressively more dependent on VWF and fibronectin, with in vivo studies of fibrinogen-deficient mice demonstrating normal or even increased rates of initial thrombus growth.35 These thrombi are invariably unstable, and studies on mice expressing mutants forms of fibrinogen that have a selective defect in their ability to engage integrin ␣IIb3 have confirmed that part of the thrombus-stabilizing effects of fibrinogen are linked to its capacity to bridge integrin ␣IIb3 receptors on the surface of adjacent platelets,39 whereas fibrin formation anchors the thrombus to the vessel wall.35 von Willebrand factor The importance of VWF in initiating platelet-platelet adhesion contacts under conditions of rapid blood flow is well established,40 with evidence that it participates over a much broader (and lower) shear range than previously anticipated.12,28,33,41 There are a range of factors that lead to the preferential binding of platelets to VWF under flow, including (a) VWF’s very large multimeric structure, which provides an array of binding sites for platelet receptors; (b) VWF’s efficiency at capturing platelets from bulk flow (through engagement of GPIb), thereby increasing its spatial proximity to integrins30; (c) potential shear-dependent changes in VWF’s globular conformation that increases the availability of receptor binding sites; and (d) the ability of the VWF-GPIb interaction to transmit signals that initiate integrin ␣IIb3 activation, thereby increasing the affinity of integrin ␣IIb3-VWF bonds.42,43 These factors, combined with the fact that VWF-GPIb bonds are positively modulated by shear, provides a mechanistic explanation for the dominant role of VWF in promoting platelet aggregation with progressive increases in blood flow. The importance of VWF multimer size in regulating platelet thrombus formation has recently been highlighted through the study of mice lacking the VWF cleaving metalloproteinase ADAMTS13 whose deficiency predisposes to thrombotic thrombocytopenic purpura.44 Mice lacking ADAMTS13 have a much faster rate of thrombus formation leading to premature vasculature occlusion. Furthermore, infusion of recombinant ADAMTS13 promotes thrombus dissolution in injured mouse arterioles, raising the possibility that cleavage of VWF multimers may represent an effective antithrombotic approach. Fibronectin For many years, fibronectin was thought to play a minor role in platelet aggregation. For example, deficiency of plasma fibronectin in mice is not associated with a defect in platelet aggregation using standard aggregometer assays, nor is it associated with a prolonged bleeding time.45 The demonstration more than 20 years ago that depleting fibronectin from plasma reduced thrombus formation under flow raised the possibility for its involvement in sheardependent platelet aggregation.29 Recent in vivo studies have supported these findings,35,36 with Wagner et al demonstrating persistent thrombus formation in arterioles of mice lacking VWF and fibrinogen, suggesting the involvement of a third adhesive ligand in platelet aggregation.35 Follow-up studies on mice expressing low levels of plasma and platelet fibronectin have confirmed an important role for fibronectin in promoting platelet aggregation and From www.bloodjournal.org by guest on September 7, 2015. For personal use only. 5090 JACKSON thrombus growth.36,46 Interestingly, all stages of thrombus development appear to be affected by fibronectin deficiency, including thrombus initiation, growth, and stability. Fibronectin influences both platelet adhesion and aggregation as crosslinking fibronectin with polymerized fibrin in combination with added plasma fibronectin, synergistically enhanced platelet adhesion, aggregation, and thrombus growth.47 Fibronectin’s role in shear-dependent platelet aggregation is incompletely understood and is likely to involve multiple factors. Fibronectin is known to bind other proteins involved in aggregation, such as fibrinogen and thrombospondin, a process that may increase the stability of platelet-platelet interactions.48 As with VWF, fibronectin has been demonstrated to undergo conformational changes in response to mechanical stress,49 a finding that may partially explain the preferential role for fibronectin in promoting thrombus formation under high shear. Fibronectin has also been noted to exhibit a distinct pattern of matrix assembly on the surface of platelets50 and have distinct binding characteristics from fibrinogen to the activated form of integrin ␣IIb3.50,51 How these distinct binding interactions with platelets facilitate platelet aggregation remains unknown. Although considerable progress has been made in recent years in defining the relative roles of VWF, fibrin(ogen), and fibronectin in promoting shear-dependent platelet aggregation, many important issues remain, not least, the mechanisms controlling deposition of these proteins on the platelet surface. It also remains to be determined what the relative contributions of platelet-derived and plasma VWF, fibrinogen, and fibronectin are in platelet aggregation. Confocal imaging of fibrinogen and VWF distribution during thrombus development reveal distinct spatial localization of the different protein pools with evidence that platelet stores of fibrinogen are important for consolidating the thrombus core.52 It is also unclear why a combination of ligands (including VWF/ fibrinogen or fibronectin/fibrin) are far more reactive to platelets than in isolation, a finding that may have direct clinical relevance because elevated plasma levels of VWF, fibrinogen, and possibly fibronectin independently increase the risk of atherothrombotic complications. Unraveling the complex dynamics of platelet aggregation One of the most striking findings from intravital studies of thrombosis is the dynamic nature of platelet aggregation in vivo. A characteristic feature of initial thrombus formation is the high proportion of platelets that translocate (movement from the point of initial attachment) on the injured vessel wall and on the surface of thrombi before forming firm adhesion contacts.12,39 This adhesive behavior is similar to the rolling interactions of leukocytes with postcapillary endothelial cells at sites of inflammation, where initial tethering and rolling is mediated by the P-selectin-PSGL-1 interaction and firm adhesion is mediated by leukocyte 2integrins.53 The initial tethering of platelets is principally mediated by the VWF-GPIb interaction, whereas firm adhesion depends on ligand engagement of 1 and 3-integrins.19 Platelet translocation is typically stop-start in nature and, depending on the type and extent of vascular injury, a considerable proportion of tethering platelets will only interact transiently with the thrombus surface.12 Defining the factors regulating the stop-start nature of platelet translocation is a critical issue, because it is one of the key variables regulating the rate and extent of thrombus growth. BLOOD, 15 JUNE 2007 䡠 VOLUME 109, NUMBER 12 Platelet shape and translocation dynamics An important factor influencing cell rolling behavior is morphology.53 In the case of leukocytes, their round morphology is well suited to rotational motion (rolling); the flat discoid morphology of platelets, however, is less ideal for smooth rolling interactions. Platelets have been demonstrated to assume different morphologies in vitro after prolonged exposure to adhesive surfaces such as VWF,54-56 raising the possibility that signals generated during surface translocation may induce morphological changes that facilitate a rolling phenotype. It has also been demonstrated that platelets can translocate as flat discs through a rotational side-toside flipping mechanism57 or through a sliding, rotational movement of the cell body.58 Recent high-resolution imaging of platelets in vivo have revealed that the majority of platelets translocate on the vessel wall and on the surface of thrombi in vivo as flat, sliding discs.59 Most platelets slide in a stop-start manner, although rotational flipping of discoid platelets can also be observed. Relative to rotating discs or rolling spheres, sliding platelets experience reduced drag force and have much lower tensile stresses on adhesive bonds. A flat disc also enables maximal surface contact area with the adhesive surface, increasing the potential for multivalent adhesive interactions. Thus, by adopting a sliding, adhesive behavior, platelets appear to have evolved a unique translocation mechanism that minimizes drag forces on adhesive bonds. Combined with their small dimensions, this translocation mechanism provides an efficient means of limiting the detaching effect of blood flow on translocating platelets. As discussed subsequently, this sliding mechanism appears to be greatly facilitated by the development of small membrane protrusions (tethers), which further reduce tensile stress on adhesive bonds. Importance of membrane tethers in the initiation of platelet aggregation A significant recent development in the study of platelet adhesive interactions under flow has been the finding that membrane tethers play an important role in the initiation of platelet-matrix and platelet-platelet adhesive interactions56,58-60 (Figure 3A). Membrane tethers are smooth cylinders of lipid bilayer that are pulled from the surface of platelets under the influence of hemodynamic drag force. These structures are dynamic and extend from localized adhesion contacts and appear to play a role in regulating platelet translocation behavior.60 Similar structures have been demonstrated in a variety of cell types, including red blood cells,61 neutrophils,62 neurons,63 fibroblasts,64 and endothelial cells,65 and similar to platelets, membrane tethers are thought to play an important role in regulating leukocyte rolling behavior.62 One of the key features of membrane tethers is their ability to reduce the pulling forces imposed on adhesive bonds66 and, as a consequence, increase the probability of sustaining adhesion in a shear field. Strikingly, tethers of up to 30 m have been observed extending from the surface of platelets,58,60 a massive extension of membrane from a cell that is only 1 to 3 m in diameter. Because surface membranes can only be normally stretched by approximately 4% before causing cell lysis,67 the creation of such tethers must involve utilization of membrane reserves from the surfaceconnecting canalicular system. These membrane reserves can also buffer changes in surface membrane tension, helping to dampen the platelet activating effects of rapid fluctuations in blood flow.64 From www.bloodjournal.org by guest on September 7, 2015. For personal use only. BLOOD, 15 JUNE 2007 䡠 VOLUME 109, NUMBER 12 PLATELET AGGREGATION 5091 Role of adhesion receptor signals and soluble agonists in coordinately regulating platelet aggregation dynamics under flow Figure 3. Membrane tethers regulate platelet-matrix and platelet-platelet adhesive interactions. (A) (Left) Scanning electron microscopy of a single discoid platelet forming membrane tethers during adhesion to immobilized VWF (1800 s⫺1). Note that tethers can be readily distinguished from filopodia in that the latter is inhibited by pretreating platelets with the actin polymerization inhibitor, cytochalasin D. (Right) Shear-dependent formation of adhesion contacts between discoid platelets involves the development of membrane tethers (arrows). (B) (Left) Reversible aggregation of discoid platelets. Note that in this image, all platelets cluster around a central activated platelet through the development of membrane tethers. (Right) The development of stable platelet aggregates is associated with classic platelet shape change characterized by sphering of the platelet body and the extension of multiple filopodial projections. (Modified from Brass et al68; used with permission.) (This research was originally published in Maxwell et al.59 and was adapted with permission. Platelets were imaged on a Hitachi S570 scanning electron microscope [Tokyo, Japan] at 20 kV accelerating voltage, 3 mm working distance.) Role of membrane tethers in a 2-stage platelet aggregation process What is the role of membrane tethers in promoting the formation of stable platelet aggregates? Recent high-resolution imaging of platelets during thrombus development has provided new insights into this process59 (Figure 3B). These studies have revealed the existence of a 2-phase platelet aggregation process, an initial reversible phase of aggregation occurring between discoid platelets and a subsequent stable aggregation phase associated with platelet shape change and granule release. The formation of discoid platelet aggregates is shear-dependent and primarily mediated through the development of membrane tethers. This initial phase of aggregation involves platelet activation and the adhesive function of both GPIb and integrin ␣IIb3; conspicuously, however, it does not require soluble agonists such as adenosine diphosphate (ADP), TXA2, or thrombin. Nonetheless, these aggregates are always reversible with conversion to stable aggregates dependent on soluble agonist generation, most notably ADP. Thus, tethers appear to play an important role in maintaining close physical proximity between platelets, which may provide a mechanism of facilitating autocrine/ paracrine stimulation by locally generated agonists. Such a mechanism may help limit the “washout” effects of blood flow on released soluble agonists, providing a means of maintaining a high local concentration of activating signals within the confines of a developing aggregate.68 One of the key features of membrane tethers is that they provide a mechanism of sustaining platelet interactions with the thrombus surface without the requirement for global platelet activation. One of the more vexing issues with respect to platelet aggregation is the significance of GPIb and integrin ␣IIb3-derived activating signals. In general, there is limited insight into the spatiotemporalsignaling relationship operating between adhesion receptors and soluble agonists in regulating platelet or leukocyte activation under flow. For example, leukocyte rolling velocity is critically influenced by the level of cell stimulation by specific chemokines69; these input signals, however, do not occur in isolation and are likely to be modified (and amplified) by signals emanating from the adhesion receptors themselves.70 Based on perfusion studies on immobilized VWF, GPIb and integrin ␣IIb3 appear to elicit distinct, cooperative activating signals71,72 that can regulate platelet translocation dynamics partially through the reversible activation of integrin ␣IIb3.71 In general, GPIb and integrin ␣IIb3 signals are inefficient at inducing global platelet activation in the absence of a costimulus such as ADP, thrombin, TXA2, or collagen.73 Most current models of thrombus development propose a key role for collagen (and possibly vessel wall-derived thrombin) in initiating platelet activation in primary adherent platelets, whereas subsequent propagation of thrombi (platelet aggregation) is primarily driven by agonists released or generated from the platelet surface, including ADP, TXA2, and thrombin. Soluble agonists do not recruit platelets into forming thrombi, however, adhesion receptors do, which begs the question whether activating signals downstream of GPIb and integrin ␣IIb3 are important to promote initial platelet activation and thrombus development. The answer to this, at least in the context of initial thrombus growth, remains uncertain and controversial.73-76 There is currently limited evidence that signals downstream of GPIb are essential for the initiation of hemostatic plug formation or thrombus development. Similarly, the role of integrin ␣IIb3-derived signals in initiating platelet aggregation and thrombus development remains unclear. It is possible that activating signals downstream of GPIb play an important role in initiating localized integrin ␣IIb3 activation necessary for membrane tether formation and reversible aggregation. Similarly, there is evidence that integrin ␣IIb3-derived signals, in combination with soluble agonists, synergistically enhance platelet activation under flow.73,77 In isolation, GPIb and integrin ␣IIb3 signals are weak; nonetheless, it is conceivable that localized signals from these receptors may play an important role in facilitating the initial formation of reversible aggregates and, in combination with soluble agonists, Figure 4. Factors stabilizing formed platelet aggregates. The intercellular space between aggregating platelets facilitates interaction between integrins and their ligands and other adhesion molecules, and promotes activation of Eph receptor kinases by cell surface ephrins. This narrow space also provides a protective environment for the accumulation of soluble agonists (ADP, thrombin, and TXA2), Gas-6, and the proteolytically shed exodomains of platelet surface proteins (GPIb, P-selectin, sCD40L). From www.bloodjournal.org by guest on September 7, 2015. For personal use only. 5092 JACKSON BLOOD, 15 JUNE 2007 䡠 VOLUME 109, NUMBER 12 Figure 5. Distinct mechanisms initiating platelet aggregation at various shear rates. (A) Platelet aggregation at shear rates ⬍ 1000 s⫺1 is predominantly mediated by the interaction of fibrinogen with integrin ␣IIb3. Under these conditions, stable aggregation typically occurs between shape-changed platelets. (B) At shear rates between 1000 and 10 000 s⫺1, a distinct 2-stage aggregation process can be identified. The initial formation of aggregates occurs between discoid platelets, is mediated by membrane tethers, and is dependent on the adhesive function of both GPIb and integrin ␣IIb3. Conversion of reversible aggregates to stable aggregates is associated with platelet shape change and is dependent on the generation of soluble agonists, particularly ADP. (C) At shear rates ⬎ 10 000 s⫺1, platelet aggregation can be initiated independent of integrin ␣IIb3 or platelet activation and is exclusively mediated by the VWF-GPIb interaction. Although the initial aggregates form between discoid platelets, at such high shears, the platelets adopt a smooth spherical morphology and roll (translocate in a rotational manner) across the VWF surface (unpublished observations, Erik Westein and Shaun P. Jackson). (Figure was adapted with permission from Maxwell et al.59) enhance the incorporation of these aggregates into stable thrombi. Definitive resolution of this issue will require the development of mouse models expressing mutant forms of GPIb and integrin ␣IIb3 that have preserved adhesive function but a selective signaling defect. Sustaining platelet aggregation and thrombus stability Platelets must not only be able to develop adhesive contacts under rapid blood flow, but must also sustain them to prevent thrombus detachment (embolization) from the site of vascular injury. Recent studies have revealed that a complex repertoire of adhesion and signaling receptors are present on the platelet surface that may serve to regulate the stability of forming aggregates (Figure 4). The details of this system have recently been reviewed68 and only the core elements are briefly presented here. Central to thrombus stability is the maintenance of high-affinity/avidity integrin ␣IIb3 adhesion bonds. Initiation of integrin ␣IIb3 activation is controlled by well-characterized signaling events operating downstream of soluble agonist (Gq and G12/13) and adhesion (nonreceptor tyrosine kinase) coupled receptors.9 Sustaining integrin ␣IIb3 activation is critically dependent on signals operating downstream of Gicoupled receptors, principally the purinergic P2Y12 receptor.78 Thus, ADP plays a key role in both initiating (through the Gq-linked P2Y1 receptor) and sustaining integrin ␣IIb3 activation necessary for the development of stable platelet-platelet adhesion contacts. There is growing evidence that the P2Y12 signals do not operate in isolation. For example, once engaged by ligand, integrin ␣IIb3 initiates the formation of membrane-proximal signaling complexes that not only serve to sustain platelet activation, but also induce cytoskeletal changes that promote integrin ␣IIb3 clustering and increased receptor avidity.79 Recent evidence suggests that members of the tetraspanin family, CD15169 and TSSC6,80 play an important role in regulating integrin ␣IIb3 outside-in signaling, with a deficiency in TSSC6 leading to a defect in thrombus stability.80 The development of close platelet-platelet contacts also enables the juxtaposition of ligands on one platelet with receptors on adjacent platelets. Examples of this include various members of the immunoglobulin superfamily (PECAM-1,81 JAM-A,82 JAMC,83 ESAM ,84 and CD22685), Eph kinases/ephrins,86 and Gas6 and its receptors, Axl-Tyro3-Mer.87 The role of these individual components in regulating the stability of platelet aggregates is only beginning to be addressed, although there is evidence that Eph kinases/ephrins88 and Gas687 and its receptors play an important role in this process. The exodomains of various platelet surface proteins, including P-selectin,89 CD40L,90 GPIb,91 GPV,92 GPVI,93 and Sema4D,68 are also shed from the surface of platelets with evidence that the soluble form of CD40L promotes thrombus stability by engaging integrin ␣IIb3.94 Multiple adhesion mechanisms initiating platelet aggregation Based on findings from in vitro perfusion systems12,28,33,34,95 and in vivo thrombosis models,35,36,39,59 at least 3 distinct mechanisms of platelet aggregation can be identified with the relative contribution of each mechanism dependent on the prevailing shear conditions. As demonstrated in Figure 5, the key components of the traditional model of platelet aggregation, in which aggregation is mediated exclusively by fibrinogen and integrin ␣IIb3, is likely to be the predominant mechanism operating under relatively low shear conditions (⬍ 1000 s⫺1). From www.bloodjournal.org by guest on September 7, 2015. For personal use only. BLOOD, 15 JUNE 2007 䡠 VOLUME 109, NUMBER 12 Under such conditions, integrin ␣IIb3 on the surface of free-flowing platelets can engage fibrinogen adsorbed onto the surface of thrombi. Note that this adhesive interaction can occur independent of VWFGPIb, although even at these shear rates, the latter adhesive interaction may have a role.41 Subsequent stimulation of platelets by locally generated soluble agonists induces platelet shape change and an increase in integrin ␣IIb3 affinity, which helps stabilize/sustain integrin ␣IIb3fibrinogen bonds. At shear rates between 1000 and 10 000 s⫺1, the processes initiating aggregation are quite distinct involving additional receptors, ligands, and membrane tethers. At these shear rates, plateletplatelet interactions become progressively more VWF-dependent with an important role for both GPIb and integrin ␣IIb3 in promoting the initial formation of discoid platelet aggregates. Whether fibronectin is also involved in these earliest stages remains to be seen; nonetheless, at these shear rates, there are essential contributions of VWF, fibrin(ogen), and fibronectin to the formation of stable platelet aggregates. Recently, a third mechanism initiating platelet aggregation has been identified that is operative at very high shear rates (⬎ 10 000 s⫺1).95 Strikingly, this aggregation mechanism does not require platelet activation or the adhesive function of integrin ␣IIb3 and is exclusively mediated by VWF-GPIb adhesive bonds. Aggregate formation is dependent on both immobilized and soluble forms of VWF, and at such high shear, marked deformation to the membrane of nucleating platelets (the nidus for aggregate formation) is a hallmark feature of the aggregation process. How important these membrane changes are to the formation/ maintenance of adhesive bonds remains to be established; nonetheless, the findings that nonactivated platelets can form large aggregates under very high shear have potentially important implications for the mechanisms promoting occlusive thrombus formation in stenosed arteries. It is important to recognize that none of these models are identical to the traditional model of aggregation developed from studies in the aggregometer. In the latter situation, regardless of the physiological agonist, the platelet response is essentially the same; platelets undergo shape change, become “sticky,” form aggregates, and release granule contents, a temporal sequence of events distinct from those occurring under flow. Translating basic insights into clinical benefit The identification of discrete shear-specific mechanisms initiating platelet aggregation, coupled with a clearer understanding of the key elements propagating and sustaining thrombi, raises the possibility that antiplatelet therapies may be developed that preferentially operate in certain vascular territories and at distinct stages of the thrombotic process. Evidence supporting such a possibility has been derived from in vivo studies targeting specific platelet adhesion events. For example, deficiency of plasma fibronectin produces a platelet aggregation defect that is distinct from that observed with VWF or fibrinogen deficiency35,36 without producing a marked prolongation in bleeding time. Given the preferential role for fibronectin in promoting thrombus growth and stability under elevated shear,46 selective blockade of the fibronectin-integrin ␣IIb3 interaction may be an attractive approach to prevent thrombotic vascular occlusion while maintaining mural thrombus formation. Similarly, blockade of the integrin ␣IIb3-binding sequence of fibrinogen (C-terminal ␥-chain dodecapeptide sequence) also produces a marked defect in thrombus formation,39,96 raising the possibility that selective inhibition of specific integrin ␣IIb3 ligands, rather than global inhibition of integrin ␣IIb3 itself, may represent a more targeted antithrombotic approach. Furthermore, given the well-defined role for the GPIb-VWF interaction in supporting platelet adhesive interactions under high shear, inhibi- PLATELET AGGREGATION 5093 tion of this adhesive event has been considered an attractive approach to limit thrombus formation in high shear flow situations. Inhibitors of the VWF-GPIb interaction are highly effective in preventing thrombotic vascular occlusion,97 although it remains to be established whether such approaches will ultimately have an improved therapeutic window relative to conventional integrin ␣IIb3 antagonists.8 Similar concepts are beginning to emerge in the context of soluble agonists. For example, there is a considerable body of evidence supporting a major role for ADP in not only potentiating platelet aggregation, but also in sustaining formed aggregates necessary for the development of stable thrombi. Genetic deletion or pharmacological blockade of the P2Y12 receptor leads to a major defect in thrombus growth and stability regardless of the primary thrombogenic stimulus.98 These amplifying effects of ADP are likely to be important for thrombus development/stability in most areas of the circulation, producing a significant hemostatic defect at high levels of receptor blockade. Several signaling components acting downstream of the P2Y12 receptor appear to also play an important role in thrombus development/stability but are less critical for hemostasis. These include 2 members of the PI 3-kinase family, p110 and p110␥, which play an important role in sustaining integrin ␣IIb3 activation and stable platelet aggregation.99,100 Other platelet components that have been demonstrated to be important for thrombus stability, but less important for initial thrombus growth, include sCD40L94 and Gas687 and its receptors. Whether other agonist receptors and their downstream signaling components regulate platelet aggregation under specific rheological conditions and at discrete stages in the thrombotic process remains to be seen. It should be acknowledged that a great deal of the recent progress in the understanding of platelet aggregation has been based on experimental animal models, particularly the mouse, which inevitably raises issues of relevance to humans. In this context, none of the commonly used in vivo thrombosis models (involving ferric chloride, photochemical or laser injury of healthy vessels) accurately reproduce the conditions of arterial thrombosis in humans, in which thrombi typically form on ruptured or eroded atherosclerotic plaques. Moreover, the rheological conditions in the mouse microcirculation used for intravital studies are likely to be significantly different from those operating in stenosed human arteries. It also remains unclear whether observations from in vivo thrombosis models can be extrapolated to platelet aggregation processes relevant to hemostatic platelet plug formation. However, as with any experimental model, the biological plausibility of findings can only be established when carefully analyzed in the context of other experimental data and clinical observations. Thus, in the same way that findings in the aggregometer helped validate the importance of various adhesive proteins, soluble agonists/ receptors, and signaling cascades in hemostasis, insights from ex vivo perfusion studies on human platelets, combined with data from carefully performed clinical studies, will be required to more precisely validate the role of the growing list of “new players” in the platelet aggregation/thrombosis process. Conclusions Great progress has been made over the last decade in unraveling the complex and dynamic processes regulating platelet aggregation, particularly in the context of arterial thrombosis. Although most of the key elements have probably been identified, some unexpected From www.bloodjournal.org by guest on September 7, 2015. For personal use only. 5094 BLOOD, 15 JUNE 2007 䡠 VOLUME 109, NUMBER 12 JACKSON findings continue to surface that challenge many long-held beliefs in the field, not least the recent demonstration that under certain experimental conditions, platelet aggregation can occur independent of platelet activation and integrin ␣IIb3 engagement.95 These new insights, although currently of primary interest to the academic community, are likely to be rapidly translated into new experimental approaches that may ultimately hold great promise in the clinic. What was once considered a straightforward phenomenon, platelet aggregation has evolved into a complex field of investigation with many of its most jealously guarded secrets only now beginning to emerge. Acknowledgments I thank Drs Denisa Wagner and Zaverio Ruggeri, members of my laboratory and the Department of Haematology, Alfred Hospital, for constructive advice on the review. The assistance with preparation of figures by Dr. Simone Schoenwaelder is also gratefully acknowledged. This work was supported by grants from the National Health and Medical Research Council of Australia, the Australian Research Council, and the National Heart Foundation of Australia. Authorship Conflict-of-interest disclosure: The author declares no competing financial interests. Correspondence: Shaun P. Jackson, Australian Centre for Blood Diseases, Monash University, 6th Floor, Burnet Tower, 89 Commercial Rd., Melbourne, Australia, 3004; e-mail: shaun.jackson@med. monash.edu.au. References 1. Bizzozero J. On a new blood particle and its role in thrombosis and blood coagulation. Virchows Arch Pathol Anat Physiol Klin Med. 1882;90:261332. 2. Eberth J, Schimmelbusch C. Experimentelle Untersuchungen uber Thrombose. Die Anfange der Thrombenbildung. Fortschr Med. 1885;3:379389. 3. Hayem G. Recherches sur l’evolution des hematies dans le sang de l’homme et des vertebres. Arch Physiol Norm Pathol II. 1878;5:692-734. 4. Zahn F. Untersuchungen uber thrombose. Centrralbl Med Wissensch. 1872;10:129-130. 5. Bizzozero J. Bizzozero’s new corpuscle. Lancet. 1882;1:446. 6. Osler W. On certain problems in the physiology of the blood corpuscles. The Medical News. 1886; 48:421-425. 7. Bhatt DL, Topol EJ. Scientific and therapeutic advances in antiplatelet therapy. Nat Rev Drug Discov. 2003;2:15-28. 8. Jackson SP, Schoenwaelder SM. Antiplatelet therapy: in search of the ’magic bullet.’ Nat Rev Drug Discov. 2003;2:775-789. 9. Ruggeri ZM. Platelets in atherothrombosis. Nat Med. 2002;8:1227-1234. 10. Goto S, Ikeda Y, Saldivar E, Ruggeri ZM. Distinct mechanisms of platelet aggregation as a consequence of different shearing flow conditions. J Clin Invest. 1998;101:479-486. 11. Frojmovic MM. Platelet aggregation in flow: differential roles for adhesive receptors and ligands. Am Heart J. 1998;135:S119–131. 12. Kulkarni S, Dopheide SM, Yap CL, et al. A revised model of platelet aggregation. J Clin Invest. 2000; 105:783-791. 13. Ruggeri ZM, Dent JA, Saldivar E. Contribution of distinct adhesive interactions to platelet aggregation in flowing blood. Blood. 1999;94:172-178. 14. Bizzozero G. Su di un nuovo elemento morfologico del sangue dei mammiferi e della sua importanza nella trombosi e nella coagulazione. L’Osservatore. 1881;17:785-787. 15. Glanzmann E. Hereditare Hamorrhagische Thrombasthenie. Ein Beitrag zur pathologie der Blutplattchen. Jahrb Kinderheilk. 1918;88:142,113-141. 16. Born GV. Aggregation of blood platelets by adenosine diphosphate and its reversal. Nature. 1962; 194:927-929. 17. O’Brien J R. Platelet aggregation. II. Some results from a new method of study. J Clin Pathol. 1962;15:452-481. 18. Bluestein D, Niu L, Schoephoerster RT, Dewan- jee MK. Fluid mechanics of arterial stenosis: relationship to the development of mural thrombus. Ann Biomed Eng. 1997;25:344-356. 19. Ruggeri ZM. von Willebrand factor. J Clin Invest. 1997;99:559-564. 20. Stemerman MB, Baumgartner HR, Spaet TH. The subendothelial microfibril and platelet adhesion. Lab Invest. 1971;24:179-186. 21. Baumgartner HR, Haudenschild C. Adhesion of platelets to subendothelium. Ann N Y Acad Sci. 1972;201:22-36. 22. Tschopp TB, Weiss HJ, Baumgartner HR. Decreased adhesion of platelets to subendothelium in von Willebrand’s disease. J Lab Clin Med. 1974;83:296-300. 23. Weiss HJ, Turitto VT, Baumgartner HR. Effect of shear rate on platelet interaction with subendothelium in citrated and native blood. I. Shear ratedependent decrease of adhesion in von Willebrand’s disease and the Bernard-Soulier syndrome. J Lab Clin Med. 1978;92:750-764. 24. Baumgartner HR. Platelet interaction with collagen fibrils in flowing blood. I. Reaction of human platelets with alpha chymotrypsin-digested subendothelium. Thromb Haemost. 1977;37:1-16. 25. Savage B, Almus-Jacobs F, Ruggeri ZM. Specific synergy of multiple substrate-receptor interactions in platelet thrombus formation under flow. Cell. 1998;94:657-666. in hemostasis and thrombosis. Proc Natl Acad Sci USA. 1998;95:9524-9529. 32. Falati S, Gross P, Merrill-Skoloff G, Furie BC, Furie B. Real-time in vivo imaging of platelets, tissue factor and fibrin during arterial thrombus formation in the mouse. Nat Med. 2002;8:1175-1181. 33. Ikeda Y, Handa M, Kawano K, et al. The role of von Willebrand factor and fibrinogen in platelet aggregation under varying shear stress. J Clin Invest. 1991;87:1234-1240. 34. Tsuji S, Sugimoto M, Miyata S, Kuwahara M, Kinoshita S, Yoshioka A. Real-time analysis of mural thrombus formation in various platelet aggregation disorders: distinct shear-dependent roles of platelet receptors and adhesive proteins under flow. Blood. 1999;94:968-975. 35. Ni H, Denis CV, Subbarao S, et al. Persistence of platelet thrombus formation in arterioles of mice lacking both von Willebrand factor and fibrinogen. J Clin Invest. 2000;106:385-392. 36. Ni H, Yuen PS, Papalia JM, et al. Plasma fibronectin promotes thrombus growth and stability in injured arterioles. Proc Natl Acad Sci USA. 2003;100:2415-2419. 37. Marguerie G, Ginsberg MH, Plow EF. Glycoproteins: the fibrinogen receptor. In: Holmsen H, ed. Platelet Responses and Metabolism. Volume III— Response-Metabolism Relationships. Boca Raton, Florida: CRC Press Inc; 1986:286-294. 26. Nieswandt B, Brakebusch C, Bergmeier W, et al. Glycoprotein VI but not alpha2beta1 integrin is essential for platelet interaction with collagen. Embo J. 2001;20:2120-2130. 38. Gralnick HR, Williams SB, Coller BS. Fibrinogen competes with von Willebrand factor for binding to the glycoprotein IIb/IIIa complex when platelets are stimulated with thrombin. Blood. 1984;64: 797-800. 27. Turitto VT, Weiss HJ, Baumgartner HR. Platelet interaction with rabbit subendothelium in von Willebrand’s disease: altered thrombus formation distinct from defective platelet adhesion. J Clin Invest. 1984;74:1730-1741. 39. Ni H, Papalia JM, Degen JL, Wagner DD. Control of thrombus embolization and fibronectin internalization by integrin alpha IIb beta 3 engagement of the fibrinogen gamma chain. Blood. 2003;102: 3609-3614. 28. Weiss HJ, Hawiger J, Ruggeri ZM, Turitto VT, Thiagarajan P, Hoffmann T. Fibrinogen-independent platelet adhesion and thrombus formation on subendothelium mediated by glycoprotein IIb-IIIa complex at high shear rate. J Clin Invest. 1989; 83:288-297. 40. Ruggeri ZM. Old concepts and new developments in the study of platelet aggregation. J Clin Invest. 2000;105:699-701. 29. Bastida E, Escolar G, Ordinas A, Sixma JJ. Fibronectin is required for platelet adhesion and for thrombus formation on subendothelium and collagen surfaces. Blood. 1987;70:1437-1442. 41. Chauhan AK, Kisucka J, Lamb CB, Bergmeier W, Wagner DD. von Willebrand factor and factor VIII are independently required to form stable occlusive thrombi in injured veins. Blood. 2006;109: 2424-2429. 30. Savage B, Saldivar E, Ruggeri ZM. Initiation of platelet adhesion by arrest onto fibrinogen or translocation on von Willebrand factor. Cell. 1996; 84:289-297. 42. De Marco L, Girolami A, Russell S, Ruggeri ZM. Interaction of asialo von Willebrand factor with glycoprotein Ib induces fibrinogen binding to the glycoprotein IIb/IIIa complex and mediates platelet aggregation. J Clin Invest. 1985;75:11981203. 31. Denis C, Methia N, Frenette PS, et al. A mouse model of severe von Willebrand disease: defects 43. Kasirer-Friede A, Cozzi MR, Mazzucato M, De Marco L, Ruggeri ZM, Shattil SJ. Signaling From www.bloodjournal.org by guest on September 7, 2015. For personal use only. BLOOD, 15 JUNE 2007 䡠 VOLUME 109, NUMBER 12 through GP Ib-IX-V activates alpha IIb beta 3 independently of other receptors. Blood. 2004;103: 3403-3411. 44. Motto DG, Chauhan AK, Zhu G, et al. Shigatoxin triggers thrombotic thrombocytopenic purpura in genetically susceptible ADAMTS13-deficient mice. J Clin Invest. 2005;115:2752-2761. 45. Sakai T, Johnson KJ, Murozono M, et al. Plasma fibronectin supports neuronal survival and reduces brain injury following transient focal cerebral ischemia but is not essential for skin-wound healing and hemostasis. Nat Med. 2001;7:324330. 46. Matuskova J, Chauhan AK, Cambien B, et al. Decreased plasma fibronectin leads to delayed thrombus growth in injured arterioles. Arterioscler Thromb Vasc Biol. 2006;26:1391-1396. 47. Cho J, Mosher DF. Enhancement of thrombogenesis by plasma fibronectin cross-linked to fibrin and assembled in platelet thrombi. Blood. 2006; 107:3555-3563. 48. Cho J, Mosher DF. Role of fibronectin assembly in platelet thrombus formation. J Thromb Haemost. 2006;4:1461-1469. 49. Hynes RO. The dynamic dialogue between cells and matrices: implications of fibronectin’s elasticity. Proc Natl Acad Sci USA. 1999;96:2588-2590. 50. Olorundare OE, Peyruchaud O, Albrecht RM, Mosher DF. Assembly of a fibronectin matrix by adherent platelets stimulated by lysophosphatidic acid and other agonists. Blood. 2001;98:117-124. 51. Plow EF, Ginsberg MH. Specific and saturable binding of plasma fibronectin to thrombin-stimulated human platelets. J Biol Chem. 1981;256: 9477-9482. 52. Matsui H, Sugimoto M, Mizuno T, et al. Distinct and concerted functions of von Willebrand factor and fibrinogen in mural thrombus growth under high shear flow. Blood. 2002;100:3604-3610. 53. Lawrence MB, Springer TA. Leukocytes roll on a selectin at physiologic flow rates: distinction from and prerequisite for adhesion through integrins. Cell. 1991;65:859-873. 54. Yuan Y, Kulkarni S, Ulsemer P, et al. The von Willebrand factor-glycoprotein Ib/V/IX interaction induces actin polymerization and cytoskeletal reorganization in rolling platelets and glycoprotein Ib/V/IX-transfected cells. J Biol Chem. 1999;274: 36241-36251. 55. Kuwahara M, Sugimoto M, Tsuji S, et al. Platelet shape changes and adhesion under high shear flow. Arterioscler Thromb Vasc Biol. 2002;22:329334. 56. Maxwell MJ, Dopheide SM, Turner SJ, Jackson SP. Shear induces a unique series of morphological changes in translocating platelets: effects of morphology on translocation dynamics. Arterioscler Thromb Vasc Biol. 2006;26:663-669. 57. Mody NA, Lomakin O, Doggett TA, Diacovo TG, King MR. Mechanics of transient platelet adhesion to von Willebrand factor under flow. Biophys J. 2005;88:1432-1443. PLATELET AGGREGATION 62. Schmidtke DW, Diamond SL. Direct observation of membrane tethers formed during neutrophil attachment to platelets or P-selectin under physiological flow. J Cell Biol. 2000;149:719-730. 63. Dai J, Sheetz MP, Wan X, Morris CE. Membrane tension in swelling and shrinking molluscan neurons. J Neurosci. 1998;18:6681-6692. 5095 endothelial cells: role in inflammatory thrombosis. Thromb Haemost. 2002;88:843-850. 83. Santoso S, Sachs UJ, Kroll H, et al. The junctional adhesion molecule 3 (JAM-3) on human platelets is a counterreceptor for the leukocyte integrin Mac-1. J Exp Med. 2002;196:679-691. 64. Raucher D, Sheetz MP. Characteristics of a membrane reservoir buffering membrane tension. Biophys J. 1999;77:1992-2002. 84. Nasdala I, Wolburg-Buchholz K, Wolburg H, et al. A transmembrane tight junction protein selectively expressed on endothelial cells and platelets. J Biol Chem. 2002;277:16294-16303. 65. Girdhar G, Shao JY. Membrane tether extraction from human umbilical vein endothelial cells and its implication in leukocyte rolling. Biophys J. 2004;87:3561-3568. 85. Kojima H, Kanada H, Shimizu S, et al. CD226 mediates platelet and megakaryocytic cell adhesion to vascular endothelial cells. J Biol Chem. 2003;278:36748-36753. 66. Shao JY, Ting-Beall HP, Hochmuth RM. Static and dynamic lengths of neutrophil microvilli. Proc Natl Acad Sci USA. 1998;95:6797-6802. 86. Prevost N, Woulfe D, Tanaka T, Brass LF. Interactions between Eph kinases and ephrins provide a mechanism to support platelet aggregation once cell-to-cell contact has occurred. Proc Natl Acad Sci USA. 2002;99:9219-9224. 67. Evans EA, Skalak R. Mechanics and thermodynamics of biomembranes: part 1. CRC Crit Rev Bioeng. 1979;3:181-330. 68. Brass LF, Zhu L, Stalker TJ. Minding the gaps to promote thrombus growth and stability. J Clin Invest. 2005;115:3385-3392. 69. Laudanna C, Alon R. Right on the spot. Chemokine triggering of integrin-mediated arrest of rolling leukocytes. Thromb Haemost. 2006;95:5-11. 70. Abbal C, Lambelet M, Bertaggia D, et al. Lipid raft adhesion receptors and Syk regulate selectindependent rolling under flow conditions. Blood. 2006;108:3352-3359. 71. Nesbitt WS, Kulkarni S, Giuliano S, et al. Distinct glycoprotein Ib/V/IX and integrin alpha IIbbeta 3-dependent calcium signals cooperatively regulate platelet adhesion under flow. J Biol Chem. 2002;277:2965-2972. 72. Mazzucato M, Pradella P, Cozzi MR, De Marco L, Ruggeri ZM. Sequential cytoplasmic calcium signals in a 2-stage platelet activation process induced by the glycoprotein Ibalpha mechanoreceptor. Blood. 2002;100:2793-2800. 73. Jackson SP, Nesbitt WS, Kulkarni S. Signaling events underlying thrombus formation. J Thromb Haemost. 2003;1:1602-1612. 74. Kuwahara M, Sugimoto M, Tsuji S, Miyata S, Yoshioka A. Cytosolic calcium changes in a process of platelet adhesion and cohesion on a von Willebrand factor-coated surface under flow conditions. Blood. 1999;94:1149-1155. 75. Liu J, Fitzgerald ME, Berndt MC, Jackson CW, Gartner TK. Bruton tyrosine kinase is essential for botrocetin/VWF-induced signaling and GPIb-dependent thrombus formation in vivo. Blood. 2006; 108:2596-2603. 76. Arias-Salgado EG, Haj F, Dubois C, et al. PTP-1B is an essential positive regulator of platelet integrin signaling. J Cell Biol. 2005;170:837-845. 77. Nesbitt WS, Giuliano S, Kulkarni S, Dopheide SM, Harper IS, Jackson SP. Intercellular calcium communication regulates platelet aggregation and thrombus growth. J Cell Biol. 2003;160:11511161. 58. Reininger AJ, Heijnen HF, Schumann H, Specht HM, Schramm W, Ruggeri ZM. Mechanism of platelet adhesion to von Willebrand factor and microparticle formation under high shear stress. Blood. 2006;107:3537-3545. 78. Gachet C. ADP receptors of platelets and their inhibition. Thromb Haemost. 2001;86:222-232. 59. Maxwell MJ, Westein E, Nesbitt WS, Giuliano S, Dopheide SM, Jackson SP. Identification of a 2-stage platelet aggregation process mediating shear-dependent thrombus formation. Blood. 2007;109:566-576. 80. Goschnick MW, Lau LM, Wee JL, et al. Impaired ‘outside-in’ integrin alphaIIbbeta3 signaling and thrombus stability in TSSC6-deficient mice. Blood. 2006;108:1911-1918. 79. Shattil SJ, Newman PJ. Integrins: dynamic scaffolds for adhesion and signaling in platelets. Blood. 2004;104:1606-1615. 60. Dopheide SM, Maxwell MJ, Jackson SP. Sheardependent tether formation during platelet translocation on von Willebrand factor. Blood. 2002;99: 159-167. 81. Newman PJ, Newman DK. Signal transduction pathways mediated by PECAM-1: new roles for an old molecule in platelet and vascular cell biology. Arterioscler Thromb Vasc Biol. 2003;23:953964. 61. Evans EA, Hochmuth RM. Membrane viscoelasticity. Biophys J. 1976;16:1-11. 82. Babinska A, Kedees MH, Athar H, et al. F11-receptor (F11R/JAM) mediates platelet adhesion to 87. Angelillo-Scherrer A, de Frutos P, Aparicio C, et al. Deficiency or inhibition of Gas6 causes platelet dysfunction and protects mice against thrombosis. Nat Med. 2001;7:215-221. 88. Prevost N, Woulfe DS, Jiang H, et al. Eph kinases and ephrins support thrombus growth and stability by regulating integrin outside-in signaling in platelets. Proc Natl Acad Sci USA. 2005;102: 9820-9825. 89. Berger G, Hartwell DW, Wagner DD. P-Selectin and platelet clearance. Blood. 1998;92:44464452. 90. Henn V, Slupsky JR, Grafe M, et al. CD40 ligand on activated platelets triggers an inflammatory reaction of endothelial cells. Nature. 1998;391: 591-594. 91. Bergmeier W, Piffath CL, Cheng G, et al. Tumor necrosis factor-alpha-converting enzyme (ADAM17) mediates GPIbalpha shedding from platelets in vitro and in vivo. Circ Res. 2004;95: 677-683. 92. Rabie T, Strehl A, Ludwig A, Nieswandt B. Evidence for a role of ADAM17 (TACE) in the regulation of platelet glycoprotein V. J Biol Chem. 2005; 280:14462-14468. 93. Stephens G, Yan Y, Jandrot-Perrus M, Villeval JL, Clemetson KJ, Phillips DR. Platelet activation induces metalloproteinase-dependent GP VI cleavage to down-regulate platelet reactivity to collagen. Blood. 2005;105:186-191. 94. Andre P, Prasad KS, Denis CV, et al. CD40L stabilizes arterial thrombi by a beta3 integrin-dependent mechanism. Nat Med. 2002;8:247-252. 95. Ruggeri ZM, Orje JN, Habermann R, Federici AB, Reininger AJ. Activation-independent platelet adhesion and aggregation under elevated shear stress. Blood. 2006;108:1903-1910. 96. Jirouskova M, Chereshnev I, Vaananen H, Degen JL, Coller BS. Antibody blockade or mutation of the fibrinogen gamma-chain C-terminus is more effective in inhibiting murine arterial thrombus formation than complete absence of fibrinogen. Blood. 2004;103:1995-2002. 97. Chang MC, Lin HK, Peng HC, Huang TF. Antithrombotic effect of crotalin, a platelet membrane glycoprotein Ib antagonist from venom of Crotalus atrox. Blood. 1998;91:1582-1589. 98. Andre P, Delaney SM, LaRocca T, et al. P2Y12 regulates platelet adhesion/activation, thrombus growth, and thrombus stability in injured arteries. J Clin Invest. 2003;112:398-406. 99. Hirsch E, Bosco O, Tropel P, et al. Resistance to thromboembolism in PI3Kgamma-deficient mice. Faseb J. 2001;15:2019-2021. 100. Jackson SP, Schoenwaelder SM, Goncalves I, et al. PI 3-kinase p110beta: a new target for antithrombotic therapy. Nat Med. 2005;11:507-514. From www.bloodjournal.org by guest on September 7, 2015. For personal use only. 2007 109: 5087-5095 doi:10.1182/blood-2006-12-027698 originally published online February 20, 2007 The growing complexity of platelet aggregation Shaun P. Jackson Updated information and services can be found at: http://www.bloodjournal.org/content/109/12/5087.full.html Articles on similar topics can be found in the following Blood collections Cell Adhesion and Motility (790 articles) Cytoskeleton (143 articles) Hemostasis, Thrombosis, and Vascular Biology (2494 articles) Signal Transduction (1930 articles) Information about reproducing this article in parts or in its entirety may be found online at: http://www.bloodjournal.org/site/misc/rights.xhtml#repub_requests Information about ordering reprints may be found online at: http://www.bloodjournal.org/site/misc/rights.xhtml#reprints Information about subscriptions and ASH membership may be found online at: http://www.bloodjournal.org/site/subscriptions/index.xhtml Blood (print ISSN 0006-4971, online ISSN 1528-0020), is published weekly by the American Society of Hematology, 2021 L St, NW, Suite 900, Washington DC 20036. Copyright 2011 by The American Society of Hematology; all rights reserved.