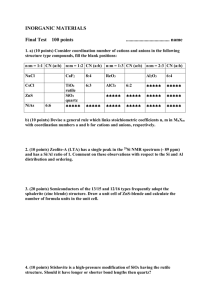
GY 302: Crystallography & Mineralogy Lecture 25: Class VIII-Silicates

UNIVERSITY OF SOUTH ALABAMA
GY 302: Crystallography &
Mineralogy
Lecture 25: Class VIII-Silicates
Tektosilicates part 1: Quartz
Last Time
(before the poster session)
Class VIII Minerals (Phyllosilicates)
A) Serpentine Group
B) Other Phyllosilicates
C) Clay Minerals
(chemistry, modes of analysis)
More Phyllosilicate Minerals
Mineral
“Clay” Minerals
Kaolinite
*”Smectite”
*Bentonite
*Illite
Formula
Al
2
Si
2
O
5
(OH)
4
(Na,Ca)
0-3
(Al,Mg)
(Na,Ca)
0-3
(Al,Mg)
2
Si
4
O
10
(OH)
2
·n(H
2
O)
2
Si
4
O
10
(OH)
2
·n(H
2
O)
(K,H
2
O)(Al,Mg,Fe)
2
(Si,Al)
4
O
10
[(OH)
2
·(H
2
O)
Serpentine Group (17 minerals)
”Serpentine”
*Antigorite
Chrysotile
(Mg, Fe, Ni)
3
Si
2
O
5
(OH)
4
Mg
3
Si
2
O
5
(OH)
4
Mg
3
Si
2
O
5
(OH)
4
Other Phyllosilicates
Stilpnomelane (series) ~K(Fe, Al)
10
Prehnite (series) Ca
2
Al(AlSi
3
Si
12
O
30
(OH)
O
10
)(OH)
2
2
Sepiolite
Garnierite
Apophylite
Astrophyllite**
Mg
4
Si
6
O
15
(OH)
2
·6(H
2
O)
(Ni,Mg)
4
Si
6
O
15
(OH)
2
·6(H
2
O)
KCa
4
(Si
4
O
10
)
2
F·8(H
2
O)
K
2
Na(Fe ++ ,Mn)
7
Ti
2
Si
8
O
26
(OH)
4
System
Triclinic
Monoclinic
Monoclinic
Monoclinic
Monoclinic
Monoclinic
Monoclinic
Triclinic
Orthorhombic
Orthorhombic
Orthorhombic
Tetragonal
Triclinic
70nm
Clay Geochemistry
(1:1 structure)
Halloysite
(Expandable)
100 nm
K + H
2
O kaolinite
(Non-expandable)
Clay Geochemistry
(2:1 structure)
Smectite
(Expandable)
K + H
2
O
Illite/Muscovite
(Non-expandable)
100 nm
Clay Geochemistry
(2:1 structure)
Smectite
(Expandable)
170 nm
K +
“glycol”
Illite/Muscovite
(Non-expandable)
Clay Mineral Analysis
Is primarily done using XRD
( X-ray diffraction ). In order to understand how this works, you need to consider physics (yuck).
Clay Mineral Analysis
Is primarily done using XRD
( X-ray diffraction ). In order to understand how this works, you need to consider physics (yuck).
Clay Mineral Analysis
Today’s Agenda
Class VIII Minerals (Tektosilicates)
1.
Quartz Group
Tektosilicate Minerals
Mineral
Quartz Group
Quartz
*Cristobalite
Coesite
Chalcedony
Opal crystalline”
*”Chert”
Formula
SiO
2
(multiple varieties)
SiO
2
SiO
2
SiO
2
SiO
2
·nH
2
O
SiO
2
(multiple varieties)
System
Trigonal
Tetragonal
Monoclinic
“non crystalline”
“non
“non crystalline”
Basic chemical composition: SiO
2
Tektosilicate Minerals
(Quartz Group)
Quartz
[ SiO
2
]
Crystal: Hexagonal (Trigonal)
Pt. Group: 32
Habit : bipyramidal, massive, drusy etc.
SG: 2.65; H: 7
L: vitreous; Str: colourless
Col: colourless (varied)
Clev: poor [0110]
Optics: Uniaxial (-); bir= 0.009
n w
=1.544; n e
=1.553
Occurrence: widespread http://webmineral.com
Name Derivation: From the German “quarz” of uncertain origin
Quartz Varieties [SiO
2
]
Agate - banded variety of chaledony
Amethyst - purple
Avanturine - translucent chalcedony
Carnelian - flesh red chalcedony
Cat's Eye - chatoyant
Chalcedony - microcrystalline quartz
Chert - cryptocrystalline quartz
Chrysoprase - apple green chalcedony
Citrine - yellow
Flint - microcrystalline quartz
Hornstone - flint
Jasper - red or brown chalcedony
Moss Agate - variety of chaledony
Plasma - green chalcedony
Prase - leek green chalcedony
Rock Crystal
Rose Quartz - rose colored
Sapphire Quartz - blue colored
Smoky Quartz - brown to black
Tiger Eye - entombed asbestos http://webmineral.com
Tektosilicate Minerals
(Quartz Group)
Coesite
[ SiO
2
]
Crystal: Monoclinic
Pt. Group: 2/m
Habit : spherical masses
SG: 2.93; H: 7.5
L: vitreous; Str: colourless
Col: colourless (varied)
Clev: none
Optics: Biaxial (+); bir= 0.005-0.006
n
α
=1.593; n
β
=1.595, n
γ
=1.599
Occurrence: High temp/pressure
(impact sites) http://webmineral.com
Named for Loring Coes, Jr. (1915-1973), American chemist, who first synthesized the phase.
Tektosilicate Minerals
(Quartz Group)
Cristabolite
[SiO
2
]
Crystal: Tetragonal
Pt. Group: 422
Habit : spherical masses
SG: 2.27; H: 6.5
L: vitreous; Str: white
Col: blue-grey, brown, yellow
Clev: none
Optics: Uniaxial (-); bir= 0.002-0.004
n w
=1.486; n e
=1.482
Occurrence: void-fill in felsic rocks with topaz and garnet http://webmineral.com
Name Derivation: From its location, Cerro San
Cristobal, Mexico.
Tektosilicate Minerals
(Quartz Group)
Chalcedony
[SiO
2
·?H
2
O]
Crystal: N/A
Pt. Group: N/A
Habit: massive (fibrous in TS)
SG: 2.57-2.63; H: 6.5
L: vitreous to waxy; Str: white
Col: blue-grey (colourless)
Clev: none
Optics: cryptocrystalline
Occurrence: diagenetic void-fill http://webmineral.com
Chalcedony in the main ingredient in “chert” but much chalcedony itself may be composed of another quartz pseudomorph; “morganite”
Tektosilicate Minerals
(Quartz Group)
Opal
[SiO
2
·nH
2
O]
Crystal: N/A
Pt. Group: N/A
Habit: massive (veinlets)
SG: 2.09; H: 5.5 to 6
L: vitreous, dull, waxy; Str: white
Col: varied
Clev: none
Optics: isotropic; n=1.43-1.46 http://webmineral.com
Occurrence: hot springs, petrified wood, biogenic
Name Derivation: From the Old Indian upala - "precious stone."
Tektosilicate Minerals
(Quartz Group)
“Chert”
[SiO
2
]
Crystal: N/A
Pt. Group: N/A
Habit: microcrystalline
SG: 2.09-2.65; H: 5.5 to 7
L: dull, waxy; Str: white
Col: varied
Clev: none
Optics: N/A
Occurrence: sedimentary http://www.uky.edu/KGS/rocksmn/images/chert_var.jpg
Chert is a rock name. Numerous varieties of chert have been identified.
Tektosilicate Minerals
(Quartz Group)
Lechatlerite (“Fulgurite)
[SiO
2
+ contaminants]
Crystal: N/A
Pt. Group: N/A
Habit: Amorphous?
SG: 2.20; H: 7.0?
L: dull; Str: white
Optics: N/A
Col: white
Clev: none
Occurrence: lightning strikes http://www.mindat.org/gphotos/0707699001129998806.jpg
Lightning strikes may pass 1,000,000 volts of electricity into the ground fusing quartz sand into “glass”.
Six Quartz Polymorphs
Displacive polymorphic transformations require relatively minor changes in the crystal lattice (e.g., modification of
α, β or γ crystallographic angles).
There is generally no change in energy at the transformation threshold so polymorphic transformations are instantaneous and reversible.
Six Quartz Polymorphs
Displacive polymorphic transformations require relatively minor changes in the crystal lattice (e.g., modification of
α, β or γ crystallographic angles).
There is generally no change in energy at the transformation threshold so polymorphic transformations are instantaneous and reversible.
β-quartz If you heat “quartz” above 600 ºC it transforms to the
α- polymorph (also known
α-quartz as high quartz). When the temperature falls below
600ºC it transforms back to the
β- polymorph (also known as low quartz).
Six Quartz Polymorphs
Displacive polymorphic transformations require relatively minor changes in the crystal lattice (e.g., modification of
α, β or γ crystallographic angles).
There is generally no change in energy at the transformation threshold so polymorphic transformations are instantaneous and reversible.
The higher the pressure of formation, the higher the specific gravity.
Specific Gravity
67 km 220 km
Quartz Phase Diagrams
Olivine-Enstatite-Quartz
System
Quartz Phase Diagrams
Quartz-Nephaline-Kalsilite
System
We could look at several other
SiO
2
phase diagrams, but that would leave Dr. Allison with nothing to torment you with in
GY 303.
Quartz: Last Words
The quartz minerals are one of the most pervasive groups on our planet. (Occurrence = everywhere)
•Igneous (Bowen’s Reaction Series)
•Sedimentary (Diagenesis)
•Metamorphic (low to high grade)
•Hydrothermal (epithermal ore emplacement)
Quartz: Last Words
Except for 2 situations:
1) Nephaline*-bearing rocks
NaAlSiO
4
+ 2SiO
2
→ NaAlSi
3
O
8
(albite)
2) Corundum-bearing rocks
Al
2
O
3
+ SiO
2
→ Al
2
SiO
5
(Sil/And/Ky)
* A feldspathoid; you’ll hear about these next time
Today’s Stuff To Do
1. Poster Session next Tuesday
(posters due to Doug by 5 PM today)
They will be printed by 2 PM Thursday
2. Phyllosilicates quiz in 1 hr
3) Final lab test/mineral notebook exam (2 weeks)
100 mineral lab final list (Thursday)
1.
Next Time
Poster Prep period (no class meeting)
GY 302: Crystallography and
Mineralogy
Lecture 25: Silicates 8: Quartz Group
Instructor: Dr. Doug Haywick dhaywick@southalabama.edu
This is a free open access lecture, but not for commercial purposes.
For personal use only.