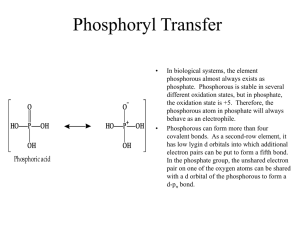
INHIBITION OF TAU KINASE ACTIVITY ... RAY BY
advertisement

INHIBITION OF TAU KINASE ACTIVITY BY ATP BY RAY RABINDRAN B.S., Texas A&M University (1985) M.B.A., The University of Chicago (1993) Submitted to the Department of Chemistry In partial fulfillment of the requirements for the degree of DOCTOR OF PHILOSOPHY in MASSACHUSETTS INSTITUTE BIOCHEMISTRY MAR 0 3 1998 at the LIBRARIES OF TECHNOLOGY MASSACHUSETTS INSTITUTE OF TECHNOLOGY February, 1998 © Ray Rabindran. All rights reserved. The author hereby grants to MIT permission to reproduce and to distribute publicly paper and electronic copies of this thesis document in whole or in part. Signature of Author Ray- indran, Department of Chemistry, October 21, 1997 Certified by_ Pr fessor Vernon M. Ingram, Thesis Supervisor ,, Accepted by // Professor Dietmar Seyferth, Chairman, Department Committee for Graduate Students i O1993 3 This thesis has been reviewed by a committee from the Massachusetts Institute of Technology Chemistry and Biology departments and the Harvard Medical School Center for Neurological Disorders. Accepted b , Professor James Williamson, MIT Chemistry Dept., Thesis Committee Chairman Accepted by Profesmsr Vernon M. Ingrfjpm-T Biology Dept., Thesis Supervisor Accepted by /Accepted /rof b r leterT. Lansbury, Harvard Medical School Accepted by Protessor Jun Liu, MIT-C istry & Biology Dept.'s INHIBITION OF TAU KINASE ACTIVITY BY ATP BY RAY RABINDRAN Submitted to the Department of Chemistry Massachusetts Institute of Technology on October 21, 1997 in partial fulfillment of the requirements for the degree of Doctor of Philosophy in Biochemistry ABSTRACT Alzheimer's disease (AD) is characterized clinically by a progressive loss of memory and other cognitive functions, resulting in a profound dementia. The decline in cognitive and adaptive functioning observed in all cases of AD is accompanied by the progressive accumulation in the brain of insoluble fibrous material, both extracellularly and intracellularly. Extracellular plaques have a core that consists of the amyloid-3 peptide (AP), an abnormal cleavage product of the amyloid 0-protein precursor (APP). Intracellular neurofibrillary tangles (NFTs) are composed of fibrillar paired helical filaments (PHFs) containing tau protein that is hyperphosphorylated. There is evidence that the two lesions contribute to the dysfunction and death of CNS neurons. The lesions may represent the final pathology upon which different initiating events converge. Accumulating evidence suggests that abnormal up-regulation of tau protein phosphorylation leads to the formation of PHF-tau. Hyperphosphorylation of tau protein has been shown to interfere with the normal function of tau to form and stabilize the microtubular cytoskeleton. Studies on the regulation of the phosphorylation state of tau have suggested that either increased activities of protein kinases and/or reduced activities of protein phosphatases may be involved. It is known that ATP metabolism is compromised in AD and in old age. Furthermore, the in vitro activities of two tau kinases, PK40O and the catalytic subunit of cyclic-AMPdependent protein kinase (PKAcat), have been shown to be significantly reduced in the presence of ATP 4" that is present in excess of total Mg 2+ (viz. ATPfe,). Ionic strength effects do not account for the reduction in activity. The effect could be due to either inhibition by ATPee or loss of activation by free Mg 2+. Thus, age-dependent loss of inhibition by ATPfrce or increase in activation by free Mg 2+ could explain the rise in tau phosphorylation observed in AD. Inhibition kinetics and equilibrium binding studies on ERK2 and PKAcat were performed with MgATP and human recombinant tau protein (hTau40) as substrates. The catalytic mechanism for ERK2 was determined to be ordered sequential, with MgATP binding first. The catalytic mechanism for PKAcat was characterized as random sequential in the absence of Mg 2+and ordered at high (10 mM) free Mg 2+, with MgATP binding first. The reduction in the in vitro activities of both ERK2 and PKAcat was determined to be due to inhibition by ATPfec, acting as a competitive inhibitor versus MgATP in the phosphorylation reaction. For both ERK2 and PKAcat, ATPee was a noncompetitive inhibitor versus hTau40. The inhibition constants (Ki's) for ATPf, were 32 and 9 M for ERK2 and PKAcat, respectively. A significant quantitative difference in the affinities of ERK2 (Km=499 p.M) and PKAcat (Km=35 M) for MgATP indicates that, in vitro, ERK2 is more sensitive to competitive inhibition by ATP&fr than is PKAcat. These results suggest that declines in physiologic levels of ATP may have quantitatively different effects on the in vivo activities of two tau kinases. ACKNOWLEDGEMENTS Every significant individual accomplishment involves the concerted actions of a network of many other individuals. As such, I cannot possibly thank everyone who has made it possible for me to reach this educational milestone. Nevertheless, I want to express my gratitude for all the "teachers" who appeared when I, the "student," needed to learn something new. Many of the lessons were not at the laboratory bench. Furthermore, the "student" did not always want to learn. Luckily, the educational process inevitably succeeds whenever a "teacher" simply refuses to give up on a "student." I am indeed grateful to all those whose lives connected with mine to make this achievement possible. Therefore, I would like to recognize some individuals who were especially influential. A successful ending to my graduate career at the Massachusetts Institute of Technology (MIT) would not have been possible without the foresight of chemistry professor William Orme-Johnson, who encouraged and challenged me to develop the fixity of purpose necessary to reach this milestone. My thesis advisor, biology professor Vernon Ingram, provided me with many important lessons, but none as memorable as the importance of being able to see the big picture and the fine details simultaneously. The members of my thesis committee (professors Peter Lansbury, Jun Liu, and Jamie Williamson) helped me to develop my ability to ask the right questions, to answer them with data, and to consider alternative interpretations of the data. Some other important "teachers" outside MIT were also instrumental to my achievement. My mother, Hannah Rabindran, taught me by example that education is a lifelong process and a privilege. My daughter, Leanna Krista Rabindran, has always reminded me of what it is like to learn with a spirit ofjoy and playfulness. From my partner, Michele Essex, I have learned about the beauty in ordinary moments when life is viewed as an everchanging art form. I am truly blessed to have had such a high caliber "faculty." TABLE OF CONTENTS ................................................................... ix ............ List of Figures ..................... . . . .... xi es............................................................................... List of Schem L ist of T ables ..................................................................................... . . . .. xii .................... xiii List of E quations................................................................................ .......... xiv . . ................ K ey to Abbreviations ...................................................... ............................... ......... 15 1. Introduction ................................. 2. Literature review .............................................................................. 20 ................ 21 Pathological protein deposits in Alzheimer's disease ................ A. 21 ............... Amyloid-3 plaques................................. i. ................. 23 Neurofibrillary tangles.............................. ............... ii. .... 27 Etiological hypotheses for Alzheimer's disease.................. B. ................... 27 Amyloid cascade hypothesis........................... i. ........................ 30 Tangle hypothesis................ ii. Oxidative energy metabolism hypothesis................................. 31 iii. 34 T au protein kinases.................................................................................. C. 36 ........... ..... PKAcat and ERK2 structures............................ D. General architecture............................... .......... 36 i. PKAcat structure ..................................................... 37 ii. 38 ERK2 structure......................................... iii. 39 ............... PKAcat and ERK2 regulation........................ E. .......... 40 ...... PKcat regulation............................ i. .......... 41 ...... ERK2 regulation............................ ii. PKA cat m echanism ................................................................................... 42 F. Regulation of kinases by ATPfe,,.....................................44 G. . . . 45 . . . . . ....... ... Regulation of kinases by free Mg 2 ....... ........ .......... H. 3. ......... 47 Inhibition of PKAt by ATP ............................................... ............... 48 ...... Experimental procedures .............................. A. ... 48 M aterials........................... ........................... i. Purification of recombinant hTau40...............................48 ii. ............................................ 49 Phosphorylation assay for PKAat iii. ...... 49 Equilibrium binding assay for PKAcat.................... iv. 50 ................ Calculation of ATPf , concentrations..................... v. Determination of sequential kinetics parameters for a vi. ..... 50 random sequential bireactant reaction.................. Determination of the effect of ATPfee on the slopes and intercepts vii. of Lineweaver-Burk plots of PKAcat initial velocities........................53 Determination of the effect of Mg 2+on the slopes and intercepts viii. of Lineweaver-Burk plots of PKAcat initial velocities.......................58 Calculation of equilibrium binding parameters for PKAca....................61 ix. B. C. 4. ii. Kinetics of inhibition with varying ATPfree and MgATP ........ iii. Kinetics of inhibition with varying ATPfee and hTau40 ..................... 73 iv. Equilibrium binding of ATPfr, to PKAcat ............... B. C. .. 68 .............. 78 .. v. Equilibrium binding of ATPce to PKAat*hTau40 complex ......... 78 vi. Kinetics of inhibition with adenosine and uridine ................................ 83 86 ................................. Discussion .............. ........................... . Inhibition of ERK2 by ATP . .............................. A. 5. Results ....................... ............... ..... ...... .................... 61 i. Two-substrate kinetics with MgATP and hTau40.............63 .................98 .......... 99 ...... Experimental procedures............................ 99 ................... M aterials .................................. i. Purification of recombinant hTau40 ........................................... 99 ii. 99 ................ Phosphorylation assay for ERK2......................... iii. Equilibrium binding assay for ERK2...............................................99 iv. Calculation of ATPfi concentrations .............................................. 100 v. vi. Determination of sequential kinetics parameters for an ordered ......................... 100 sequential bireactant reaction............................ vii. Determination of the effect of ATPfre on the slopes and intercepts of Lineweaver-Burk plots of ERK2 initial velocities ........................ 100 viii. Determination of the effect of Mg 2+ on the slopes and intercepts of Lineweaver-Burk plots of ERK2 initial velocities ........................ 100 101 Results ................................................ Two-substrate kinetics with MgATP and hTau40 ............................ 101 i. Kinetics of inhibition with varying ATP&,, and MgATP .................... 101 ii. Kinetics of inhibition with varying ATPo,, and hTau40.................109 iii. Equilibrium binding of ATPfre to ERK2........................................ 109 iv. Discussion.................................................115 Inhibition of GST-ERK2 by ATP ...................................................................... 120 Experimental procedures................................... ......... 121 A. 121 ............ . ........................ M aterials ..................... i. Purification of recombinant hTau40 ................................................ 121 ii. Phosphorylation assay for GST-ERK2. ......................................... 121 iii. Calculation of ATP or,concentrations ............................................. 121 iv. Determination of the effect of ATPfre on the slopes and intercepts v. of Lineweaver-Burk plots of GST-ERK2 initial velocities .............. 122 Determination of the effect of Mg2+ on the slopes and intercepts vi. of Lineweaver-Burk plots of GST-ERK2 initial velocities............. 122 ............................................. 122 Results ............................ B. varying ATP e and MgATP ................. 122 with Kinetics of inhibition i. ii. C. Kinetics of inhibition with varying ATPo, and hTau40.................. 126 Discussion................................................ .................. 126 6. Sum m ary ................... ... .................. ..................... ..... .............. Summary of conclusions.......... A. ............ ...... Summary of contributions........................... B. Future research................................................................ C. 133 133........................... 134 136 140 7. References ................................................. 142 8. ...................... ...... .. Appendices............................. sequential random of a for inhibition equations Rate A. bireactant reaction........................ ................ ............................ Rate equations for inhibition of an ordered sequential B. bireactant reaction............................................................... Rate equations for non-essential activation of a random sequential C. ............ .... ... ... ... bireactant reaction ..................... .. ...... sequential Rate equations for non-essential activation of an ordered D. bireactant reaction ................. .............................. ...................... 146 147 151 155 158 LIST OF FIGURES 3-1 Dependence of PKAcat initial velocity on [MgATP] at different fixed values of [hTau40] ............................................ 64 A. Michaelis plots of the dependence of PKA activity on MgATP and hTau40.........65 B. Lineweaver-Burk plots of the activity data presented in (A) ............................. 66 3-2 Dependence of PKAat initial velocity on [MgATP] at different ...... ...................... 69 fixed values of [ATP ,.] ............................... ..... A. Michaelis plots of the dependence of PKAat activity on MgATP and ATPfe ...... 70 B. Lineweaver-Burk plots of the activity data presented in (A)............................71 3-3 Dependence of PKAcat initial velocity on [hTau40] at different ........... ................... 74 fixed values of [ATPf .......................................................... A. Michaelis plots of the dependence of PKAcat activity on hTau40 and ATPe ....... 75 B. Lineweaver-Burk plots of the activity data presented in (A) ......................... 76 C. Replot of Lineweaver-Burk Slope*Intercept versus [ATPfrc]/[MgATP]..............77 3-4 80 A. Binding of ATPr to PKcat ........................................................ B. Scatchard plot of ATPfGe binding to PKAcat.........................................81 3-5 Effect of uridine and adenosine nucleosides on PKAcat initial velocity at different fixed values of [M BP] .......................................................................... 85 4-1 Dependence of ERK2 initial velocity on [MgATP] at different fixed values of [hTau40]......................................................................................102 A. Michaelis plots of the dependence of ERK2 activity on MgATP and hTau40.....103 B. Lineweaver-Burk plots of the activity data presented in (A)...........................104 4-2 Dependence of ERK2 initial velocity on [MgATP] at different fixed values of [A T P ] ................................................. .............. ................... ..106 A. Michaelis plots of the dependence of ERK2 activity on MgATP and ATPfee.....107 B. Lineweaver-Burk plots of the activity data presented in (A) ............... .......... 108 4-3 Dependence of ERK2 initial velocity on [hTau40] at different fixed values of [ATPfai ] ...................................................................... ................ 110 A. Michaelis plots of the dependence of ERK2 activity on hTau40 and ATPfr=e......111 B. Lineweaver-Burk plots of the activity data presented in (A) ............................ 112 C. Replot of Lineweaver-Burk slope*intercept versus [ATPfee]/[MgATP]........ 113 5-1 Dependence of GST-ERK2 initial velocity on [MgATP] at different ..........123 ..................... fixed values of [ATP ,] ...................................................... A. Michaelis plots of the dependence of GST-ERK2 activity on ...... 124 M gA TP and A TP .................................................... ............................ B. Lineweaver-Burk plots of the activity data presented in (A)..........................125 5-2 Dependence of GST-ERK2 initial velocity on [hTau40] at different ........................ ............... 127 ................ fixed values of [ATP e,,] .......... A. Michaelis plots of the dependence of GST-ERK2 activity on 128 ............................ MgATP and ATPf...................................... B. Lineweaver-Burk plots of the activity data presented in (A) ........................... 129 C. Replot of Lineweaver-Burk slope*intercept versus [ATPfee]/[MgATP]........ 130 6-1 Dependence of PKAcat and ERK2 activities on [hTau40] at hypothetical 137 Alzheimer's disease concentrations of [MgATP] and [ATP.].................. A. PKAcat activity in normal versus hypothetical Alzheimer's disease................. 138 B. ERK2 activity in normal versus hypothetical Alzheimer's disease...................... 139 LIST OF SCHEMES 2-1 General protein phosphorylation reaction ............... 3-1 Equilibria for a random sequential bireactant reaction................................ 51 3-2 Equilibria for an ordered sequential bireactant reaction.................. 3-3 Equilibria for inhibition of a random sequential bireactant reaction by inhibitor I............................. ......................................... 43 ... ............................ ... 52 54 3-4 Equilibria for inhibition of an ordered sequential bireactant reaction by inhibitor I .................................... .. .................... 55 3-5 Equilibria for activation of a random sequential bireactant reaction by non-essential activator M .................................................... 59 3-6 Equilibria for activation of an ordered sequential bireactant reaction by non-essential activator M.....................................................60 3-7 Ordered sequential mechanism for PKAcat-catalyzed ........ ............................. phosphorylation of hTau40 ............ 89 Random sequential mechanism for PKAcat-catalyzed ................. phosphorylation of hTau40 ............................... .................90 3-8 3-9 Proposed mechanism for ATPf~, inhibition of ordered sequential pathway for PKA at .................... ......................................................................... 95 3-10 Proposed mechanism for ATPe inhibition of random sequential p athw ay for P KA .t......................................................................... .................. 96 4-1 Ordered sequential mechanism for ERK2-catalyzed ............................................. 117 phosphorylation of hTau40...................... 4-2 Proposed mechanism for ATP&ee inhibition of ordered sequential pathw ay for G ST-ERK 2 ..................................................................... 5-1 .............. 119 Proposed mechanism for ATPee, inhibition of ordered sequential pathway for ERK2............................................................................. 131 LIST OF TABLES 3-1 Effects of varying inhibitor I on the slopes and intercepts of Lineweaver-Burk plots of a random sequential bireactant reaction........................56 3-2 Effects of varying inhibitor I on the slopes and intercepts of Lineweaver-Burk plots of an ordered sequential bireactant reaction......................57 3-3 Estimated kinetic parameters for PKAcat................................................67 3-4 Effect of ATPfi. on Lineweaver-Burk plots for PKAcat, ERK2, and GST-ERK2......................................... 3-5 Binding of ATPfr to PKAcat*hTau40 complex........................ 4-1 Estimated kinetic parameters for ERK2................................ 4-2 Binding of ATPfre to ERK2 ........................................... ......... ................. 72 82 105 .......... 114 LIST OF EQUATIONS 3-1 Rate equation for a random sequential bireactant reaction....................53 3-2 Rate equation for an ordered sequential bireactant reaction....................................53 3-3 Multiple regression model for testing the effect of ATPfree on slopes and intercepts of Lineweaver-Burk plots...................... 3-4 Simple equilibrium binding of ligand to protein....................... X11i ................ .................. 58 61 KEY TO ABBREVIATIONS AD AD APO E APP ATP [y- 33P]ATP ATPree cAMP CaMK II CDK2 DTT EDTA ERK ERK2 FAD GSK-3 GST-ERK2 hTau40 MAPK MBP PAGE PC12 PKA PKAcat PHF PMSF PP2A1 PP2B PSI PS2 SDS SF TM Amyloid-3 peptide Alzheimer's disease Apolipoprotein E Amyloid precursor protein Adenosine triphosphate gamma- 33P labeled ATP Uncomplexed ATP Cyclic adenosine monophosphate Calcium/calmodulin-dependent protein kinase II Cyclin-dependent kinase 2 Dithiothreitol Ethylenediamine Extracellular signal-regulated kinase Extracellular signal-regulated kinase 2 Familial Alzheimer's disease Glycogen synthase kinase 3 Glutathione S-transferase-ERK2 fusion protein Recombinant human tau protein (441 amino acid isoform) Mitogen-activated protein kinase (aka ERK) Myelin basic protein Polyacrylamide gel electrophoresis Rat pheocytochromata cell line Cyclic-AMP-dependent protein kinase Catalytic subunit of PKA Paired helical filaments Phenyl methyl sulfonyl fluoride Protein phosphatase 2A1 Protein phosphatase 2B (aka calcineurin) Presenilin 1 Presenilin 2 Sodium dodecyl sulfate Straight filaments Transmembrane Chapter 1: Introduction In 1906, the German physician Alois Alzheimer first described a peculiar case of dementia in a non-elderly woman. That report initiated a search for the etiology of the disease that continues today. The term "Alzheimer's disease" (AD) was first used by Kraepelin (1910) in his classic psychiatry textbook. AD is now the most common cause of old-age associated dementia in humans. It is prevalent among all races and ethnic groups, although the prevalence in developed nations is increased because of greater life expectancy. AD is the fifth most common cause of death in the United States. An estimated $80 billion is spent each year in caring for AD sufferers in the United States. Alzheimer's disease is characterized clinically by a progressive loss of memory and other cognitive functions, resulting in a profound dementia. AD accounts for 50-70% of the typical, late-onset cases of dementia. The progressive decline in intellectual and adaptive functioning manifests as deficits in memory, attention, orientation, abstraction, and judgment. Because of the resemblance between AD dementia and the dementia of other organic brain syndromes, definitive diagnosis relies on the detection of characteristic neuropathology at autopsy. The major morphological changes observed in the brains of AD sufferers at autopsy include the presence of cerebrovascular and neuritic (senile) plaques and intracellular neurofibrillary tangles in the neocortical areas, hippocampus and other brain regions. Additionally, cortical atrophy and loss of neurons are also observed. The etiology of AD is now known to be heterogeneous. The heterogeneity of the disease is reflected in the fact that only 10% of cases are the familial (autosomal-dominant) form of AD (FAD). The other 90% of cases are sporadic. The decline in cognitive and adaptive functioning observed in all cases of AD is accompanied by the progressive accumulation in the brain of insoluble fibrous material, both extracellularly and intracellularly. In fact, the presence of abnormal proteins is regarded to be one of the hallmarks of AD. Extracellular plaques have a core that consists of the amyloid-3 peptide (AP), a cleavage product (39 to 43 amino acids in length) of the amyloid 0-protein precursor (APP). Intracellular tangles are derived from fibrillar aggregates of tau protein that is hyperphosphorylated. These two AD lesions may represent the "final common pathway" upon which different initiating events converge. There is evidence that the lesions contribute to the dysfunction and death of CNS neurons and ultimately to the dementia of AD. The convergence of different initiating events on the development of plaque and tangle lesions in the AD brain may be crucial for the treatment of AD because these "final" lesions could be pursued as therapeutic targets common to all forms of AD. Genetics studies of FAD have identified four distinct genes that are linked to this form of the disease. The gene encoding APP on chromosome 21 is now known to be the site of at least six different missense mutations that cause an early onset form of FAD. A second gene, presenilin 1 (PS1), has been located on chromosome 14. The PS1 gene defect is associated with a very early and severe amyloid-plaque phenotype, with clinical symptoms presenting by age 45. A third gene, presenilin 2 (PS2), has been mapped to chromosome 1. It is now clear that mutations in the APP, PS1, and PS2 genes alter APP processing in a way that causes a shift in the metabolism of APP such that more of the A3 protein is produced. The fourth genetic trait that has been linked to FAD is the naturally occurring polymorphism of the APO E gene on chromosome 19. A large number of genetic epidemiological studies indicate that the inheritance of one or two APOE4 alleles significantly increases the likelihood of developing AD and decreases the age of onset of the disease, compared with people who carry no APOE4 alleles. The APOE4 allele on chromosome 19 has been shown to be a risk factor for both FAD and sporadic AD. Accumulating evidence suggests that alterations in the normal metabolism and posttranslational processing (e.g. hyperphosphorylation) of tau protein lead to the formation of neurofibrillary tangles. Ultrastructurally, NFTs contain abnormal paired helical filaments (PHFs) as their major fibrous component. Tau protein, in a hyperphosphorylated form, has been shown to be the major subunit of PHFs in AD. Hyperphosphorylation of tau protein has been shown to interfere with the normal function of tau to form and stabilize the microtubular cytoskeleton. It has also been shown that all six human isoforms of tau protein contribute to the formation of AD PHFs. Studies on the mechanisms that might account for the disruption in the regulation of the phosphorylation state of tau have suggested that either aberrant activation of protein kinases and/or inactivation of protein phosphatases may be involved. Furthermore, studies on the abnormal phosphorylation sites that distinguish PHF tau from normal tau have led to the identification of candidate kinases and phosphatases. Some of these candidate kinases include extracellular signal-regulated kinases (ERKs), glycogen synthase kinase-3 (GSK-3), calcium/calmodulin-dependent protein kinase (CaMK II), and cyclic-AMP-dependent protein kinase (PKA). Although less is known about phosphatases that might be involved, there is some evidence suggesting that protein phosphatase 2A1 (PP2A1) and protein phosphatase 2B (PP2B or calcineurin) are likely candidates. Given the significance of tau hyperphosphorylation in the etiology of AD, an improved understanding of the cellular (metabolic) processes that regulate the activities of tau kinases might yield some insights on the aberrant processing of tau in AD. Roder and Ingram (1991) reported that the activity of the tau kinase PK40" 2 is significantly reduced by ATP 4- (ATPfree) k2 that is added in excess of Mg 2+. This apparent inhibition of PK40er by ATPfree provided the basis for an interesting hypothesis regarding the disruption of tau kinase regulation in AD. It is known that deficits in oxidative energy metabolism, and consequently ATP levels, accompany normal aging and may be related to an age-dependent increase in mitochondrial DNA mutations. Age-dependent impairment of oxidative energy metabolism may therefore contribute to the pathogenesis of AD by disrupting the normal inhibition of tau kinases by ATPfree. Other studies (unpublished data from Barbara Blanchard) in this laboratory suggested that the activity of the catalytic subunit of PKA (PKAcat) is reduced by up to 80% when 4 mM 4 ATP 4 is added to the reaction containing 2 mM Mg 2+ and 2 mM ATP -. There are two likely 4 explanations for the reduced activity of both PK40k2 and PKAcat when ATP is added in 2+ excess of Mg2+: inhibition by ATPr, or a loss of activation by free Mg . The objectives of this thesis are to: 1) determine whether the reduction in the activity of both PKAcat and PK40 k2 2 involves inhibition by ATPftee or loss of activation by free Mg +; 2) characterize the kinetic mechanism of the inhibition or activation; and, 3) determine the relevant inhibition or activation constants. Chapter 2: Literature review A. Pathological protein deposits in Alzheimer's disease The major morphological changes observed in the brains of AD sufferers at autopsy include the presence of cerebrovascular and neuritic (senile) plaques and intracellular neurofibrillary tangles in the neocortical areas, hippocampus and other brain regions. Additionally, cortical atrophy and loss of neurons are also observed. The decline in cognitive and adaptive functioning observed in all cases of AD is accompanied by the progressive accumulation in the brain of insoluble fibrous material, both extracellularly and intracellularly. In fact, the presence of abnormal proteins is regarded to be one of the hallmarks of AD. Extracellular plaques have a core that consists of the amyloid- peptide (AP), a cleavage product (39 to 43 amino acids in length) of the amyloid -protein precursor (APP). Plaques are surrounded by neurons containing neurofibrillary tangles (NFTs). Intracellular NFTs are derived from fibrillar aggregates of tau protein that is hyperphosphorylated. These two AD lesions may represent the "final common pathway" upon which different initiating events converge. i. Amyloid- plaques Amyloidosis is defined as a group ofbiochemically diverse conditions in which normally innocuous, soluble proteins polymerize to form insoluble fibrils. The growing mass of amyloid fibrils then associates with plasma and extracellular matrix proteins and proteoglycans to form amyloid deposits or plaques. Amyloidosis accompanies numerous medical conditions and disorders including cancer, rheumatoid arthritis, AD, chronic renal dialysis, familial amyloid poylneuropathy, and metabolic diseases such as diabetes. The term amyloid, meaning starch-like, was introduced by Virchow in 1851 based on his concluding that the insoluble fibrils were composed of cellulose. The name remained despite work by Friedrich and Kekule during the same decade demonstrating that the major component of amyloid deposits is protein. Amyloid plaques are commonly defined by three physical characteristics: fibrous morphology, birefringent staining, and a distinct X-ray fiber diffraction pattern (Glenner, 1980). Without polarizing optics, amyloid deposits in tissue sections appear homogeneous, eosinophilic, and amorphous when analyzed by light microscopy (Glenner & Page, 1976). With polarizing optics, unstained amyloid deposits exhibit birefringence, indicating the presence of highly ordered structures (Divry & Florkin, 1927). In 1984, Glenner and Wong (1984) purified and sequenced a 28-amino-acid peptide, which they called amyloid beta peptide (AP) , from amyloid plaque-laden leptomeningeal tissues of a patient who died with AD. Subsequently, the subunit peptide from the core amyloid plaque was found to be a larger form of AP, consisting of 40-43 amino acids extended at the carboxy terminus (Masters et al., 1985). Staining with Congo red dye indicates that the linear dye molecules are arranged in parallel along the amyloid fibril axis (Glenner & Page, 1976). Eanes and Glenner (1968) determined from X-ray diffraction patterns of isolated amyloid fibrils that the subunits are arranged in a cross-p-pleated sheet conformation, with the polypeptide backbones oriented perpendicular to the long axis of the fibril. Infrared spectroscopic analysis revealed that the orientation of the polymerized polypeptides in antiparallel (Termine et al., 1972). To date, relatively little is known about the normal biological function of the AP peptides. The A3 peptide is derived from processing of the amyloid precursor protein (APP). APP is a ubiquitously-expressed transmembrane glycoprotein with no known biological function. Four mRNA species are generated from the APP gene by alternative splicing, APP695, 714, 751, and 770, all of which encode AP. The two largest APP transcripts contain sequences homologous to a Kunitz-type proteinase inhibitor (protease nexin II) (Van Nostrand et al., 1989). APP has a short half-life and is metabolized rapidly down two pathways in all cells. Normal processing of APP leads to the production of a 39 amino acid form of the A3 peptide, referred to as AP1-39. Abnormal processing of APP generates a slightly longer, and pathologically more significant, peptide of 42-43 residues, referred to as A31-42(43). 42(43) Both forms of A3 occur normally in the brain and in other tissues, although A31- occurs in lower amounts. The A3 peptide sequence overlaps the putative transmembrane sequence of APP, which is postulated (Kang et al., 1987) to have an outer membrane border between Lys28 and Gly29 in the A3 sequence. Relatively little is known about the normal biological function of the APP protein. Several functions have been proposed. Nishimoto et al. (1993) suggested that APP is a neuronal receptor mediating its effects via a G protein, GO. Another suggestion, that APP forms an ion channel, derives from the observation that AP forms channels which are permeable to metal ions in artificial lipid bilayers (Arispe et al., 1993). ii. Neurofibrillary tangles In AD, intracellular fibril deposits are formed in fairly specific neuronal populations. The cell types include pyramidal cells in CAl of the hippocampus, layers 2 and 4 of the entorhinal cortex, layers 3 and 5 of the association cortex, and relatively large neurons in the nucleus basalis of Meynert, the septal nucleus, the nucleus raphe dorsalis, and the locus ceruleus. These are the subsets of neurons lost during the progression of the disease. Ultrastructurally, the NFTs contain abnormal paired helical filaments (PHFs) as their major fibrous components and straight filaments (SFs) as their minor fibrous components. The PHF consists of two strands of subunits which twist around one another in a helical fashion. When viewed in the electron microscope, the helical twist and relative disposition of the two strands give rise to images in which the width alternates between about 8 and 20 nm, with an apparent periodicity of 80 nm (Arriagada et al., 1992). The unusual solubility characteristics of PHF, in particular, insolubility in sodium dodecyl sulfate (SDS), slowed progress in the elucidation of the nature of PHF. Using an immunochemical approach with PHF-specific antibodies (Ihara et al., 1983), researchers showed that tau is a major component of PHF (Nukina et al., 1985; Grundke-Iqbal et al., 1986). Later protein chemistry and molecular cloning studies directly established that tau protein is the principal component of PHF (Wischik et al., 1988; Kondo et al., 1988; Goedert et al., 1988). PHFs were found to consist of fibrils of tau (PHF-tau), a heat-stable microtubule-associated protein necessary for the correct assembly and stabilization of axonal microtubules (Weingarten et al., 1975; Kanai et al., 1989). Tau has been known to be a potent promoter of tubulin polymerization in vitro (Weingarten et al., 1975). The binding of tau to microtubules reduces their dynamic instability (Dreschel et al., 1992). Analysis of the dynamics of microtubule formation indicates that tau increases the rate of association and decreases the rate of dissociation of tubulin molecules at the growing end and inhibits the transition to the catastrophic shortening phase. Tau is subject to developmentally-regulated alternative mRNA-splicing. In adult human brain, six tau isoforms are produced from a single gene through alternative splicing (Lee, 1990). The isoforms range from 352 to 441 amino acids and differ from each other by the presence or absence of three inserts. In fetal brain, only the transcript encoding the shortest isoform is expressed. The most striking feature of the tau sequences is the presence of three or four tandem repeats of 31 or 32 amino acids located in the carboxy-terminus half Studies with recombinant tau proteins have shown that the repeats constitute microtubulebinding domains (Butner & Kirscher, 1991). Therefore, tau consists of a carboxy-terminus microtubule-binding domain and an amino-terminus projection domain. Tau is a phosphoprotein (Butler & Shelanski, 1986) and its phosphorylation is also developmentally regulated (Goedert et al., 1993). Phosphorylation has been shown to negatively regulate the ability of tau to bind to microtubules. Tau phosphorylated by ERK kinase has one-tenth the ability of non-phosphorylated tau to bind to microtubules (Dreschel et al., 1992). Tau from fetal brain is phosphorylated at more sites than is tau from adult brain. In fetal brain, the shortest tau isoform is phosphorylated at six to eight sites, whereas in adult brain all six isoforms are phosphorylated at two or three sites (Ksiezak-Reding et al., 1992). Mass spectroscopic and immunological studies have been used to identify several phosphorylation sites. In fetal brain, a significant fraction of tau is phosphorylated at serine residues 202, 396, and 404 (according to the numbering of the largest human tau isoform). In adult brain, serine 404, 202 and 396 are phosphorylated. All of these serine residues are followed by a proline, suggesting that proline-directed protein kinases are responsible for the phosphorylation of tau in normal brain. Three lines of evidence have been used to show that PHF-tau consists of a hyperphosphorylated form of tau. First, antibodies to PHF recognize a phosphorylated form of tau with slower mobility on sodium dodecyl sulfate (SDS)-polyacrylamide gel electrophoresis (PAGE) (Ihara et al., 1986). Second, one monoclonal antibody, tau 1, that recognizes tau in the normal brain can label tangles in situ and abnormal species of tau bound to PHF only after alkaline phosphatase treatment (Grundke-Iqbal et al., 1986; Iqbal et al., 1986). Third, alkaline phosphatase treatment brings about normal mobilities in SDS-PAGE for PHF-tau (Lee, et al., 1991; Flament et al., 1989). PHF-tau contains all six adult brain isoforms in a hyperphosphorylated state. The estimates of the number of phosphorylation sites in PHF-tau range from six to eight (Ksiezak-Reding et al., 1992). Several sites that are abnormally phosphorylated in isolated PHF-tau have been identified using mass spectrometry and the use of phosphorylation-dependent antibodies. Those sites include serine 202, threonine 231 and serines 235, 262, and 396 (Lee et al., 1991; Goedert et al., 1993; Hasagewa et al., 1992). The known hyperphosphorylated sites in PHF-tau are all located outside the tandem repeat region. Hyperphosphorylated tau is prevented from binding to microtubules and is believed to self-assemble into PHF through interactions between the tandem repeat regions. The phosphorylation state of tau, as with other phosphoproteins, is the result of a balance between protein kinase and protein phosphatase activities. The hyperphosphorylation of tau that is observed in AD is suggestive of a breakdown in protein kinase and/or protein phosphatase regulation. The findings from research on how that regulation might become disrupted in AD are discussed later in Section C of this chapter. B. Etiological hypotheses for Alzheimer's disease Data from various lines of research support the notion that AD is a heterogeneous disorder. The evolving evidence suggests that AD represents a group of phenotypically similar neurodegenerative conditions with different initiating events. Thus, as in cancer or infectious diseases, different strategies may be required for the treatment of etiologically distinct forms of AD. An important theme in our understanding of AD etiology is stratification of groups by age of onset. The dividing line that clinicians have established between early and late-onset cases is age 60. Although less than 10% of the total AD cases are the familial form of the disease (FAD), the majority of those cases tend to be have an early onset. There is an increase in the incidence of the sporadic form of the disease with increasing age of onset. A general model for the disease etiology is: strong mendelian inheritance (4060 yrs); oligogenic (few genes) (60-75 yrs); and then polygenic (many genes) (75 yrs and above). Three major mechanistic hypotheses for the pathogenesis in AD will be reviewed here. Given the heterogeneity in the etiology of the disease it is likely that more than one mechanism is operative. i. Amyloid cascade hypothesis The proponents of the amyloid cascade hypothesis have postulated that mismetabolism of APP and the consequent deposition of A3 is the central event in AD pathogenesis (Hardy & Allsop, 1991). It is postulated that AD involves a decades-long pathogenic cascade that most likely begins with altered production, aggregation, or clearance of AP1-42(43). Two lines of evidence provide the strongest support for the amyloid cascade hypothesis. First, three genes linked to FAD have been identified: the APP gene on chromosome 21; the presenilin 1 (PSI) gene on chromosome 14; and, the presenilin 2 (PS2) gene on chromosome 1. It is now evident that mutations in all three genes alter APP processing in a similar way: they shift the metabolism of APP such that more of the longer A 1-42(43) peptide is produced. The increased production of the longer A31-42(43) peptide is of particular importance because the longer peptide has been shown to be the earliest and most abundant species of A3 in neuritic plaques (Mann et al., 1996), and in biophysical experiments it has been shown to be the species most prone to fibril formation (Jarrett, et al., 1993). Second, several studies have provided strong evidence for a neurotoxic role for the A3 peptides. Yankner et al. (1990) reported that the synthetic A3 peptides A01-3 8, AI31- 40, and AP 2 5-3 5 were neurotoxic in rat hippocampal cultures. Studies by Mattson (1992) with human cortical neurons showed that the AI-.3 8 and AP 25-35 peptides enhanced glutamate neurotoxicity, while a peptide with a scrambled sequence was without effect. The gene encoding APP on chromosome 21 is now known to be the site of at least six different missense mutations that cause an early onset form of FAD (Hardy, 1992). Although the APP mutations are quite rare, their existence has strengthened the argument placing Ap peptide as the central cause of AD. Particularly since all the known mutations are clustered in or immediately around the critical region of APP and lead to changes in the quantity of the longer A3 1-42(43 ) that is proteolytically cleaved from APP. The PS1 and PS2 genes were identified through positional cloning strategies (Sherrington et al., 1995; Levy-Lahad et al., 1995). They are transmembrane (TM) proteins with six to nine TM domains and cytoplasmic N- and C-termini. Like APP, they appear to be expressed ubiquitously and are processed. In most systems that have been examined, the presenilins are cleaved in exon 9 yielding an -25 kDa N-terminus fragment and a - 19 kDa Cterminus fragment. It is not at all clear whether the holoprotein or either or both of the protein fragments are physiologically important. A large number of PS mutations have been described. All but one of these are missense mutations: the single exception is an in-frame deletion of exon 9. The preponderance of mis-sense mutations suggest that the mutant proteins may have a toxic gain-of-function. This gain-of-function might relate to the normal function of the PS or it might be that the mutant PS inactivate the normal allele of the PS. Although the PS mutations occur throughout the protein, there are some patterns in their distribution. First, they generally occur at residues conserved between the PS. Second, the mutations are predominantly found in the TM domains. Third, the cluster of mutations in one TM domain (domain 2) line up on one side of an a-helix, suggesting that this helix may interact with other TM domains or other TM proteins. The "amyloid cascade hypothesis" is presently the dominant hypothesis concerning the etiology of AD, primarily on the strength of the genetic linkage to FAD and the demonstrated neurotoxicity of A3 peptides. However, there are two lines of evidence which diminish the persuasiveness of the hypothesis. First, from pathological studies, amyloid deposition has not been shown to correlate well with degree of dementia (Roses, 1994). In fact, it is an unresolved puzzle why large amounts of A3 deposits can be found in the brains of elderly individuals who show no antemortem evidence of cognitive deficits (Dickson et al., 1991). As will be described in the next section, NFT deposition has proven to be a better correlate of disease progression. Second, behavioral deficits preceded amyloid deposition in transgenic animal studies with mice overexpressing APP with a pathogenic mutation (Hsiao, et a., 1996). ii. Tangle hypothesis The tangle hypothesis relies on three well-established facts about AD. First, hyperphosphorylated tau protein is the principal component of the PHF that is found in neurofibrillary tangles. The evidence in support of this fact has been presented earlier in this chapter. Second, and in striking contrast to the roles of APP or A3 in the amyloid hypothesis, the role of tau protein is known and is vital to the structural integrity and functioning of neurons. Third, the spatial and temporal distribution of NFTs is significantly correlated with disease progression. Proponents of the tangle hypothesis postulate that conversion of normal tau to hyperphosphorylated tau destabilizes microtubules in the neuronal cytoskeleton, leading to a disruption of axoplasmic flow which produces dystrophic changes in affected neurites. Additionally, destabilization of the microtubular system in the perikaryon causes breaking of the Golgi apparatus into small fragments (Steiber et al., 1996). This dispersion of Golgi would be expected to have an effect on processing and targeting of proteins, including APP. In contrast to the variability in the development of AP deposits, the spatial and temporal development of neurofibrillary tangles follows a more predictable pattern. This pattern has been used to define six neuropathological stages of AD (Braak & Braak, 1991). The nerve cells in the brain which develop the initial neurofibrillary lesions are located in layer pre-alpha of the trans-entorhinal region, thus defining stage I. Stage II is a more extensive involvement of this region, as well as a mild involvement of the pre-alpha layer of the enthorhinal cortex. Patients with this pathology are asymptomatic, indicating that Stages I and II represent clinically-silent stages of AD. Mild impairments of cognitive function become noticeable in Stages III and IV. Stage III is characterized by severe neurofibrillary lesions in the pre-alpha layers of both entorhinal and trans-entorhinal regions. The vast majority of nerve cells in those regions show NFTs. In Stage IV, the deep pre-alpha layer develops extensive NFTs. The major feature of Stages V and VI is the massive development of neurofibrillary lesions in isocortical association areas. The principal weakness of the tangle hypothesis is that it has proven to be difficult to identify the mechanism that changes normal tau into PHF-tau. Presumably, the production of hyperphosphorylated tau requires activation of brain kinases and/or inactivation of brain phosphatases. Although many studies (as previously described) have led to the in-vitro identification of candidate kinases and phosphatases, there is a limited number of reports based on in vivo studies. Two. recent reports in the literature have shed some light on the in vivo question. First, Bush et al. (1995) reported that one effect of ATP-depletion in rat pheochromacytoma (PC 12) cells differentiated with nerve growth factor (NGF) is the activation of a tau kinase. This suggested a putative in vivo mechanism, which will be described in more detail below, for the activation of a tau kinase. Second, Trojanowski and Lee (1995) reported that phosphatase activity is suppressed in the neurons of AD victims. It was not clear whether the phosphatases are underproduced or somehow inhibited in their activity. iii. Oxidative energy metabolism hypothesis Despite all the improvements in our understanding of the pathological features of AD, the most significant risk factor for AD (and for several other neurodegenerative diseases) is advancing age. The incidence and prevalence of AD increases steeply with age after age 60, increasing to as much as 47% of patients older than age 85 in one study (Terry & Katzman, 1983). One hypothesis that accounts for both aging and age-dependent onset of neurodegenerative diseases is that an accumulation of mitochondrial genome mutations during life results in a progressive impairment of oxidative phosphorylation. The impairment of oxidative phosphorylation would lead to a decline in ATP, which is likely to have an affect on many cellular processes requiring the high-energy y-phosphate group in ATP. In support of this hypothesis, it has been reported that ATP metabolism is compromised in AD subjects (Hoyer, 1992; 1993; 1994). The human mitochondrial DNA (mtDNA) is a 16,569 base pair (bp) closed circular molecule that codes for 13 of the subunits required for oxidative phosphorylation. The genes for the other subunits are in the chromosomal DNA. The rate of mutation in mitochondrial DNA (mtDNA) is -10 times greater than that in chromosomal DNA (Linnane et al., 1989). mtDNA may be particularly susceptible to age-dependent oxidative damage due to its lack of protective histones, its high information density due to the absence of introns, its close proximity to the inner mitochondrial membrane, where reactive oxygen species are generated, and limited repair mechanisms (Linnane et al., 1989; Ames et al., 1993; Wallace, 1992). Mitochondrial oxidative phosphorylation generates most of the free radicals in cells. Approximately 2% of the oxygen utilized in oxidative phosphorylation is converted into free radicals. An age-dependent increase in oxidative damage to mtDNA in human postmortem brain tissue has been reported by Mecocci et al. (1993). There appears to be even greater oxidative damage in AD patients. Wallace et al. (1991) have reported that there is an approximately two-fold increase in the frequency of a well-characterized 4977 bp deletion in human mtDNA in brain tissue (cortical regions) from AD patients, compared with agematched controls. Mecocci et al. (1994) have shown that there is a significant three-fold increase in the amount of OH'dG in mtDNA in parietal cortex of AD patients, compared with age-matched controls. The potential effects of mtDNA damage have been directly observed in biochemical studies on components of the oxidative phosphorylation system. A 16-26% decrease in cytochrome c oxidase (COX) activity in the cerebral cortex of AD patients was reported by Kish et al. (1992). Characterization of COX in both peripheral tissue and brain of AD patients by Parker and Parks (1995) led to the discovery of a kinetically abnormal COX with an unidentifiable low Km binding site. A weakness of the oxidative metabolism hypothesis is that a causal connection to the two cardinal histopathological lesions in AD, A-3 plaques and NFTs, has not been strongly demonstrated. Hence, it is not clear whether the oxidative damage is primary to the fibrillar protein deposits or vice versa. There are, however, some reports that suggest a primary role for age-dependent declines in oxidative metabolism. Gabzuda et al. (1994) reported that inhibition of energy metabolism in COS cells by mitochondrial uncouplers alters the processing of APP and induces a potentially amyloidogenic derivative. Bush et al. (1995) reported that a tau kinase is activated when ATP is depleted in rat pheochromacytoma (PC12) cells by the mitochondrial uncoupler FCCP. C. Tau protein kinases The degree of phosphorylation is the only known structural difference between PHF-tau and normal adult tau. As described earlier in this chapter, much research has been conducted to identify the abnormal sites that distinguish PHF-tau from normal tau in AD. This research has laid the groundwork for identifying the kinases that might be involved in the generation of PHF-tau in the AD brain. Several criteria have been used to identify the Alzheimer-like state of tau (Hasegawa et al., 1992). First, certain monoclonal antibodies recognize tau from PHFs, but do not recognize normal tau or PHF-tau after dephosphorylation. For example, antibody AT8 depends on the phosphorylation of Serl99 and Ser202. Antibodies SMI31 and SMI34 depend on the phosphorylation of Ser396 and Ser404. Antibodies have also been developed which recognize normal tau, but not PHF-tau. Antibody SMI33 requires an unphosphorylated Ser235, whereas antibody TAU1 requires unphosphorylated Serl99 and Ser202. Second, PHF-tau exhibits a shift in electrophoretic mobility to a higher apparent Mr. Third, tryptic digestion and phosphopeptide sequencing has been used to determine specific phosphorylation sites on tau. These criteria have been used to identify kinase activities in brain extracts which are capable of transforming tau into a state resembling that of PHFs. Several candidate kinases have been identified which might be involved in the conversion of normal tau into PHF-tau. The candidate kinases include the ERK kinases, glycogen synthase kinase-3 (GSK-3), calcium/calmodulin-dependent protein kinase (CaMK II), and cyclic-AMP-dependent protein kinase (PKA). It is likely that a combination of kinases (and/or phosphatases), perhaps some not yet identified, are simultaneously or sequentially involved in the conversion of normal tau to PHF-tau in AD. Roder and Ingram (1991) showed that a 40 kDa kinase, PK40, isolated from bovine brain could induce the characteristic Mr shift in tau. PK40 was shown to be capable of producing the phosphoepitopes on tau that are recognized by the SMI31 and SMI34 antibodies. Later, it was reported that PK40 is a member (ERK2) of the ERK family of kinases (Roder et al., 1995) and was renamed PK40". The ERKs, also known as mitogen activated protein kinases (MAPKs), are highly conserved enzymes which serve as common signaling components in distinct transduction pathways initiated by many stimuli. Drewes et al. (1992) also isolated a 42 kDa ERK kinase capable of transforming tau into the PHF-like state, as judged by several criteria including gel mobility shift, antibody response and phosphorylation sites. The purified enzyme was capable of incorporating -14-16 phosphates per molecule of tau. Mandelkow et al. (1992) showed that the proline-directed kinase GSK-3 can induce an Alzheimer-like immune response involving several distinct and phosphorylatable epitopes at Ser-Pro motifs, as well as a gel mobility shift. Two isoforms of GSK-3 (o, 51 kDa; 0, 45 kDa) had similar effects. GSK-3 is normally involved in the control of insulin-stimulated signal transduction. Surprisingly, GSK-3 is associated with microtubule-associated proteins (MAPs) as defined by the classical criterion of co-purification through cycles of microtubule assembly. GSK-3 also directly associates with PHFs. D. PKA, and ERK2 structures Protein kinases are involved in every aspect of signal transduction in eukaryotic cells, from primary transmembrane signaling to control of transcription and cellular metabolism. Distinct physiological roles have been determined for well-over 60 mammalian kinases, despite the fact that they possess eleven subdomains of sequence identity and are likely to have similar structures. The kinases, however, differ greatly in specificity and mechanism of regulation. How such diverse regulatory mechanisms and specificities are accommodated in the same overall protein fold is an interesting problem. The solution of five protein kinase structures has shed some light on this problem. The first protein kinase structure to be solved was that of mouse cyclic-AMPdependent protein kinase (PKA) in a complex with a substrate analog inhibitor, protein kinase inhibitor (PKI) (Knighton et al., 1991). The structures of four other kinases have since been reported: the cyclin-dependent kinase (CDK2); the extracellular signal regulated kinase (ERK2); the myosin light chain kinase (MLCK) homolog twitchin; and, casein kinase I (CKI). In addition, the structure of a second PKA, purified from porcine heart, has also been reported. The CDK2, ERK2, and twitchin structures were solved in low-activity conformations and in the absence of substrates or substrate analogs, whereas the structure of CKI was solved in an active conformation as a complex with ATP. Only the structures of PKA and ERK2 will be reviewed here. i. General architecture Like metabolic kinases such as hexokinase, the protein kinases have two domains (lobes). The smaller amino-terminal domain consists of a single 0-sheet, while the carboxy- terminal domain is almost entirely composed of ao-helices, so that the overall structure is c/J3. Catalysis occurs at the cleft between the lobes. The function of the small lobe is to bind MgATP leaving the y-phosphate poised for transfer. The larger lobe provides sites for peptide recognition and catalysis. The MgATP binding site is at an interior position between the two domains, whereas the protein or peptide substrate binding site is along the outer edge of the catalytic cleft. The domains close around the substrates to form ternary complexes. Conserved catalytic residues extend from both domains. Those include a glycine-rich 3ribbon containing residues which bind the a- and P-phosphates of MgATP. The kinase structures are most variable outside the kinase core, where the amino- and carboxy-terminal extensions follow completely different paths on the surface of the molecule. ii. PKAct structure The first protein kinase structure to be solved was that of mouse PKA, in a complex with the inhibitor protein PKI. That structure has provided a useful model for the active conformation of the enzyme and its substrate binding mode. Two structures have been solved so far. The binary complex contains the recombinant mouse catalytic subunit and the high affinity inhibitor peptide PKI. The ternary complex contains both MgATP and PKI. PKA serves as the protein kinase archetype structure The smaller lobe, consisting of residues 40 through 125 is associated primarily with the binding of nucleotide and is characterized by a dominance of 0 structure. This domain contains five antiparallel 0 strands and a short helical insert between 1-strands 3 and 5. The larger lobe, in contrast is predominantly helical and has seven a helices. Interestingly, the only region of 0 structure in this lobe is located on the surface of the cleft at the interface between the two lobes where four antiparallel 0 strands form a sheet. Most of the residues which are important for peptide recognition, as well as residues involved in catalysis, are located within this larger lobe. Within the small lobe of PKA, a highly conserved glycine-rich loop extends between f-strands 1 and 2 (Lys72 and Glu91) and contributes to nucleotide binding and, in particular to stabilizing the portion of the nucleotide that is not directly involved in phosphotransfer. Zheng et al. (1994) showed that several residues bind the MgATP in the PKA*PKI peptide*MgATP ternary complex. The adenine ring is enclosed in a hydrophobic pocket, formed at the domain interface, consisting of residues Leu49, Va157, Ala70, Metl20, Tyr122, Val123, and Leu173. Two hydrogen bonds serve to anchor the adenine ring in this hydrophobic pocket. The first hydrogen bond contact is between the N6 amino group and the main-chain carbonyl of Glu 121. The second hydrogen bond contact is between the N7 nitrogen and the side-chain hydroxyl of Thr183. Two hydrogen bonds also stabilize the ribose ring. The oxygen of the 2'-OH is located 2.6 A from the side chain of Glul27. The 3'-OH is 2.6 A from the ox-carbonyl oxygen of Glu 170. iii. ERK2 structure The solved structure of the unphosphorylated low-activity conformation of recombinant ERK2 was reported by Zhang et al., (1994). ERK2 achieves maximum activity only when both Thr183 and Tyr185 are phosphorylated through the action of MEK, a dual specificity kinase (Anderson et al., 1990; Ahn et al., 1991). The singly phosphorylated forms of ERK2 have less than 1% of the activity of the fully phosphorylated enzyme (Robbins et al., 1993). The structure spans the conserved kinase homology region (Hanks et al., 1988) and has N- and C-terminal extensions that lie on the surface of the molecule. The two domains of unphosphorylated ERK2 are farther apart than in the active conformation of PKA. The discrepancy is a result of rotation of the N-terminal lobe of unphosphorylated ERK2 by 170 relative to that of active PKA, about an axis passing between the two domains, which leaves the active site of ERK2 more open (Zhang et al., 1994). This suggests that activation of ERK2 is likely to involve both global and local conformational changes. Domain rotations upon binding of substrate have been postulated for PKA (Cox et al., 1994). Zhang et al. (1994) incubated crystals of ERK2 for up to 6 hours in MgATP or the non-hydrolyzable analogue AMP-PNP to obtain data for nucleotide-bound forms of unphosphorylated ERK2. The nucleotides were loosely bound deep within the N- and C-terminal domain interface. The N6 amino group of the adenine ring is hydrogen bonded to the backbone carbonyl of Asp 104. The ribose 02' and 03' hydroxyls form a hydrogenbonding network to Asp 109(Glu 127) and Lys 12. No reorientation of the domains occurred upon ATP binding and the interactions of the glycine-rich loop with the phosphates of ATP are absent. E. PKAcat and ERK2 regulation Protein kinases are regulated by a plethora of mechanisms, consistent with the diverse and complex functions which they regulate. Many protein kinases, particularly those which are under the control of a second messenger, possess a regulatory domain containing a pseudosubstrate sequence which binds to the catalytic site and prevents access of external substances to the catalytic site. Binding of the second messenger to the regulatory domain releases the internal inhibitory effect (presumably by a conformational change). Examples of this regulatory mechanism include PKA (regulated by cAMP), the calcium/calmodulin kinases (CaM) (regulated by calcium), and protein kinase C (regulated by calcium, phosphatidylserine, diacylglycerol, and free fatty acids). For other kinases, phosphorylation of the catalytic domain is required for enzymatic activity. Examples of this mechanism include the ERKs, S6 kinases, and phosphorylase kinase. Autophosphorylation, occurring either as an intrachain reaction or between two adjacent peptide chains, also occurs. Examples of this include ERK2 and CaM kinase II. Interactions with other proteins also serve to regulate protein kinase activity, as is the case with cyclin-dependent kinases and c-raf Protein kinases have also been shown to be regulated by many different types of ligands and metals which are not second messengers. i. PKAct regulation Hormones and other agents with receptors that signal through heterotrimeric G proteins control the production and release of second messengers such as cAMP, diacylglycerol and intracellular calcium. The release of cAMP leads to the release of PKA from pseudosubstrate inhibition via its regulatory subunit. The PKA holoenzyme consists of two cAMP-binding regulatory subunits and two protein kinase catalytic subunits (Zheng et al., 1993). In the absence of cAMP, the enzyme is sequestered as an inactive tetrameric complex containing both regulatory (R) and catalytic (C) subunits. Upon binding of cAMP, the holoenzyme (R 2C2) dissociates into an R2-cAMP 4 dimer and two active, monomeric C subunits (aka PKAct). There appear to be at least four genes encoding the regulatory subunit isoforms and three separate genes encoding the catalytic subunits (Scott, 1991). The pseudosubstrate sequences in the regulatory subunits of PKA act as an autoinhibitor. Activation of the protein kinase is achieved by removing the pseudosubstrate from the active site and allowing protein substrate access. This form of regulation has been termed intrasteric regulation to emphasize that it is occurring within the active site as opposed to acting at another site, and that the pseudosubstrate interaction mimics some aspects of the substrate's structure. The activity of PKA is also influenced by phosphorylation, particularly at a conserved threonine reside (Thr197 in mouse PKA). Phosphorylation at this site is required both for effective regulatory subunit binding and for maximum catalytic activity (Steinberg, 1993). This site is probably autophosphorylated soon after translation; once the phosphate is in place, it is extremely stable and does not turn over at a significant rate. Thus, phosphorylation of PKA can probably be viewed more as a protein maturation event than as a regulatory event. ii. ERK2 regulation The ERK kinases are a family of enzymes which are activated by rapid tyrosine phosphorylation when a wide range of extracellular stimuli are added to quiescent cells. These stimuli include almost all of the known growth factors that bind to receptor tyrosine kinases and lymphokines that engage receptors that activate tyrosine kinases (Cobb et al., 1991). The ERK kinases are also activated by several agonists that stimulate the hydrolysis of polyphosphoinositides and phosphatidylcholine leading to the activation of protein kinase C, and by inhibitors of protein-serine/threonine phosphatases, protein-tyrosine phosphatases and microtubule assembly (Cobb et al., 1991). ERK2 (and also ERK I) require phosphorylation of threonine and tyrosine residues for maximal activation. For ERK2 the phosphorylation sites have been localized to Thrl83 and Tyr 185 in mouse (Payne et al., 1991) and to Thrl88 and Tyrl90 in X. laevis (Posada & Cooper, 1992). The location of the regulatory site just before the highly conserved nucleotide binding region suggests that phosphorylation within this region renders the enzyme more accessible to ATP. Studies in which the Thrl88 and Tyrl90 phosphorylation sites in X. laevis ERK2 have been mutated have demonstrated that their phosphorylations are not interdependent (Posada & Cooper, 1992). Two potential mechanisms of ERK2 activation have been suggested. Purified recombinant ERK2 autophosphorylates in a manner that generates a small amount of tyrosineand threonine-phosphorylated enzyme (Robbins & Cobb, 1992). This has suggested that the enzyme may autoactivate. A second mechanism has been suggested by the identification of an enzyme, known variously as MAP kinase/ERK activator or ERK kinase which appears to catalyze the phosphorylation of both the tyrosine and the threonine required for maximum activity of ERK2 (Seger et al., 1992). Evidence has accumulated that the ERK2 kinase is itself a protein kinase with a very restricted substrate specificity (Seger et al., 1992). It has been reported that ERK2 kinase is regulated via phosphorylation by p74ran (Dent, 1992). There is some evidence for physical association of activated p7 4 ral with the ligand occupied platelet-derived growth factor receptor, however other signal transducing proteins may also interact with p74raf (Ullrich & Schlessinger, 1990). F. PKAcat mechanism Protein kinases are phosphotransferases which catalyze the transfer of the y-phosphoryl group of the substrate MgATP to an amino acid side chain of a protein co-substrate. Phosphoryl acceptors include the alcohol group of serine and threonine, and the phenol group of tyrosine. The catalytic subunit of PKA catalyzes the phosphorylation of a variety of protein substrates according to the general reaction described in Scheme 2-1. MgATP + protein - phosphoprotein + MgADP (Scheme 2-1) A common feature of the amino acid sequence of many PKA substrates is the presence of one or two arginyl residues on the amino-terminal side of the phosphorylated serine or threonine. The best substrate found for the kinase is the Ser-peptide (Leu-Arg-Arg-Ala-Ser-Leu-Gly) (Hjelmquist et al., 1974). The kinetic mechanism for the PKA catalytic subunit has been suggested by Cook et al., (1982) to be random sequential in the direction of protein substrate phosphorylation, with ordered release of phosphopeptide and MgADP. This conclusion resulted from initial velocity studies in the absence and presence of the product MgADP, other nucleotide analogues (adenosine and AMP), and guanethidine (a peptide substrate analogue). Adenosine (Ki = 35 rM), AMP (Ki = 640 pM), and MgADP (Ki = 14 p.M) were found to be competitive versus MgATP at 10 mM free Mg 2+. Two combinations of adenosine with enzyme were suggested by the inhibition kinetics patterns: one with free enzyme and one with the enzyme*Ser-peptide complex. The first was competitive and the second uncompetitive to produce the net noncompetitive inhibition observed with respect to varying Ser-peptide. Similarly, two combinations of guanethidine were allowed (one with free enzyme and one with the enzyme*MgATP complex) to produce noncompetitive inhibition versus MgATP. Kong and Cook (1988) later used isotope partitioning experiments at low (0.5 mM) and high (10 mM) free Mg 2+ to directly show that PKAcat enzyme is capable combining with either MgATP or Ser-peptide. The mechanism was determined to be random sequential at both low and high free Mg2+ concentrations, with the pathway in which MgATP adds to the enzyme first being preferred at high free Mg 2+ . An alternative mechanism in which ordered addition of MgATP and peptide is followed by an ordered release of phosphopeptide and MgADP has also been proposed (Whitehouse & Walsh, 1983; Whitehouse et al., 1983). The alternative conclusion resulted from experimental results that were very similar to those obtained in Cook et al. (1982) and Kong and Cook (1988), with one exception. In initial velocity studies, the ATP analogue, AMP-PNP was determined to be a competitive inhibitor versus MgATP and a noncompetitive inhibitor versus the peptide substrate Kemptide. The Kemptide analogue Ala-peptide was found to be noncompetitive versus MgATP and noncompetitive versus Kemptide (at 1 jM fixed [MgATP]). That result led the authors to conclude that Kemptide and Ala-peptide competed for different forms of the enzyme at low ATP concentrations. An ordered sequential mechanism would require that Ala-peptide combine with free enzyme and the enzyme*MgATP*Kemptide complex; and that Kemptide combine only with the binary enzyme*MgATP complex (see Appendices 8A and 8B for further explanation of the kinetic properties of random and ordered sequential bisubstrate reactions, respectively). G. Regulation of kinases by ATPee The first report of an enzyme that is inhibited by ATPfree is phosphofructokinase, the second enzyme in glycolysis (Lowry & Passoneau, 1966). This enzyme is regulated in a complex manner presumably to control the amount of ATP produced in a cell by controlling glycolysis and thereby the citric acid cycle and oxidative phosphorylation. ATPfee was found to be much more inhibitory than the substrate MgATP (product inhibition) and the inhibition was increased or decreased by a number of small molecules. Based on enzyme kinetic studies, Lowry and Passoneau postulated the existence of a single allosteric ATPee inhibitor site. In studies with phosphofructokinase from Plasmodium berghei, Buckwitz et al. (1990) also found allosteric inhibition by ATPe, binding to a single inhibitory site. The Ki determined for ATPaf was 720 iM. More recently, it was reported that the protein kinase PK40 , 2 (ERK2) is strongly inhibited in the presence of millimolar concentrations of ATP in excess of total magnesium (Roder & Ingram, 1991). This finding is particularly relevant to Alzheimer's disease, since ERK2 is capable of hyperphosphorylating the cytoskeletal protein tau and ATP levels decline in AD. There were two likely explanations for the reduced activity of ERK2 when ATP is added in excess of Mg 2+: inhibition by ATPee, or a loss of activation by free Mg 2+. Furthermore, it prompted interest in whether the inhibition by ATPfee applied to other tau kinases. And if so, whether the degree of inhibition would be the same. Unpublished work by B. Blanchard (1994) in this laboratory had shown that the inhibition ATPfee extended to at least two other tau kinases, PKA and PKC. The biochemical studies needed to further characterize the observed reduction in ERK2 activity formed the basis for the research work to be presented later in Chapters 3, 4, and 5. H. Regulation of kinases by free Mg2+ Buckwitz et al. (1990) also observed that phosphofructokinase from Plasmodium 2 berghei was inhibited by high (8 mM and 16 mM) concentrations of free Mg +. The inhibition was not caused by the increase in ionic strength. Free Mg2+ has been shown to activate the catalytic fragment of PKC, with ATPaf being neither a substrate or inhibitor (Hannun & Bell, 1990). Free Mg 2+, in the range of 1-10 mM, activated the catalytic fragment of PKC with half-maximal activation obtained at 1 mM free Mg 2+. Hannun and Bell also found that PKC activity was -90% inhibited by 30 mM divalent ion ZnCl 2; indicating an ionic strength inhibition effect at high salt concentrations or an inhibitory effect from Zn 2+ combining with free -SH groups on the PKC enzyme. Chapter 3: Inhibition of PKAcat by ATPfre. A. Experimental procedures i. Materials The catalytic subunit of cAMP-dependent protein kinase (PKAcat), myelin basic protein (MBP), ultrapure disodium ATP, adenosine, and uridine were purchased from Sigma Chemical Company. [y- 33 P]ATP with a specific activity of 10 mCi/ml was obtained from Dupont/NEN. Phosphocellulose filters (P81) were purchased from Whatman. Liquiscint scintillation cocktail was purchased from National Diagnostics. ii. Purification of recombinant hTau40 Recombinant human tau protein hTau40 (derived from the cDNA clone of Goedert in the pRK172 vector) was expressed and purified from Escherichia coli BL21 cells as follows: BL21 cells containing the hTau40 cDNA were grown in LB medium at 37 C to an OD600 of 0.6-1.2, then induced with 0.4 mM IPTG and harvested after 2 hours. The cell pellet from 8 liters of culture was resuspended in 200 ml of 50 mM PIPES, pH6.8 containing 1 mM DTT, 1 mM EDTA, 0.2 mM PMSF and 0.5 mg/ml lysozyme. The resuspended cells, in a 250 ml centrifuge bottle, were then sonicated over ice for 3 X 15 min with two 5 minute intermissions. The supernatant was divided into four 50 ml aliquots and chromatographed on four phosphocellulose columns (3 X 1 mm) equilibrated with 50 mM PIPES, pH6.8 containing 1 mM DTT, 1 mM EDTA, 0.2 mM PMSF. Fractions containing hTau40 protein as determined by 10% SDS-PAGE were pooled and concentrated to approximately 1 ml using Amicon Centriprep 30 and Centricon 30 ultrafiltration tubes. The final concentrate was dialyzed overnight against 20 mM HEPES buffer. Total protein was determined by the Pierce Coomassie Plus assay kit. iii. Phosphorylation assay for PKAcat Initial velocity phosphorylation assays were performed in 100 mM HEPES (pH7.0) and 1 mM DTT. The reaction was initiated by the addition of 0.001 unit of PKAcat. After incubation at 30 C for 4-8 hours, 20 tldaliquots of the reaction mix were spotted onto 2.4 cm diameter phosphocellulose disks to stop the reaction. The phosphocellulose disks were then washed four times for 5 minutes in 75 mM phosphoric acid (10 ml/disk). Afterwards, the phosphocellulose disks were washed for 1 minute with acetone (5 ml/disk) before being dried under a heat lamp for at least 5 minutes. Radioactivity was measured by liquid scintillation counting (Beckman LS 2500) with 5 ml of"Liquiscint" for 1 min. iv. Equilibrium binding assay for PKAcat Equilibrium dialysis was performed in a polycarbonate apparatus (Micro Filtration Products Inc.) containing two halfspheres of 150 ml volume, separated by a dialysis membrane. Both chambers contained 120 tld of 100 mM HEPES, pH 7.0 and 1 mM DTT. One chamber contained 0.4 ipM PKAcat. The other chamber contained the total (bound+unbound) amount of [y- 33P]ATP (72,000 - 360,000 cpm/pmol) for the experiment. After dialysis for 24 hours at 4 oC with gentle shaking, 1 pl1 aliquots were taken from each chamber and measured by liquid scintillation counting (Beckman LS 2500) with 5 ml of "Liquiscint" for 1 min. v. Calculation of ATPfee concentrations The experimental concentrations of MgCl 2 and Na2ATP used to obtain the indicated concentrations of MgATP and ATPfre were calculated using the computer program SPE (available in Martell & Motekaitis, 1992). The program calculates species distributions as a function of pH and total ion concentration. The program requires as input critical stability constants (-log K.q) for all relevant species equilibria and the total concentrations of each ion in solution. The critical stability constants used were as follows: MgATP/Mg 2+ ATP, 4.06; MgHATP/MgATPH', 4.55; HATP/H+ATP, 6.51; H2 ATP/HATPH, 4.06 (Martell & Smith, 1975). The program was executed on an IBM-compatible computer. vi. Determination of sequential kinetics parameters for random and ordered sequential bireactant reactions Initial velocity data were analyzed using the ENZYME KINETICSTM program on an IBM-compatible computer. The ENZYME KINETICSTM program, written by R. Viola, was purchased from Sci Soft. The ENZYME KINETICSTM program calculates kinetic parameter estimate for unireactant and bireactant reactions. The mechanistic models that can be fitted by the program are random sequential, ordered sequential, and ping-pong. Additionally, the program can fit unireactant inhibition data to competitive, noncompetitive, uncompetitive inhibition models. The curve fitting is performed using weighted nonlinear regression, with weights of I/velocity. The equilibrium relationships for a random and ordered sequential bireactant reactions are shown in Schemes 3-1 and 3-2. The two substrate enzyme kinetic data was fitted to the EAB k- aKA EB +B +BKB /aK, KA E -A _ EA Scheme 3-1: Equilibria for a Random Sequential Bireactant Reaction Products EAB KE +B KA E -A -EA Scheme 3-2: Equilibria for an Ordered Sequential Bireactant Reaction v Products equations for random (Equation 3-1) and ordered (Equation 3-2) sequential bireactant mechanisms, ~V =[A][B]Vmax KKB+ aKA[B] + aKB[A] + [A][B] [A][B]V max (3-1) (3-2) KAKB + KB[A] + [A][B] where V is the initial velocity, Vmx is the maximum initial velocity, and a, KA, and KB are defined in Schemes 3-1 and 3-2. vii. Determination of the effect of ATPfr,. on the slopes and intercepts of Lineweaver-Burk plots of PKAcat initial velocities The totality of possible equilibrium relationships for inhibition of random or ordered sequential bireactant reactions by inhibitor I are shown in Schemes 3-3 and 3-4. There are 15 alternative inhibition mechanisms for such each system. The 15 result from all possible combinations of inhibitor complexes. Tables 3-1 (random sequential) and 3-2 (ordered sequential) summarize the effects of varying inhibitor I on the slopes and intercepts of Lineweaver-Burk plots of kinetics data when one substrate is varied while the other substrate is fixed. Detailed rate equations are provided in Appendix 8A (random sequential) and 8B (ordered sequential). The Origin 4.1TM program on an IBM-compatible computer was used to perform multiple regression analysis on the Lineweaver-Burk transformed data. EBI cyKA EABI El EAB +I E EA Scheme 3-3: Equilibria for inhibition of a random sequential bireactant reaction by inhibitor I 0- Products EBI myKA EABI y7K, El EAB -v +I E EA Scheme 3-4: Equilibria for inhibition of an ordered sequential bireactant reaction by inhibitor I Products Table 3-1. Effects of varying inhibitor I on the slopes and intercepts of Lineweaver-Burk plots of a random sequential bireactant reaction Varying substrate B Fixed substrate A Varying substrate A Fixed substrate B Inhibitor Complexes formed in Varying I affects L-B plot Reaction Pathway EABI slopes intercepts pattern EBI EAI El Varying I affects L-B plot slopes intercepts pattern Co I SCo 0 / UN / INC UN NC NC " " / F-, / / 4 _ " " _ " " CO / UN UN / NC NC NC NC NC NC / / / / NC NC NC 1 _ " " " NC NC " / 1 UN co CO " SCO I I I T " " NC NC NC NC NC Table 3-2. Effects of varying inhibitor I on the slopes and intercepts of Lineweaver-Burk plots of an ordered sequential bireactant reaction Varying substrate B Fixed substrate A -- Varying substrate A Fixed substrate B Inhibitor Complexes formed in Reaction Pathway Varying I affects L-B plot El EAI EBI EABI slopes intercepts pattern CO V S I L II I II V NC CO V CO I / V V V " " " , " INC NC I NC " NC NC NC " " NC NC NC NC NC " V ; I CO V UN [ I Varying I affects L-B plot slopes intercepts pattern I UN I UN I UN NC II V" I1 I / V / " _ " / I 1 CO CO I I co " / " NC NC NC / , " NC NC NC NC NC ~_~li_~ ;__~III__ _i _i__YI(~.~ The following multiple regression model was used to test the effect of ATP&ee on the slopes and intercepts of the Lineweaver-Burk plots.: -_ m-+ += IDI+02D 2 Vo Vm +3DI+ +- S 4D2 (3-3) Vm where Km is the apparent Michaelis constant, Vo is the initial velocity, Vm* is the apparent maximal initial velocity, 31 and 32 are slope effect coefficients, 03 and 04 are intercept effect coefficients, and D 1 and D2 are paired variables which take on row orthogonal values of 0 or 1 for any three concentration levels of ATPce. To calculate inhibition constants, LineweaverBurk slopes and intercepts were replotted according to the determined rate equation. viii. Determination of the effect of free Mg2+ on the slopes and intercepts of Lineweaver-Burk plots of PKAcat initial velocities The totality of possible equilibrium relationships for non-essential activation of random and ordered bireactant reactions by activator M are shown in Schemes 3-5 and 3-6, respectively. A non-essential activator binds to all forms of an enzyme and enhances the rate of catalysis of the central complex (EAB). For both random and ordered sequential mechanisms, varying non-essential activator M affects both the slopes and intercepts of Lineweaver-Burk plots of kinetics data when one substrate is varied while the other substrate is fixed. Detailed rate equations are provided in Appendices 8C (random sequential) and 8D (ordered sequential). otyKA Ok EABM -O Products YPKM EAB E - Products EA Scheme 3-5: Equilibria for activation of a random sequential bireactant reaction by non-essential activator M Okp EABM - Products Y KM EAB E kEABProducts - Products EA Scheme 3-6: Equilibria for activation of an ordered sequential bireactant reaction by non-essential activator M ix. Calculation of equilibrium binding parameters for PKAcat Equilibrium binding data were analyzed using the OriginTM 4.1 program on an IBM- compatible computer. The data were fitted to the equation for simple equilibrium binding of ligand to protein: [PL] = [Lu][Pt] Kd + [Lu] (3-4) where [PL] is the concentration of bound ligand, [Pt] is the total concentration of protein, [La] is the concentration of unbound ligand, and Kd is the equilibrium dissociation constant. B. Results Previous experiments (unpublished data from Barbara Blanchard) in this laboratory suggested that the in vitro activity of PKAcat is inhibited by up to 80% when 4 mM ATP4 is added to the reaction containing 2 mM Mg2+ and 2 mM ATP 4-. The inhibitory effect could only be partially explained by an ionic strength effect, based on experiments with NaCl added to comparable ionic strength levels. Further interpretation of those results was impossible due to the nature of the species equilibrium relationships in those experiments. One complication with experiments where total Mg 2+ is held constant while total ATP 4- is increased is that there is a concomitant decrease in the concentration of free Mg2+ due to the law of mass action. Under those conditions, it is impossible to determine whether the loss of activity is due to an increase in a putative inhibitor, ATPf,ce, or to a decrease in a putative non-essential activator, free Mg 2+ . Non-essential activation of a kinase refers to activation that is separate from that required for activation required for transfer of the g-phosphate group of ATP4 . Another complication is that the concentration of MgATP, a co-substrate, also increases with increasing total ATP 4-. The simultaneous changes in the concentrations of MgATP, ATPfee, and free Mg2+ makes it impossible to unambiguously interpret the experimental results. To avoid the problems associated with interactions between the three species, MgATP, ATPee, and free Mg2+, critical stability constants were used to calculate actual equilibrium concentrations. Furthermore, the calculations were used to determine conditions where the concentration of one species could be held constant while varying the concentrations of the other two at a given pH. Greater control over the MgATP, ATPree, and free Mg2+ species concentrations in the kinase assay permitted the careful design of inhibition kinetics experiments for elucidating the mechanism of inhibition. The general experimental strategy was to perform an initial set of experiments to characterize the kinetic properties of the enzyme, with respect to MgATP and hTau40 as substrates, in the absence of the putative inhibitor (total Mg 2+= 10 mM) and then to perform two types of inhibition experiments. The first set of inhibition kinetics experiments involved fixing hTau40 concentration while MgATP and ATPfre,(.'. also free Mg 2+) concentrations were varied. The second set inhibition kinetics experiments involved fixing MgATP concentration while hTau40 and ATPfrte (.'. also free Mg2+) concentrations were varied. Two strategies were employed to analyze the experimental data. Data from the inhibitor-free experiments were fitted to the equation (Equation 3-1) for a random sequential bireactant system in order to directly estimate kinetic parameters. Data from the inhibition experiments were transformed to Lineweaver-Burk form and fitted to an equation (Equation 3-2) which determined the effect of inhibitor on slope and intercept parameters of the Lineweaver-Burk plots. The large number of parameters in the rate equations for inhibition of a two-substrate reaction did not allow for adequate model discrimination by nonlinear regression analysis. Furthermore, the data could not be fitted to rate equations for inhibition of a single-substrate reaction because of the inability to work at saturating concentration of hTau40, due to protein solubility considerations. Determining the effect of inhibitor on slope and intercept parameters of the Lineweaver-Burk plots proved to be the most appropriate method for discriminating between alternative inhibition mechanisms and for calculating inhibition/activation constants. i. Two-substrate kinetics with MgATP and hTau40 Initial velocity studies, in the absence of ATPfree, were performed over a wide range of MgATP and hTau40 concentrations (25 to 500 jtM and 15 and 200 iM, respectively), in order to assess the catalytic mechanism and the kinetic constants of the uninhibited phosphorylation reaction. Total Mg 2+concentration was fixed at 10 mM. In Figure 3-1A, initial velocity (represented as the picomoles of phosphate incorporated over time) is plotted versus MgATP at constant hTau40. In Figure 3-1B, reciprocal initial velocity is plotted versus the reciprocal of MgATP at constant hTau40. The data were fitted to the equations for random (Equation 3-1) and ordered (Equation 3-2) sequential kinetics. The results are shown in Table 3-3. The analysis favored either an ordered sequential mechanism, with MgATP binding first or a random sequential mechanism. An ordered sequential mechanism with hTau40 binding first gave the poorest fit, with negative estimates being obtained for KhTau40 and CKMgATP. An ordered sequential mechanism with hTau40 binding first was ruled out because of negative parameter estimates for KhTau40 and aKMgATP. The random sequential model, however, could not be completely ruled out based on the large standard errors of FIGURE 3-1. Dependence of PKAcat initial velocity on [MgATPI at different fixed values of [hTau40]. The assay for protein kinase activity was performed with 0.06 U PKA catalytic subunit plus the indicated concentrations of MgATP and hTau40. Conditions were 2+ as described under "Experimental Procedures" except that total Mg concentration was fixed at 10 mM while ATP 4 concentration was varied at fixed hTau40 concentrations of 15 (0), 30 (0), 50 (*), 100 (V), and 200 (A) t M. The data for each point is the average of two measurements. Error bars are +/-1 SD. (A) Michaelis plots of the dependence of PKA activity on MgATP and hTau40. (B) Lineweaver-Burk plots of the activity data presented in (A). pM hTau40 0.8 200 0.7 0.6 100 0.5 0 a) E0 O CL o 0.4 0.3 _50 E 0.2- S--- 30 --- 15 0.1 0.0 -I 100 200 300 tM MgATP FIG 3-1A 400 500 600 gM hTau40 15 20 1816 ,14 0. Q 12 0 E lo o 0 E 30 8 6- 50 4 100 2 200 0.00 0.01 0.02 0.03 (rM MgATP) FIG 3-1 B 0.04 0.05 Table 3-3. Estimated kinetic parameters for PKAcat Catalytic Mechanism Ordered MgATP binds first Ordered hTau40 binds first 1.6 (0.2) 1.3(0.1) 5.3 (0.06) 20 (8) 189 (147)" 24 (14) a 35 (6) 223 (46) 0.02 169 (24) 0.02 Random Vmax (pmoles/min) KMgATP (iM) KhTau4O (JIM) aKMgATP (pM) aKhTau40 (pM) RMSE -181 (29) -53 (12) 0.08 Parameter estimates were determined as described in Chapter 3 "Experimental Procedures." PKAcat concentration was 0.001U. Total Mg 2+ concentration was 10 mM. MgATP and hTau40 concentration ranges were 25-500 CtM and 15-200 pM, respectively. Standard errors are in parentheses. 'Not significantly different from zero by Student t test at t=5%. parameter estimates because the imprecision could have resulted from our inability to achieve higher (> 200 pM) concentrations ofhTau40 due to protein solubility and pH buffering constraints. ii. Kinetics of inhibition with varying ATPf,e. and MgATP Inhibition kinetics studies were performed at a fixed concentration of hTau40 (20 pM) with varying concentrations of MgATP and ATPfre. In Figure 3-2A, initial velocity (represented as the picomoles of phosphate incorporated over time) is plotted versus MgATP at constant ATPfe. In Figure 3-2B, reciprocal initial velocity is plotted versus the reciprocal of MgATP at constant ATPee. The effect of inhibitor on slope and intercept parameters of the Lineweaver-Burk plots is shown in Table 3-4. ATPr, affected only the slopes of the Lineweaver-Burk plots. In these experiments, free Mg 2+concentration varied inversely to ATPf., 2 concentration, in the range of 0.01 mM to 9.5 mM. If free Mg +were acting as a non- essential activator, double reciprocal plots from an experiment where MgATP concentration is also varied would be predicted to have the same slopes but different intercepts (see Appendices 8C and 8D for the rate equations for non-essential activation of random and ordered sequential bireactant reactions, respectively); this was not observed (see Table 3-4). The pattern observed, however, was consistent with ATPfee acting as a competitive inhibitor of MgATP. FIGURE 3-2. Dependence of PKAcat initial velocity on [MgATP] at different fixed values of [ATPJe,] the assay for protein kinase activity was performed with 0.06 U PKA catalytic subunit plus the indicated concentrations of MgATP. Conditions were as described under "Experimental Procedures" except that hTau40 concentration was fixed at 30 p.M at ATP ee concentrations of 5 (0) , 15 (0), and 500 (*) pM. The data for each point is the average of two measurements. Error bars are +/- 1 SD. (A) Michaelis plots of the dependence of PKA activity on MgATP and ATPfree. (B) Lineweaver-Burk plots of the inhibition data presented in (A). pM ATPfree 0.7 500 0.61 0.5 0 - 0. 3 E o 0.2 0.1 0 100 200 300 iM MgATP FIG 3-2A 400 500 600 IM ATP ree 25 500 20 0 15 U) E 0 10 E - 15 5 -5 0.000 FIG 3-2B 0.005 0.010 0.015 (pM MgATP)- 0.020 0.025 Table 3-4. Effect of ATPfr. on Lineweaver-Burk plots for PKAcat, ERK2, and GST-ERK2 Varying MgATP " and ATPfree " Unequal Unequal intercepts slopes Pattern (p-value) (p-value) PKAcat ERK2 GST-ERK2 <0.0001 0.11 <0.0001 0.26 0.49 <0.01 CO CO CO Varying hTau40 and ATPfree Unequal Unequal intercepts slopes Pattern (p-value) (p-value) 0.01 <0.005 <0.002 0.01 0.04 <0.001 NC NC NC iii. Kinetics of inhibition with varying ATPf,ee and hTau40 Inhibition kinetics studies were performed at a fixed concentration of MgATP (50 tM) with varying concentrations of MgATP and ATPfree. In Figure 3-3A, initial velocity (represented as the picomoles of phosphate incorporated over time) is plotted versus hTau40 at constant ATPree. In Figure 3-3B, reciprocal initial velocity is plotted versus the reciprocal of hTau40 at constant ATPree. The effect of inhibitor on slope and intercept parameters of the Lineweaver-Burk plots is shown in Table 3-2. ATPfr, affected both the slopes and intercepts of the Lineweaver-Burk plots. For an ordered sequential mechanism with MgATP binding first, that L-B pattern is compatible only with ATPce being noncompetitive versus hTau40. For a random sequential mechanism that pattern is compatible with ATPe being either noncompetitive or uncompetitive versus hTau40. Graphical analysis (Figure 3-3C) of the varying hTau40 and varying ATPfe inhibition kinetics data, using rate equations for noncompetitive inhibition of an ordered sequential reaction or noncompetitive inhibition of a random sequential reaction yielded Ki's of 12 ± 0.2 itM and 6 ± 0.1 average value for Ki was 9 pM. M, respectively. The FIGURE 3-3. Dependence of PKAcat initial velocity on [hTau40] at different fixed values of [ATPfree]. The assay for protein kinase activity was performed with 0.06 U PKA catalytic subunit plus the indicated concentrations of hTau40. Conditions were as described under "Experimental Procedures" except that MgATP concentration was fixed at 50 pM at ATPafe concentrations of 5 (N) , 15 () , and 50 (*) mM. The data for each point in Figures 3-3A&B is the average of two measurements. Error bars are +/- 1 SD. (A) Michaelis plots of the dependence of PKA activity on hTau40 and ATPfree. (B) Lineweaver-Burk Plots of the inhibition data presented in (A). (C) Replot of Lineweaver-Burk Slope*Intercept versus [ATPfree]/[MgATP]. The line was fitted by linear regression and the correlation coefficient was 0.99. 0.7 pM ATPfree 0.6 0.5 0.4 CL 0& S 0.3 " I 15 050 O 0.2 50 0.1 0.0 0 25 50 1M FIG 3-3A 75 hTau40 100 125 LM ATPfree 50 0 15 E 0 E 0.00 FIG 3-3B 0.01 0.02 0.03 ( M hTau40) 0.04 0.05 0.06 350 300 250 2200 a) C150 O 100 50 l 0.0 I 0.2 ' I I I 0.4 0.6 [ATPfree]/[MgATP] FIG 3-3C I • 0.8 1.0 iv. Equilibrium binding of ATP,ee to PKAcat The inhibition experiments were complemented by equilibrium dialysis studies to directly measure the affinity of the PKAcat for ATPfree. Figure 3-4A shows that saturable binding of ATPe, to PKAcat occurred over the range of ATPfe concentrations indicated. Figure 3-4B shows a Scatchard plot of the data presented in Figure 3-4A. The maximum ATPfo. bound was 1 mol per mol of PKAat based on a nonlinear regression fit of the data to Equation 3-3. The dissociation constant was estimated to be 1.1 4jM. The observed binding of ATPfree to the free PKAcat enzyme ruled out a random sequential mechanism with uncompetitive inhibition by ATP&, (see Table 3-1). v. Equilibrium binding of ATPfr. to the PKAcat*hTau40 complex Equilibrium binding experiments were performed in order to assess the affinity of ATPo, for a PKAcat*hTau40 complex. Initial experiments indicated that the binding affinity of ATPfr:, for the PKAcat*hTau40 complex was lower than for free PKAcat. This necessitated the use of higher concentrations of both ATPe,, PKAcat enzyme (4 pM), and hTau40 in the binding experiments. Time constraints, as well as enzyme and hTau40 availability, prevented the performance of sufficient experiments to produce a saturation binding curve for ATPfre binding to the PKA,.t*hTau40. Table 3-5 shows the results of the experiments that were performed. Binding of ATPfo to the PKAat*hTau40 complex was weaker than to the free PKAcat enzyme. An approximate value of Kd, based on the concentration of [unbound ATP] giving 50% binding of the PKAcat*ATPfe, complex, is 18 jLM. FIGURE 3-4. A: Binding of ATPfree to PKAcat. Assays were performed as described under "Experimental Procedures." [PKAt]o=0.4 pM, 1 mM DTT, pH 7.0, 4 C. The solid line is the fitted curve drawn according to equation 3-1 with Kd= 1.1 LM and Bmax=0.94 mol ATP per mol PKAcat. The data for each point is the average of five to ten measurements. Error bars are +/- 1 SD. B: Scatchard plot of ATPfre binding to PKAcat. The line was fitted by linear regression and the correlation coefficient was -0.97. 1.0 I 0.8 0.6 0 -5 E 0a 0.4 C -0.2 0 m Bmax 0.94 ± 0.04 (mol/mol) Kd 1.1 ± 0.2 gM 0.0 2 4 6 8 10 [Unbound ATP] (rM) FIG 3-4A 12 14 16 0.4 r. O S0.2 CL c 0.1O 0.0 1 0.0 FIG 3-4B 0.1 0.2 [Bound ATP] (rM) 0.3 0.4 Table 3-5. Binding of ATPfree to PKAcat*hTau40 complex [Unbound ATP] (p.M) 3.6 %Bound ATP 9.2 +/- 0.4% 16.4 21.5 40 +/-2% 60 +/-2% Assays were performed as described in Chapter 3 "Experimental Procedures." [PKAcat]o=4 iM, [hTau40]=200 pM, 1 mM DTT, pH 7.0, 4 "C. The data for each point is the average of ten measurements. vi. Kinetics of inhibition with adenosine and uridine To investigate the specificity of the ATPfri, inhibition effect on PKAcat, kinetics studies were performed with adenosine and uridine as potential inhibitors. Under standard conditions, total Mg 2+and ATP 4- concentrations were both fixed at 2 mM while myelin basic protein concentration was varied from 7 to 21 rpM. Adenosine and uridine were added to the standard assay to a final concentration of 4 mM to assess their effects on PKAat activity. In Figure 3-6, initial velocity (represented as the picomoles of phosphate incorporated over time) is plotted versus MBP for each experimental condition. Adenosine, but not uridine, inhibited the phosphorylation reaction, suggesting that the specificity of the ATPfee inhibition of PKAcat depends on structural determinants in the adenine ring. In experiments (unpublished) by Barbara Blanchard, the inhibition observed following the addition of 4 mM was determined to be partially due to an ionic strength effect. FIGURE 3-5. Effect of uridine and adenosine nucleosides on PKAcat initial velocity at different fixed values of [Myelin Basic Protein]. The assay for protein kinase activity was performed with 0.06 U PKA catalytic subunit plus the indicated concentrations of MBP. Conditions were as described under "Experimental Procedures" except that total Mg2+ concentration was fixed at 2 mM and ATP 4 , Adenosine, and Uridine concentrations were varied to achieve the following final concentrations 2 mM ATP 4 (0, Control), 2 mM ATP4+4 mM Adenosine(*), 6 mM ATP 4- (U) mM, and 2 mM ATP 4 +4 mM Uridine (*). The data for each point is the average of four measurements. Error bars are +/-1 SEM. 400 350 -Control 300 i +4 mM Uridin( .- E O 250 O 200200 o 150 0 .. 100 10 50 - j+4 mM ATP j +4 mM Adeno 00 5 10 15 [tM MBP FIG 3-5 20 25 C. Discussion Previous experiments in this laboratory (Barbara Blanchard, unpublished results) suggested three possible explanations for the reduced in vitro activity of PKAcat when ATP 4 is added in excess of Mg 2+: an ionic strength effect, inhibition by ATPfee, or a loss of activation by free Mg 2+ . Later experiments by Barbara Blanchard, under conditions where ionic strength was increased by the addition of NaCI, indicated that the ionic strength effect only partially accounted for the reduced in vitro activity of PKAcat when ATP 4 is added in excess of Mg 2+. The goals of this research project were to test the two remaining hypotheses, to draw conclusions regarding mechanism, and to relate the findings to a possible in vivo role for PKAat in the hyperphosphorylation of tau observed in AD. Our experiments with varying MgATP and ATPfr, indicated that loss of activation by decreasing free Mg2+ was not an explanation for the reduction in the in vitro activity of PKAcat when ATP 4- is present in excess of Mg2+. In those experiments, free Mg 2+ concentration varied inversely to ATPf e concentration. If free Mg 2+were acting as a non-essential activator, the double reciprocal plots from an experiment where MgATP is also varied would be predicted to have different intercepts; this was not observed. Additional experiments (not shown) with varying concentrations of NaCl demonstrated that ionic strength effects were negligible in the ranges of Mg 2+ and ATP 4 considered. Hence, the conclusion from our experiments with varying MgATP and ATPfree is that ATPfre is a competitive inhibitor versus MgATP. To characterize the catalytic mechanism for PKAcat in the absence of the putative inhibitor, we performed initial velocity experiments under conditions where the concentration of the putative inhibitor ATPfre, was greatly lowered by high concentrations of total Mg 2+ (10 mM). The recombinant human tau construct hTau40, the largest of the six adult tau isoforms, was used a protein substrate. Analysis of the uninhibited reaction data favored an ordered sequential mechanism with MgATP binding first. An ordered sequential mechanism with hTau40 binding first was ruled out because of negative parameter estimates for KhTau40 and aoKMgATP. The random sequential model, however, could not be completely ruled out based on the large standard errors of parameter estimates because the imprecision could have resulted from our inability to achieve higher (> 200 WiM) concentrations of hTau40 due to protein solubility and pH buffering constraints. Both the ordered and random sequential mechanisms for the PKAct-catalyzed phosphorylation of hTau40 are depicted in Schemes 3-7 and 3-8, respectively. Both ordered and random sequential mechanisms have been reported in the literature for PKAat. An ordered sequential mechanism has been suggested for PKAcat based on initial velocity studies using the ATP analogue AMP*PNP and the PKA Inhibitor (PKI) peptide (Whitehouse & Walsh, 1983). AMP*PNP was determined to be a competitive inhibitor versus MgATP and a noncompetitive inhibitor versus the peptide substrate Kemptide. PKI was found to be a competitive inhibitor versus Kemptide and an uncompetitive inhibitor versus MgATP, indicating that binding of PKI requires prior binding of MgATP. Both experiments were performed at total Mg 2+concentration of 20 mM. Binding studies with [3H]acetyl-Kemptide and excess unlabeled AMP*PNP were reported as further evidence that binding of the nucleotide substrate precedes the binding of the peptide substrate. No binding experiments were described for [3H]acetyl-Kemptide in the absence of excess unlabeled AMP*PNP. PKAat *MgATP*hTau4 0 kpmo /14 +hTau40 / aKhTau40 KMgATP PKAcat +MgATP PKAcat * MgATP Scheme 3-7: Ordered Sequential Mechanism for PKAcat-catalyzed Phosphorylation of hTau40 Products PKAct*hTau40 PKAcat +MgATP oKgATp PKAct*MgATP*hTau40 kpON PKAcat*MgATP Scheme 3-8: Random Sequential Mechanism for PKAcat-catalyzed Phosphorylation of hTau40 Products Kong and Cook (1982, 1988) have reported that the catalytic mechanism of PKAcat follows a random sequential pathway, with a preference for MgATP binding first at high free Mg2+ (10 mM). That conclusion was based on experiments at 0.5 mM free Mg 2+ (low) and 10 mM free Mg 2+(high). In those experiments, the Ser-peptide analogue guanethidine was found to be a competitive inhibitor versus Ser-peptide and a noncompetitive inhibitor versus MgATP, indicating that guanethidine was capable of binding to either free PKAcat enzyme or the PKAcat*MgATP complex. Isotope partitioning experiments with both PKACt*[y- 32P]MgATP and PKAct* [3H]Acetyl-Ser-peptide complexes demonstrated that both nucleotide and peptide substrates were capable of binding to free PKAct enzyme at low and high free Mg2 +concentrations. However, the affinity of MgATP for the enzyme is increased at high concentrations of free Mg 2+ . Our conclusion that PKat follows an ordered sequential pathway is compatible with Kong and Cook's observations under high free Mg2+ conditions and with the observations by Whitehouse and Walsh (1983). Our conclusions, however, do not rule out a random mechanism for PKAct at lower free Mg 2+ concentrations. For an ordered sequential pathway, inhibition of PKAcat by ATPfre could involve direct binding of ATPfrs to the free PKAcat enzyme or to one or both of the two transitory complexes (PKAt*MgATP, PKAat*MgATP*hTau40) depicted in Scheme 3-7. For a random sequential pathway, inhibition of PKAcat by ATP,. could involve direct binding of ATP, to the free PKAcat enzyme or to one or more of the three transitory complexes (PKAcat*MgATP, PKAcat*hTau40, PKAcat*MgATP*hTau40) depicted in Scheme 3-8. In our experiments, ATPfr, was a competitive inhibitor versus MgATP. Competitive inhibition suggests mutually exclusive binding of MgATP and ATPfee; with both presumably binding to the same site. That conclusion eliminates from consideration two possible inhibitor complexes: PKAcat*MgATP*ATPse and the PKA,,at*MgATP*hTau40*ATPe. The conclusion that ATPfre is a noncompetitive inhibitor versus hTau40 is also consistent with that observation. If ATPce is a noncompetitive inhibitor versus hTau40, then the only possible inhibitor complexes are PKAcat*ATPf, and PKAcat*hTau40*ATPfee. In the case of the ordered sequential pathway, formation of the PKAcat*hTau40*ATPf", complex would require that ATPe,bind prior to hTau40. The Ki determined for ATPfi, was 9 WiM. The equilibrium binding experiments demonstrated that ATPfe, is capable of binding to both the free PKAat enzyme and the PKA,.t*hTau40 complex under low free Mg2+ conditions, consistent with ATPfree acting as a noncompetitive inhibitor versus hTau40. ATPfo, showed a greater affinity for PKAcat (Kd = 1.1 iM) than for the PKAcat*hTau40 complex (Kd 18 pM). The difference in affinity suggests a conformational change upon binding of the hTau40 protein. Circular dichroism experiments (Reed & Kinzel, 1984; Reed et al. 1985) with blue dextran bound to the C-subunit suggest that Ser-peptide binding occurs in three steps, with the first involving interaction of the Arg-Arg subsite with the enzyme resulting in a closed conformation. This closing of the site is followed by a second change that results in the peptide assuming a specific coil conformation, and this is followed by a third change that results in some movement of the serine hydroxyl group. In the case of an ordered sequential catalytic mechanism, we would postulate that the PKAcat*ATPe, complex is capable of binding hTau40 and that binding results in increase in the off-rate for ATPfee. For a random sequential mechanism, there might also be a decrease in the on-rate for ATPfre resulting from prior binding of hTau40 to the free PKAcat enzyme. The observation that ATPof, and MgATP compete for the same enzyme forms, namely PKAcat and PKAt*hTau40, suggests that ATPee might be mimicking MgATP in reaction pathway. Binding of the uncomplexed nucleotide to the active site of PKAcat would be the most likely explanation. High-resolution 3D structural data on PKAat (Zheng et al., 1993 and Bossemeyer et al., 1993) are consistent with that explanation. Zheng et al. showed that several residues bind the MgATP in the PKAcat*PKI peptide*MgATP ternary complex. The adenine ring is enclosed in a hydrophobic pocket, formed at the domain interface, consisting of residues Leu40, Va157, Ala70, Metl20, Tyr122, Va1123, and Leul73. Two hydrogen bonds serve to anchor the adenine ring in this hydrophobic pocket. The primary hydrogen bond contact is between the N6 amino group and the main-chain carbonyl of Glul21. The second hydrogen bond contact is between the N7 nitrogen and the side-chain hydroxyl of Thr183. Two hydrogen bonds also stabilize the ribose ring. The oxygen of the 2'-OH is located 2.6 A from the side chain of Glu 127. The 3'-OH is 2.6 A from the x-carbonyl oxygen of Glul70. The structural data prompted us to compare the nucleosides adenosine and uridine as inhibitors of PKAC.t. Presumably, adenosine would possess most of the structural determinants for inhibition; uridine would not. Using myelin basic protein as substrate, we observed that adenosine was also an inhibitor of PKAcat; uridine was not. This suggests that discrimination at the PKAcat active site is stringent enough to exclude the pyrimidine nucleoside uridine but not enough to exclude the two structural analogs of MgATP, adenosine and ATPfree. On the basis of magnetic resonance studies, Granot et al. (1979) found substantial changes in the torsional angle of the glycosidic linkage, suggesting the enzyme interacts very strongly with the adenosine portion of the nucleotide. This was further substantiated in experiments by Cook et al. (1982) in experiments with Ser-peptide. They reported that adenosine was a competitive inhibitor versus MgATP and a noncompetitive inhibitor versus Ser-peptide; a Ki of 40 pM was reported. The lower Ki (10 WiM) for ATPfee, determined from our experiments, likely reflects the contribution made by the triphosphate group to binding stabilization at the inhibitory site. The inhibitory site for ATPee is likely to be the MgATP binding site. However, an alternative site is also possible. A single site model with binding of ATPfree away from the MgATP site which prevents the binding of MgATP (but not the binding of hTau40) is consistent with the data. A two site model is also possible, with the binding of hTau40 to the free PKAcat enzyme creating a second (lower affinity) inhibitory site for ATPe. The primary evidence against those alternatives is the 3D structural data indicating that there is only a single binding site for MgATP. Presumably, MgATP would also be capable of binding to the alternative inhibitory site. In summary, our enzyme kinetic and equilibrium binding data suggest that the reduced in vitro activity of PKAcat when ATP 4- is added in excess of Mg2+ is due to an inhibition of PKAcat by ATPfre. The proposed mechanism of inhibition is described in Schemes 3-9 and 3-10. The inhibition occurs along either an ordered sequential catalytic pathway or a random sequential pathway, depending on the concentration of free Mg 2+. For the ordered sequential pathway, the enzyme is inhibited by binding of ATPfree to the free PKAcat enzyme. The resulting PKAcat*ATPfrC inhibitor complex is capable of binding to hTau40. For the random sequential pathway, the enzyme is inhibited by binding of ATPfr,, to the free PKAcat enzyme or PKAc,t*ATPfre*hTau4 0 PKAcat*ATPfree PKAcat*MgATP*hTau40 -- Products K, +hTau40 +ATPfree +hTau40 / aKhTau4O KMgATP +MgATP PKAcat*MgATP Scheme 3-9: Proposed Mechanism for ATPfree Inhibition of Ordered Sequential Pathway for PKAcat PKAcat * ATP free *hTau4 0 PKhTau4O PKi PKAcat*ATPfree kp oKMgATP PKAcat*MgATP*hTau4 0 PKACt*hTau40 +ATPfree KhTau40 OxKhTau40 KMgATP PKAcat +MgATP PKAcat*MgATP Scheme 3-10: Proposed Mechanism for ATPfree Inhibition of Random Sequential Pathway for PKAcat Products to the PKAcat*hTau40 complex. Inhibition by ATPf,,e along both pathways is competitive versus MgATP and noncompetitive versus hTau40. The inhibition constant (Ki) determined for ATPf, is 10 pM. The biological implications of these conclusions and their relationship to Alzheimer's disease will be discussed further in Chapter 6. Chapter 4: Inhibition of ERK2 by ATPf.. A. Experimental procedures i. Materials Active mouse recombinant ERK2 enzyme was purchased from New England Biolabs. Brij 35 was purchased from Sigma Chemical Company. Other materials were obtained as described in the Experimental Procedures section of Chapter 3. ii. Purification of recombinant hTau40 Recombinant human tau protein hTau40 was obtained as described in the Experimental Procedures section of Chapter 3. iii. Phosphorylation assay for ERK2 Initial velocity phosphorylation assays were performed in 100 mM HEPES (pH7.0), 1 mM DTT, and 0.01% Brij 35. The reaction was initiated by the addition of 0.0033 units of ERK2. After incubation at 30 oC for 2 hours, 20 jpl aliquots of the reaction mix were spotted onto 2.4 cm diameter phosphocellulose disks to stop the reaction. The phosphocellulose disks were then washed and counted as described in the Experimental Procedures section of Chapter 3. iv. Equilibrium binding of ATPfr~ to ERK2 Equilibrium dialysis was performed in a polycarbonate apparatus (Micro Filtration Products Inc.) containing two halfspheres of 150 ml volume, separated by a dialysis membrane. Both chambers contained 120 tplof 100 mM HEPES, pH 7.0, 1 mM DTT, and 0.01% Brij 35. One chamber contained 0.3 M ERK2. The other chamber contained the total (bound+unbound) amount of [y- 33 P]ATP (180,000 cpm/pmol) for the experiment. After dialysis for 24 hours at 4 C with gentle shaking, 1 pl. aliquots were taken from each chamber and measured by liquid scintillation counting (Beckman LS 2500) with 5 ml of"Liquiscint" for 1 min. v. Calculation of ATPfree concentrations The experimental concentrations of MgCl 2 and Na2ATP used to obtain the indicated concentrations of MgATP and ATPre were calculated as described in the Experimental Procedures section of Chapter 3. vi. Determination of sequential kinetics parameters for random and ordered sequential bireactant reactions Initial velocity data were analyzed as described in the Experimental Procedures section of Chapter 3. vii. Determination of the effect of ATPfre, on the slopes and intercepts of Lineweaver-Burk plots of ERK2 initial velocities The effect of ATPfee on the slopes and intercepts of Lineweaver-Burk plots of ERK2 initial velocities were determined as described in the as described in the Experimental Procedures section of Chapter 3. Determination of the effect of free Mg 2+ on the slopes and intercepts of viii. Lineweaver-Burk plots of ERK2 initial velocities The effect of free Mg2+ on the slopes and intercepts of Lineweaver-Burk plots of ERK2 initial velocities were determined as described in the as described in the Experimental Procedures section of Chapter 3. 100 B. Results i. Two-substrate kinetics with MgATP and hTau40 Initial velocity studies, in the absence of ATPfe, were performed over a wide range of MgATP and hTau40 concentrations (50 to 2000 iM and 5 and 100 pM, respectively), in order to assess the kinetic mechanism and the kinetic constants of the uninhibited phosphorylation reaction. In Figure 4-1A, initial velocity (represented as the picomoles of phosphate incorporated over time) is plotted versus MgATP at constant hTau40. In Figure 4-1B, reciprocal initial velocity is plotted versus the reciprocal of MgATP at constant hTau40. The data were fitted to the equations for random (Equation 3-1) and ordered (Equation 3-2) sequential. The results are shown in Table 4-1. The ordered mechanism, with MgATP binding first, provided the best fit of the data. The kinetic parameters obtained for an ordered mechanism with hTau40 binding first and a random mechanism were unacceptable due to high imprecision and negative estimates, respectively. ii. Kinetics of inhibition with varying ATPr.. and MgATP Inhibition kinetics studies were performed at a fixed concentration of hTau40 (20 pM) with varying concentrations of MgATP and ATPr,. In these experiments free Mg 2+ concentrations varied in the range of 0.04 mM to 40 mM. In Figure 4-2A, initial velocity (represented as the picomoles of phosphate incorporated over time) is plotted versus MgATP at constant ATPfe. In Figure 4-2B, reciprocal initial velocity is plotted versus the reciprocal of MgATP at constant ATPee,. The effects of inhibitor on slope and intercept parameters of the Lineweaver-Burk plots are shown in Table 3-4. ATPfree affected only the slopes of the Lineweaver-Burk plots, indicating that it is a competitive inhibitor versus MgATP. 101 FIGURE 4-1. Dependence of ERK2 initial velocity on [MgATP] at different fixed values of [hTau40]. The assay for protein kinase activity was performed with 0.02 U ERK2 plus the indicated concentrations of MgATP and hTau40. Conditions were as described under "Experimental Procedures" except that total Mg 2 + concentration was fixed at 10 mM while ATP 4" concentration was varied at fixed hTau40 concentrations of 5 (a), 20 (0), 50 (*), and 100 (V) pM. The data for each point is the average of two measurements. Error bars are +/1 SD. (A) Michaelis plots of the dependence of ERK2 activity on MgATP and hTau40. (B) Lineweaver-Burk plots of the activity data presented in (A). 102 2.5- gM hTau40 v 100 2.0- Ef --- 1.5 - 50 O0 E 1.0- 0 0 13_ 20 5 0.5- 0.0 -I 500 1000 1000 pM FIG 4-1A 00 1500 MgATP 2000 2500 14 pM hTau40 5 12 10 0 CL 0 0E 20 O rE 50 100 0 -0.000 0.005 0.010 ([M MgATP) FIG 4-1B 0.015 0.020 Table 4-1. Estimated kinetic parameters for ERK2 Catalytic Mechanism Ordered MgATP binds first Random Vmax (pmoles/min) KMgATP (M) KhTau4 0 (pM) aKMgATP (pM) aKhTau40 (PM) RMSE 7 (3) 4.2 (0.9) 76 (95) 20 (28) a 799 (546) 499 (146) Ordered hTau40 binds first 2.2 (0.2) -152 (28) -418 (140) 205 (111) 84 (33) 0.1 0.01 0.2 Parameter estimates were determined as described in Chapter 4 "Experimental Procedures." ERK2 concentration was 0.0033U. Total Mg 2+concentration was 10 mM. MgATP and hTau40 concentration ranges were 5-20000 pM and 5-100 pM, respectively. Standard errors are in parentheses. 'Not significantly different from zero by Student t test at ca=5%. FIGURE 4-2. Dependence of ERK2 initial velocity on [MgATP] at different fixed Values of [ATPfree]. The assay for protein kinase activity was performed with 0.02 U ERK2 catalytic subunit plus the indicated concentrations of MgATP. Conditions were as described under "Experimental Procedures" except that hTau40 concentration was fixed at 20 pM at ATPre concentrations of 5 (0), 100 (0), and 150 (*) pM. The data for each point is the average of two measurements. Error bars are +/- 1 SD. (A) Michaelis Plots of the ERK2 inhibition by ATPfe,. (B) Lineweaver-Burk Plots of the inhibition data presented in (A). 106 2.5 r- O 5 M ATPfree 2.0 1.5 100 gM ATPf a) E 1.0 0.5 0.5 150 gM ATPfree 0.0 1 0 500 1000 [M MgATP FIG 4-2A 1500 2000 pM ATPfree 12 150 10 8 100 O a) E 6 0 4- E 5 2 00.0000 FIG 4-2B 0.0025 0.0050 (pM MgATP) - 0.0075 0.0100 The observed pattern was not consistent with free Mg 2+acting as a non-essential activator (see Appendices 8C and 8D for rate equations for non-essential activation of random and ordered sequential bireactant reactions, respectively). iii. Kinetics of inhibition with varying ATPfr,. and hTau40 Inhibition kinetics studies were performed at a fixed concentration of MgATP (500 pM) with varying concentrations of MgATP and ATPffr. In Figure 4-3A, initial velocity (represented as the picomoles of phosphate incorporated over time) is plotted versus hTau40 at constant ATPree. In Figure 4-3B, reciprocal initial velocity is plotted versus the reciprocal of hTau40 at constant ATPfee. The effects of inhibitor on slope and intercept parameters of the Lineweaver-Burk plots are shown in Table 3-4. ATP., affected both the slopes and intercepts of the Lineweaver-Burk plots. For an ordered sequential mechanism with MgATP binding first, that L-B pattern suggests that ATPfee is noncompetitive versus hTau40. Analysis of the replot of L-B Slope*Intercept versus ATPf~e/MgATP (Figure 4-3C) yielded a Ki for ATPe, of 32 + 6 pM. iv. Equilibrium binding of ATPf,,. to ERK2 The inhibition experiments indicated that ATPf,ee was competitive versus MgATP and noncompetitive versus hTau40, with two inhibitor complexes being formed: ERK2*ATPfre and ERK2*hTau40*ATPfc. Furthermore, formation of the ERK2*hTau40*ATPee complex is ordered with the binding of ATPe preceding the binding of hTau40. To confirm the binding between ERK2 and ATPe, an equilibrium binding experiment was performed with 0.3 mM ERK2. Table 4-2 shows that ATPfrs, is able to bind ERK2, with 20% of enzyme bound at a ATPfe, concentration of 1.9 mM. 109 FIGURE 4-3. Dependence of ERK2 Initial Velocity on [hTau40] at different fixed values of [ATPfr.]. The assay for protein kinase activity was performed with 0.02 U ERK2 catalytic subunit plus the indicated concentrations of hTau40. Conditions were as described under "Experimental Procedures" except that MgATP concentration was fixed at 500 M at ATPffr concentrations of 15 (0), 50 (0), and 100 (*) tM. The data for each point in Figures 4-3A&B is the average of two measurements. Error bars are +/- 1 SD. (A) Michaelis Plots of the ERK2 inhibition by ATPfre. (B) Lineweaver-Burk Plots of the inhibition data presented in (A). (C) Replot of Lineweaver-Burk Slope*Intercept versus [ATPfree]/[MgATPj. The line was fitted by linear regression and the correlation coefficient was 0.96. 110 3.0M ATPfree 3.0 - I 15 2.5 c 2.0- E O .. 1.5 S 1.0 1.0- o 50 100 - C.. 0.5 0.00 50 100 150 M hTau40 FIG 4-3A 200 250 M ATPfree 3.0 I 100 2.5 Ot 0 o) 2.0 . 50 1.5 E o0 0 -- 15 1.0 E 0.5 0.0 0.00 FIG 4-3B 0.02 0.04 0.06 ( M hTau40) 0.08- 0.10 0.12 18 16 1412 CL (D 0o 10 O 8 0 6 4 2 00.00 111~ I- 0.05 0.10 0. 15 [ATPfree]/[MgATP] 4-JUC 0.20 Table 4-2. Binding of ATPfree to ERK2 [Unbound ATP] (iM) %Bound ATP 10+/- 0.3% 0.37 20 +/- 0.7% 1.92 Assays were performed as described under "Experimental Procedures." [ERK2]o=0.3 WM, 1 mM DTT, pH 7.0, 4 oC. The data for each point is the average of five to ten measurements. C. Discussion Roder and Ingram (1991) reported that the in vitro activity of the brain protein kinase PK40O 2, which is capable of hyperphosphorylating the cytoskeletal protein Tau, is strongly inhibited by the presence of mM concentrations of ATP 4- added in excess of Mg 2+ . Three possible explanations for the reduced activity of ERK2 are ionic strength effects, inhibition by ATPfroe, or a loss of activation by free Mg 2+ . The goals of the ERK2 research project were to examine the plausibility of those three explanations, to draw conclusions regarding mechanism, and to relate the findings to a possible in vivo role for ERK2 in the hyperphosphorylation of tau observed in AD. Loss of activation by decreasing free Mg 2+was not the cause of the reduction in the in vitro activity of PKACt when ATP4 is present in excess of Mg2+. If free Mg2+ were acting as a non-essential activator of ERK2, the double reciprocal plots from an experiment where MgATP and free Mg 2+ are varied would be predicted to have different slopes; this was not observed. Experiments with varying concentrations of NaCl also ruled out ionic strength effects as an explanation. Our experiments on the catalytic mechanism of ERK2 with MgATP and hTau40 substrates suggest that the reaction is ordered sequential, with MgATP binding first, at 10 mM total Mg 2+ . The experiments, however, don't prove such a mechanism. Additional experiments, with non-hydrolyzable analogues of MgATP and inhibitory peptide substrates would further strengthen the argument for an ordered sequential mechanism. Furthermore, it is not known what effect Mg2+ concentration has on the catalytic mechanism. The ordered sequential pathway, with MgATP binding first, is illustrated in 115 Scheme 4-1. The pathway involves two transitory complexes, ERK2*MgATP and ERK2*MgATP*hTau40. Inhibition of ERK2 by ATPe, would involve direct binding of ATPee, to the free ERK2 enzyme or to one or both of the two transitory complexes. In our experiments, ATPfe is a competitive inhibitor versus MgATP. Competitive inhibition suggests mutually exclusive binding of MgATP and ATPfre; with both presumably binding to the same site. That observation eliminates from consideration two possible inhibitor complexes: ERK2*MgATP*ATPf, and ERK2*MgATP*hTau40*ATPf~.. Our data indicating that ATPe, is a noncompetitive inhibitor versus hTau40 are also consistent with that observation. If ATPf, is a noncompetitive inhibitor versus hTau40, then the only possible inhibitor complexes are ERK2*ATPfee and ERK2*hTau40*ATPife. In the case of the latter inhibitory complex, hTau40 binds after binding of ATPfr. A Ki of 32 pLM for ATPfr, was determined. Equilibrium binding experiments confirmed that ATPo binds to ERK2, with a Kd greater than 2 pM. The available structural data on ERK2 indicates that, like with other protein kinases, the ATP-binding site is at an interior position between two domains; whereas the protein binding site is on the outside. Zhang et al. (1994) showed that MgATP or the nonhydrolyzable ATP analogue AMP-PNP are loosely bound deep in the back of the domain interface between the C-terminal and N-terminal domains. The N6 amino group of the adenine ring is hydrogen bonded to the backbone carbonyl of Asp 104. The ribose 02' and 03' hydroxyls form a hydrogen-bonding network to Asp109(Glu127) and Lysll12. Our experiments suggest that the specificity of ERK2 nucleotide binding extends to ATP that is uncomplexed to Mg 2+ and that such binding inhibits the phosphorylation reaction. It would be 116 ERK2*MgATP*hTau40 kp -O / +hTau40 ERK2 R A 1TPD oCKhTau40 ERK2*MgATP +lyl.• Scheme 4-1: Ordered Sequential Mechanism for ERK2-catalyzed Phosphorylation of hTau40 Products interesting to determine whether the specificity also extends to ADP that is uncomplexed to Mg 2+. If so, then ADPfr would also be predicted to behave as an inhibitor of the phosphorylation reaction. 4 In summary, we have determined that the reduction in activity of ERK2 by ATP in excess of Mg 2+is due to inhibition by ATPsf. The proposed mechanism of inhibition is described in Scheme 4-2. Our enzyme kinetic and equilibrium binding data, suggest a mechanism of inhibition which is competitive versus MgATP and noncompetitive versus hTau40. Furthermore, the ordered sequential reaction mechanism of ERK2 implies that increasing concentrations of hTau40 serve to drive the equilibrium towards the form of the enzyme bound to ATPe. The biological implications of these results and the possible relationship to Alzheimer's disease will be discussed further in Chapter 6. 118 ERK2*ATPfree*hTau40 fKhTau40 ERK2*ATPree ERK2*MgATP*hTau40 - PON Products / +ATPfree +hTau40 / aKhTau4O KMgATP ERK2 +MgATP ERK2*MgATP Scheme 4-2: Proposed Mechanism for ATPfree Inhibition of Ordered Sequential Pathway for ERK2 Chapter 5: Inhibition of GST-ERK2 by ATPfree 120 A. Experimental procedures i. Materials Active recombinant p44mapk enzyme was purchased as a glutathione-S-transferase fusion protein from Upstate Biotechnology Inc.. Other materials were obtained as described in the Experimental Procedures section of Chapter 3. ii. Purification of recombinant hTau40 Recombinant human tau protein hTau40 was obtained as described in the Experimental Procedures section of Chapter 3. iii. Phosphorylation assay for GST-ERK2 Initial velocity phosphorylation assays were performed in 100 mM HEPES (pH7.0), and 1 mM DTT. The reaction was initiated by the addition of 0.00034 unit of GST-ERK2. After incubation at 30 C for 2 hours, 20 tl aliquots of the reaction mix were spotted onto 2.4 cm diameter phosphocellulose disks to stop the reaction. The phosphocellulose disks were then washed and counted as described in the Experimental Procedures section of Chapter 3. iv. Calculation of ATPfre, concentrations The experimental concentrations of MgC12 and Na2ATP used to obtain the indicated concentrations of MgATP and ATPe, were calculated as described in the Experimental Procedures section of Chapter 3. 121 Determination of the effect of ATPfree on the slopes and intercepts of v. Lineweaver-Burk plots of GST-ERK2 initial velocities The effect of ATPfe, on the slopes and intercepts of Lineweaver-Burk plots of ERK2 initial velocities were determined as described in the as described in the Experimental Procedures section of Chapter 3. Determination of the effect of free Mg2+ on the slopes and intercepts of vi. Lineweaver-Burk plots of GST-ERK2 initial velocities The effect of free Mg 2+ on the slopes and intercepts of Lineweaver-Burk plots of ERK2 initial velocities were determined as described in the as described in the Experimental Procedures section of Chapter 3. B. Results Studies were also performed with ERK2 as a glutathione-S-transferase fusion protein. Based on the results from our studies with non-fusion ERK2 (and PKAcat), inhibition by ATPfr, involves mutually exclusive binding of MgATP and ATPfree. Presumably, MgATP and ATPfee bind at the active site cleft. If ATPee were instead binding to an allosteric regulatory site, that interaction might be altered in the GST-ERK2 fusion protein. These studies aimed to determine what differences existed between ERK2 and GST-ERK2 in their sensitivity to ATPfre inhibition. i. Kinetics of inhibition with varying ATPfee and MgATP Inhibition kinetics studies were performed at a fixed concentration of hTau40 (50 jiM) with varying concentrations of MgATP and ATPfr . In these experiments, the range of free Mg 2 + concentration was 0.04 - 40 mM. In Figure 5-1A, initial velocity (represented as the picomoles of phosphate incorporated over time) is plotted versus MgATP at constant ATPfre. 122 FIGURE 5-1. Dependence of GST-ERK2 initial velocity on [MgATP] at different fixed values of [ATPfr,]. The assay for protein kinase activity was performed with 0.02 U GSTERK2 catalytic subunit plus the indicated concentrations of MgATP. Conditions are as described under "Experimental Procedures" except that hTau40 concentration was fixed at 50 pM at ATPfr, concentrations of 5 (0), 100 (0), and 150 (*) pM. The data for each point is the average of two measurements. Error bars are +/- 1 SD. (A) Michaelis Plots of the GST-ERK2 inhibition by ATPfree. (B) Lineweaver-Burk Plots of the inhibition data presented in (A). 123 pM ATPfree 0.8- 100 150 5 0.6C E O 0.4 - 0 E 00 0.2- 0.0 500 1000 1500 tM MgATP FIG 5-1A I I 2000 2500 pM ATPfree 70 150 60 50 O 40 0 100 E 0 30 E - 20 5 101 00.000 0.005 0.010 0.015 (riM MgATP) FIG 5-1B 0.020 0.025 0.025 In Figure 5-1 B, reciprocal initial velocity is plotted versus the reciprocal of MgATP at constant ATP,.. The effect of inhibitor on slope and intercept parameters of the LineweaverBurk plots is shown in Table 3-4. ATPfee affected only the slopes of the Lineweaver-Burk plots, indicating that it is a competitive inhibitor versus MgATP. The observed pattern was not consistent with non-essential activation by free Mg2+. ii. Kinetics of inhibition with varying ATPfre, and hTau40 Inhibition kinetics studies were performed at a fixed concentration of MgATP (500 VIM) with varying concentrations of MgATP and ATPfe. In Figure 5-2A, initial velocity (represented as the picomoles of phosphate incorporated over time) is plotted versus hTau40 at constant ATPfr,. In Figure 5-2B, reciprocal initial velocity is plotted versus the reciprocal of hTau40 at constant ATPre. The effect of inhibitor on slope and intercept parameters of the Lineweaver-Burk plots is shown in Table 3-4. ATPfre affected both the slopes and intercepts of the Lineweaver-Burk plots, suggesting that ATPfr, is noncompetitive versus hTau40. Analysis of the replot of L-B Slope*Intercept versus ATPfr /MgATP (Figure 5-2C) yielded a Ki for ATP&, of 15 ± 2 p.M. C. Discussion Our limited studies with recombinant GST-ERK2 indicate that ATPfe, acts as an reversible inhibitor of the phosphorylation reaction. The inhibition of GST-ERK2 by ATPfec is competitive versus MgATP and noncompetitive versus hTau40. Mechanistically, the inhibition of GST-ERK2 by ATPf e is similar to that observed with ERK2 (see Scheme 5-1). There is, however, evidence suggesting that the affinity of ATP~o, for the GST-ERK2 enzyme (Ki = 15 pM) is greater than its affinity for ERK2 (Ki = 32 p.M). We are unable to draw any 126 FIGURE 5-2. Dependence of GST-ERK2 initial velocity on [hTau40] at different fixed values of [ATPr,]. The assay for protein kinase activity was performed with 0.02 U GSTERK2 catalytic subunit plus the indicated concentrations of hTau40. Conditions are as described under "Experimental Procedures" except that MgATP concentration was fixed at 50 LM at ATPr, concentrations of 15 (E), 50 (0), and 100 (*) rtM. The data for each point is the average of two measurements. Error bars are +/- 1 SD. (A) Michaelis Plots of the GST-ERK2 inhibition by ATPfee. (B) Lineweaver-Burk Plots of the inhibition data presented in (A). (C) Replot of Lineweaver-Burk Slope*Intercept versus [ATPfree]/[MgATP]. The line was fitted by linear regression and the correlation coefficient was 0.99. 127 M ATPfree 1.4 15 1.2 1.0 50 0.8 0 0.6 0 S 0.4 100 0.2 0.00 50 100 150 ,M hTau40 FIG 5-2A 200 250 pM ATPfree 4.5 100 4.0 50 3.5 3.0 0 o E o0 15 2.5 2.0 1.5- E 1.00.5n n v.v -I I 0.00 FIG 5-2B a 0.01 I 0.02 Ii I 0.03 0.04 (pM hTau40) - I I I 0.05 0.06 definitive conclusions from that data about differences between GST-ERK2 and ERK2 with respect to their affinities for the two substrates MgATP and hTau40. Any such estimates obtained from the existing GST-ERK2 data would be apparent values, dependent on the concentrations of ATPre and of the fixed substrate. That information could be obtained from future studies on kinetics of the uninhibited phosphorylation reaction. It would be especially important to determine if the increased affinity of GST-ERK2, relative to ERK2, for ATPe, also reflects a similar increase in affinity for MgATP. 132 80- 70 CL 60 C) C 40 30 20 0.00 0.05 0.10 0.15 [ATPfree]/[MgATP] FIG. 5-2C 0.20 GST-ERK2*ATPfA*hTau40 3 KhTau40 GST-ERK2*ATPfree GST-ERK2*MgATP*hTau40 -10 Products +ATPfree +hTau40 / aKhTau40 KMgATP GST-ERK2 +MgATP - GST-ERK2*MgATP Scheme 5-1: Proposed Mechanism for ATPfree Inhibition of Ordered Sequential Pathway for GST-ERK2 Chapter 6: Summary 133 A. Summary of conclusions This research work has concluded that the reported reduction in the in vitro activities of the two tau kinases, PKAat (Barbara Blanchard, unpublished results) and ERK2 (Roder & Ingram, 1983), by ATP 4- that is present in excess of Mg 2+ is due primarily to inhibition by ATPfre. Studies done on GST-ERK2 also led to the same conclusion. For all three kinases, the inhibition by ATPee is competitive versus MgATP and noncompetitive versus the recombinant tau protein hTau40. The results indicate that binding of ATPfe and MgATP to all three enzymes is mutually exclusive. We conclude that ATP,, inhibits the three tau kinases by acting as a non-reactive substrate analogue. The inhibition constants (Ki's) determined for ATPfee were 9, 32, and 15 CtM for PKAat, ERK2, and GST- ERK2, respectively. We also concluded that the catalytic mechanism for both PKAcat- and ERK2 catalyzed phosphorylations of hTau40 is ordered sequential at 10 mM free Mg 2+ . The relevance of those conclusions with respect to possible in vivo roles for PKAcat and ERK2 in the pathology of AD will be discussed below. ATPr has been described as an inhibitor of the glucose metabolic enzyme phosphofructokinase via binding to an allosteric regulatory site (Lowry & Passoneau, 1966; Buckwitz et al., 1990). Those studies concluded that inhibition by ATP&ee is via binding to a single site which is different from the active site. Our conclusions regarding the three tau kinases suggest, but do not prove, that the inhibitory site for ATPfree is the MgATP binding site. The strongest evidence against a separate regulatory site comes from 3D structural data indicating the presence of a single MgATP-binding site for both PKAcat and ERK2. However, 134 the crystal structure of a protein might be quite different from its solution structure. A twosite model is also possible, but would not be very consistent with the structural data. Intracellular total ATP 4- concentrations have been determined by 3 1 P-NMR measurements and are in the range of 3 to 5 mM, depending on cell type (Cerdan & Seelig, 1990). The concentration of total Mg 2+ is in the range of 5 to 30 mM, also depending of the 4 cell type (Romani & Scarpa, 1992). Approximately 98-99% of intracellular ATP - is in the form of MgATP (Li et al., 1993), suggesting that intracellular ATPfre concentrations are in the range of 30 to 100 pM. Cytosolic free Mg 2+ concentration is in the range of 200 pM to 1 mM (Li et al., 1993). Direct measurements of antemortem ATP levels in AD brain has 31 proven to be difficult due to limitations in the use the of P-NMR methodology on tissue. Studies of glucose metabolism, indirectly measuring ATP production, in both incipient and advanced late-onset AD patients by Hoyer suggested that ATP production was reduced, in comparison to health age-matched controls, by approximately 46% in incipient patients and 58% in advanced patients Our studies indicate that the affinity of PKAcat for ATPfree is greater than the affinity of ERK2 for ATPfree. The difference between the affinities of the two enzymes for MgATP is much larger though; PKAcat and ERK2 have Km's for MgATP of 35 and 499 mM, respectively. The order of magnitude difference in the affinities of ERK2 and PKAcat for MgATP indicates a significant difference in their sensitivity to competitive inhibition by ATPree. It appears that ERK2 is less efficient at discriminating between the catalyticallyproductive (MgATP) and -unproductive (ATP&,e) forms of ATP; ERK2 affinity for ATPe, is an order of magnitude stronger than its affinity for MgATP. 135 The observed difference between the two tau kinases in their sensitivity to ATPfree inhibition might be physiologically relevant vis-A-vis the decline in ATP production that is observed in AD. Figures 6-1A&B show that there would be a significant increase in the activity of ERK2, but not of PKAcat, when total ATP 4- is assumed to drop from 3 mM to 1 mM (a 66% reduction) while ATPf~e drops from 100 W.M to 2 pM. This is, of course, a hypothetical comparison in the absence of direct measurements of intracellular ATP 4 and ATPee in AD brain. Nevertheless, our results provide a biochemical basis for up-regulation of tau kinases due to declining ATPee concentrations. Furthermore, we have demonstrated how a lifting of inhibition by ATPe, can have quantitative different effects on the in vitro activities of two tau kinases (PKAcat and ERK2). In light of the fact that in vitro studies demonstrating that ERK2 is also capable of phosphorylating tau protein at some of the sites which are unique to PHF-tau (Roder & Ingram, 1993; Drewes et al., 1992), our conclusions provide further support for future studies on the in vivo regulation of ERK2 activity in AD. B. Summary of contributions There is currently no effective treatment for AD. A major impediment to the development of more effective treatments is the striking heterogeneity in the etiology of the disease. Such heterogeneity might require that many different therapeutic strategies be developed concurrently. Fortuitously, the evolving evidence also suggest that different initiating events may converge on a final common pathway producing the two characteristic lesions in AD, amyloid-3 plaques and neurofibrillary tangles. There is support for the notion that it is these "final" lesions which lead to the dysfunction and death of CNS neurons and ultimately to the dementia of AD. This convergence of different initiating events on the 136 FIGURE 6-1. Dependence of PKAcat and ERK2 activities on [hTau40] at hypothetical Alzheimer's disease concentrations of [MgATP] and [ATPfree,]. (A) PKAcat activity in normal versus hypothetical Alzheimer's disease. (B) ERK2 activity in normal versus hypothetical Alzheimer's disease. Reported normal concentrations of [MgATP] and [ATPf&] are 3 mM and 200 C.M, respectively. Hypothetical concentrations of [MgATP] and [ATP&,] are 1 mM and 2 p.M, respectively. Activity dependence is as predicted for noncompetitive inhibition of PKAcat and ERK2 by ATPre. 137 100 90 Hypothetical AD: 1000 M MgATP, 2 gM ATPfree 8070 E 60 O Normal: 3000 gM MgATP, 100 giM ATPfree 50 440 D 40 1- 30 20 10 0 I 0 I 200 400 [hTau40] (PM) FIG. 6-1A 600 800 100 Hypothetical AD: 1000 pM MgATP, 2 M ATPfree 90 - 8070- E 604- 0 50 D 40- a- 30 Normal: 3000 pM MgATP, 100 gM ATPfree , ' 20 10 0 0 200 400 [hTau40] ([rM) FIG. 6-1B I I I 600 800 development of plaque and tangle lesions in the AD brain is highly beneficial for the development of a treatment for AD. The "final" lesions could serve as therapeutic targets common to many etiologically distinct forms of AD. The tangle hypothesis for AD postulates that abnormal up-regulation of tau phosphorylation leads to the formation of PHF-tau in neurons. Hyperphosphorylation of tau protein has been shown to interfere with the normal function of tau to form and stabilize the microtubular cytoskeleton. Studies on the regulation of the phosphorylation state of tau have suggested that either increased activities of protein kinases and/or reduced activities of protein phosphatases may be involved. As described in Chapter 2, several candidate kinases and phosphatases have been identified based on a combination of in-vitro criteria. This work establishes a an in vitro basis for a possible link between the activity of tau kinases and physiologic ATP levels in AD. This research helps to further narrow the list of tau kinases that might be involved in the transformation of tau to PHF-tau. The studies provide justification for pursuing a therapeutic strategy of selective inhibition of ERK2 as a means of preventing an up-regulation of activity that possibly accompanies the decline in physiologic ATP levels observed in AD and old age. C. Future research Future research should follow two lines: biochemical and cell biological. The biochemical studies that most directly follow from this work are those that would further define the nature of the interactions between PKAcat or ERK2 and ATPfee. Those studies include completing the experiments to directly measure the dissociation constants for ATPfree binding to the PKAcat*hTau40 complex and to the free ERK2 enzyme. Additionally, 140 inhibition experiments with MgADP, nonhydrolyzable ATP analogues such as adenosine 5'(0,y-imidotriphosphate) (MgAMPPNP), and inhibitor peptides should be performed to further characterize the ordered sequential mechanism of ERK2. Additional experiments to characterize the uninhibited phosphorylation reaction for GST-ERK2 would also make possible further comparisons with ERK2. The mechanism of inhibition of PKAat and ERK2 by ATPfr,, implies that other tau kinases which do not discriminate between ATPf, and MgATP would be predicted to be inhibited by ATPr,. Other tau kinases for study include GSK-3, PKC, and CaM kinase. Another line of biochemical study would involve characterizing the kinetics of ERK2 and PKAat using pre-phosphorylated tau substrate. In this research project, unphosphorylated recombinant hTau40 protein was used as co-substrate. The adult form of tau found in neurons, however, is phosphorylated at two or three sites. It is likely that the kinetic parameters for the phosphorylated protein are different from that for the unphosphorylated protein. Cell biological studies should include the development of a human neuronal cell line for the ATP-depletion model of AD. Prior work in this laboratory on the effects of ATP depletion on tau phosphorylation relied on rat pheochromacytoma (PC 12) cells. Alternative cell line to be considered are the hnT, a differentiated human teratocarcinoma cell line and the SY5Y, a human neuroblastoma cell line. It would be more relevant to characterize, using the existing antibodies to phosphorylated tau protein, the effect of ATP uncouplers on tau phosphorylation in either the hnT or SY5Y cells. The effect ofERK2-specific inhibitors, such as hypericin, on tau phosphorylation should also be studied. Chapter 7: References 142 Ahn N. G. et al. (1991) J Biol. Chem. 266, 4220-4227. Ames, B. N., Shigenaga, M. K., & Hagen, T. M. (1993) Proc. Natl. Acad. Sci. USA 90, 79157922. Anderson N. G. et al. (1990) Nature 353, 670-674. Arispe, N., Rojas, E., & Pollard, H. (1993) Proc. Natl.Acad. Sci. USA 90, 567-571. Arriagada, P. V. et al, (1992) Neurology 42, 631-639. Bossemeyer D. et al. (1993) EMBO J. 12, 849-859. Braak, H., & Braak, E. (1991) Acta Neuropathol.82, 239-259. Buckwitz, D., Jacobasch, G., & Gerth, C. (1990) Biochem J 267, 353-3 57. Bush, M. L., Miyasharo, J. S., & Ingram, V. M. (1995) Proc.Natl. Acad. Sci. USA 92, 18611865. Butler M., & Shelanski, M. L. (1986) J. Neurochem. 47, 1517-1522. Butner, K. A., & Kirschner, M.W. (1991) J. CellBiol. 115, 717-730. Cerdan S., & Seelig, J. (1990) Annu. Rev. Biophys. Biophys. Chem. 19, 43-67. Cobb, M. H., Boulton, T. G., & Robbins, D. J. (1991) CellRegul. 2, 965-978. Cook P. F. et al. (1982) Biochemistry 21, 5794-5799. Cox, S., Radzio-Andzelm, E., & Taylor, S. S. (1994) Curr. Opin. Struct. Biol. 4, 893-901. Dent, P. et al. (1992) Science 257, 1404. Dickson D. W. et al. (1991) Neurobiol.Aging 13, 179-189. Divry, P., & Florkin, M. (1927) C. R. Soc. Biol. 97, 1808-1810. Dreschel D. N. et al. (1992) Mol. Biol. Cell 3, 1141-1154. Drewes G. et al. (1992) EMBO J. 11, 2131-2138. Eanes, E. D., & Glenner, G. G. (1968) J. Histochem. Cytochem. 16, 673-677. Flament, S. & Dalacourte, A. (1989) FEBS Lett. 247, 213-216. Gabzuda D. et al. (1994) J. Biol. Chem. 269 (18), 13623-13628. Glenner, G. G. (1980a) N. Engl. J. Med. 302, 1283-1292. Glenner, G. G. (1980b) N. Engl. J. Med. 302, 1333-1343. Glenner G. G., & Page, D. L. (1976) Int. Rev. Exp. Pathol. 15, 1-92. Glenner, G. G., & Wong, C. W. (1984) Biochem. Biophys. Res. Commun. 120, 855-890. Goedert, M. et al. (1988) Proc.Natl. Acad. Sci. USA 85,4051-4055. Goedert, M. et al. (1993) Proc.Natl. Acad Sci. USA 90, 5066-5070. Granot, J. et al. (1979) Biochemistry 18, 2339-2345. Grundke-Iqbal, I. et al. (1986) Proc. Natl. Acad. Sci. USA 83, 4913-4917. Hanks, S. K., Quinn, A. M., & Hunter, T. (1988) Science 241, 42-52. Hannun, Y. A., & Bell, R. M. (1990) J. Biol. Chem. 265, 2962-2972. Hardy, J., & Allsop, D. (1991) Trends Pharmacol.Sci. 12, 383-388. Hardy, J. (1992) Nature Genet. 1, 233. Hardy, J. (1997) TINS 20, 154-159. Hasegewa et al. (1992) J. Biol. Chem. 267, 17047-17054. Hjelmquist G. et al. (1974) Biochem. Biophys. Res. Commun. 61, 559. Hoyer, S. (1992) Mol. Chem. Neuropathol. 16, 207-224. Hoyer, S. (1993) Ann. NYAcad. Sci. 695, 77-80. Hoyer, S. (1994) Ann. NYAcad. Sci. 719, 248-256. Hsiao, K et al. (1996) Science 274, 99-102. Ihara, Y., Abraham, C., Selkoe, D.J. (1983) Nature 304, 727-730. 143 Ihara Y. et al., (1986) J.Biochem. (Tokyo) 99, 1807-1810. Iqbal K. et al. (1986) Lancet 2, 421-426. Jarrett J. T. et al. (1993) Biochemistry 32, 4693-4697. Kanai Y. et al. (1989) J Cell Biol. 109, 1173-1184. Kang, J. et al. (1987) Nature 325, 733. Kish, S. et al. (1992) J.Neurochem. 59, 776-779. Knighton D. R. et al. (1991) Science 253, 407-414. Kondo J. et al. (1988) Neuron 1, 827-834. Kong C-T., & Cook, P.F. (1988) Biochemistry 27, 4795-4799. Kraeplin, E. (1910) Psychiatrie: Ein Lehrbuch fir Studierende und irtze, Verlag von Johann Ambrosius Barth, Leipzig, Germany. Ksiezak-Reding, H., Liu, W-K, & Yen, S-H (1992) Brain Res. 597, 209-219. Lee G. (1990) Cell Motil. Cytoskel. 15, 199-203. Lee, V. M-Y. et al. (1991) Science 251, 675-678. Levy-Lahad, E. et al. (1995) Science 269, 973-977. Li, H-Y, Dai, L-J, & Quamme, G. A. (1993) J Lab Clin. Med. 122 (3), 260-272. Linnane A. W. (1989) Lancet 1, 642-645. Lowry O.H., & Passoneau, J. V. (1966) J. Biol. Chem. 241, 2268-2279. Mandelkow E.-M. et al. (1992) FEBS Lett. 314, 315-321. Mann D. M. A. et al. (1996) Ann.Neurol. 40, 149-156. Martell, A. & Smith, R. (1975) Critical Stability Constants, Plenum Publishing Corp. New York. Martell, A. & Motekaitis, R. (1992) Determination and Use of Critical Stability Constants, Wiley-VCH Publishers, New York Masters, C. L., et al. (1985) Proc.Natl. Acad. Sci. USA 82, 4245-4247. Mattson, M. P. et. al. (1992) J.Neuroscience 12, 376-389. Mecocci, P. et al. (1993) Ann Neurol.34, 609-616. Mecocci, P., MacGarvey, U., & Beal, M. F. (1994) Ann Neurol. 36, 102-106. Nishimoto, I. et al. (1993) Nature 362, 75-79. Nukina N., & Ihara, Y. (1985) J Biochem (Tokyo) 98, 1715-1718. Parker, W. D., & Parks, J. K. (1995) Neurology 45, 482-486. Payne, D. M. et al. (1991) EMBO J.10, 885-892. Posada, J., & Cooper, J. A. (1992) Science 255, 212-215. Reed J., & Kinzel, V. (1984) Biochemistry 23, 968. Reed J. et al. (1985) Biochemistry 24, 2967. Robbins, D. J., & Cobb, M. H. (1992) Mol. Biol. Cell 3, 299-308. Robbins D. J. et al. (1993) J.Biol. Chem. 268, 5097-5106. Roder H. M., & Ingram, V. M. (1991) J Neurosci. 11, 3325-3343. Roder, H. M., Hoffman, F. J., & Schroder, W. (1995) J Neurochem. 64, 2203-2212. Romani A., & Scarpa, A. (1992) Arch. Bioch. Biophys. 298 (1), 1-12. Roses, A. D. (1994) J.Neuropathol.Exp. Neurol. 53, 429-437. Scott, J. D. (1991) Pharmacol. Ther. 50, 123. Segel, I. H. (1992) Enzyme Kinetics, John Wiley & Sons, New York. Seger, R. et al. (1992) Proc.Natl.Acad.Sci. USA 88, 6142-6146. Sherrington, R. et al. (1995) Nature 375, 754-760. 144 Steiber, A., Mourelatos, Z., Gonatas, N. K. (1996) Amer. J. Path. 148, 415-426. Steinberg, R. A. et al. (1993) Mol. Cell Biol. 13, 2332-2341. Termine et al., (1972) Biopolymers 11, 1103-1113. Terry, R.D., & Katzman, R. (1983) Ann. Neurol. 14, 497-506. Trojanowski, J. Q. & Lee, V.M-Y. (1995) FASEB J 9, 1570-1576. Ullrich, A, & Schlessinger, J. (1990) Cell 61, 203. Van Nostrand et al., (1989) Nature 341, 546-549. Wallace et al. (1991) Cytogenet. Cell. Genet. 58, 1103-1123. Wallace, D. W., (1992) Science 256, 628-632. Weingarten, M. D. et al. (1975) Proc. Natl. Acad Sci. USA 72, 1858-1862. Whitehouse S., & Walsh, D. A. (1982) J. Biol. Chem. 258, 3682-3692. Whitehouse S. et al. (1983) J. Biol. Chem. 258, 3693-3701. Wischik, C. M. et al. (1988) Proc. Natl. Acad. Sci. USA 85, 4506-4510. Yankner B. A., Duffy, L. K., & Kirscher, D. A. (1990) Science 250, 279-282. Zhang J. et al. (1994) Nature 367, 704-710. Zheng J. et al. (1993) Biochemistry 32, 2154-2161. 145 Chapter 8: Appendices 146 Appendix A: Rate equations for inhibition of a random sequential bireactant reaction A sequential bireactant reaction with substrate A and B binding randomly, with the binding of one substrate changing the dissociation constant for the other substrate by a factor ca, is described by the equilibria shown in Scheme 3-1. In the equilibria scheme shown in Scheme 3-3, I is an inhibitor that is capable of binding any form of the enzyme E to form up to 4 unique inhibitor complexes. There are 15 possible combinations of inhibitor complexes, each yielding a unique inhibition rate equation. Using the rapid equilibrium assumption, the rate equations for each possible combination of inhibitor complexes can be obtained by writing the reaction velocity as follows: (8A-1) v = kp[EAB] where [EAB] is the concentration of the species which yields product and kp is the catalytic rate constant. Dividing equation 8A-1 by the total concentration of enzyme, [E]t, yields an equation of following form: v kp[EAB] (8A-2) [E]t [E]+[EA]+[EB]+.-,and so on. where [E]t is expressed as the sum total of all species: [E] + [EA] + [EB] + .... Finally, the concentration of each species is expressed in terms of [E] to yield an equation of the following form [A][B] V [E]t oK AK 1+ [A] KA B (8A-3) [B]+(8 KB Equation 8A-3 can then be rearranged to the following Michaelis-Menton forms: v (8A-4) Vmax [A] acKA (slope terms) + [A](intercept terms) v Vmax [B] aKB (slope terms) + [B](intercept terms) (8A-5) and where Vmx = kp[E]t. All the species in Scheme 3-3 can be expressed in terms of [E] as shown below in Table 8A-1: 147 Table 8A-1. Expression of Scheme 3-3 species in terms of [E] Multiplier of [E] [A] Species [EA] KA [B] [EB] KB [EI] [I] KI [A][B] [EAB] OKAKB [A][I] [EAI] YKA K , [B][I] [EBI] PKBK, [A][B][I] [EABI] t3PyKAKBK, Fifteen different rate equations can be derived for the inhibition of a random sequential bireactant reaction. The Michaelis slope and intercept terms for the 15 rate equations, in the form described in Equations 8A-4 and 8A-5, are shown below in Tables 8A-2 and 8A-3, respectively. 148 Table 8A-2. Michaelis-Menton slope and intercept terms for random sequential bireactant reaction: Varying substrate A Inhibitor Complexes formed in Reaction Pathway EBI EABI EAI El Intercept terms Slope terms [B] [B] KB 1+ 1+ K [B] KB [B] 1+ + [B] [I] PK, [B] 1+ K + 1+ K, [I] K, [B] K [B] [B] K, [B] 1+ yK, [B] [I] aKB [I] [B] + yK, [B] [B] PyK, KB KB [I] [I] K K CI] B [B] K, [B] BK, [B] yK, [B] K, [B] + K, [I] K, [B] 1+ aK, + oKB [I] + [I] [B] yK, [B] ByK 1 + [I] PK, KB [B] + K, [B] + 1K, + [B] PK, KB [I] 149 1+ [I] PyK, 1+ OK B + otK B [I] + [I] 1+ K B + [I] [B] fyK [I] K [I] PKI I] 1+ + 3yK, + 1+ 1+ KB + KB [ [B] K, [B] 1+ [B] ciKB [B] B [I] + [B] PK, [B] atKB 1+ 1+ KB K yK, [B] [B] K_ 1+ B+ I] +KB 3K, ] 1+ K [B] PyK 1+K 1+ [I] K [I] + [B] K, [B] + aK B [B] zK B K B K B [I] + [I] 1+1+ [B] yK, [B] 1+ K + IB [I] aKB + 1+ K1+ [B] 1+K, 1+ KB 1+ K[I] K, [B] 1+ K [I] PK, 1 1+ [B] yK, [B] OyK, K, oKB [I] [I] [B] + + yK, [B] 3yK Table 8A-3. Michaelis-Menton slope and intercept terms for random sequential bireactant reaction: Varying substrate B Inhibitor Complexes formed in Reaction Pathway EABI EBI EAI El Intercept - terms Slove 1 terms 1+A + [A] 1+ aoKA KA [B] [I] K, [B] + [A] 1+ 1+aA yK [A] aKA A[I] 0tKA + PK, [A] [A] 1+ [A] 1+ 1+ K, [B] [A] 1+ aKA + [I] yK, + [I] yK1 [A] aKA [A] 1+ yK, KA [I] -+ AcKA + K, [B] +oKA cK A KA [I] + [A] K, [B] OLIA [A] c A [I] OK. [A] + [I] ByK, [A] [I] + + 1+ PyK, LKA [I] aK KA [I] 3K, [A] 1yK, +KA +caKA [I] ++ 1+ [A] PK, [A] PyK, CLKA + [I] 1+--[A] yK, 150 1+ yK 1 1+ -- + [A] K, [B] I+ PK, [A] [A] [I] [I] + [I] iK 1+ CKA + [I] K, [B] [A] [I] + [I] [A] PyK, +aKA 1+-+ [A] 1+ aKA + KA [I] + +KA [I] OK, [A] [A] 1 1+ 1+ a1KA [A] [A] 1yK, KA + [I] 1 1+ [A] OyK, 1aKA + ICKA [I] 1+ 13K [A] [A] 1+ aK A + KA [I] [A] K, [B] 1+ + [] 1+ K A [A] KA [I] A A [A] 1+ KA + KA [I] + [I] K, [B] yK, [A] aK [A] [I] 1+ aK [A] 1+ A A 1+ + [I] yK, 1+ XK [A] + aKA [I] + [I] PK, [A] 1yK, Appendix B: Rate equations for inhibition of an ordered sequential bireactant reaction A sequential bireactant reaction with substrate A and B binding in an ordered fashion, with substrate A binding first, is described by the equilibria shown in Scheme 3-2. In the equilibria scheme shown in Scheme 3-4, I is an inhibitor that is capable of binding any form of the enzyme E to form up to 4 unique inhibitor complexes. There are 15 possible combinations of inhibitor complexes, each yielding a unique inhibition rate equation. Using the rapid equilibrium assumption, the rate equations for each possible combination of inhibitor complexes can be written in the following Michaelis-Menton forms: v [A] Vma,, KA(slope terms) + [A](intercept terms) and v Vma. S[B] [B] KB (slope terms) + [B](intercept terms) (8B-2) where V, = kp[E]t. All the species in Scheme 3-3 can be expressed in terms of [E] as shown below in Table 8B-1: Table 8B-1. Expression of Scheme 3-4 species in terms of [E] Multiplier of [E] [A] Species [EA] KA [EI] [I] K, [A][B] [EAB] KA KB [A][I] [EAI] YKAK, [B][I] [EBI] PKBKI [A][B][I] [EABI] _pyKAK,K Fifteen different rate equations can be derived for the inhibition of an ordered sequential bireactant reaction. The Michaelis slope and intercept terms for the 15 rate equations, in the form described in Equations 8A-1 and 8A-2, are shown below in Tables 8B-2 and 8B-3, respectively. 152 Table 8B-2. Michaelis-Menton slope and intercept terms for ordered sequential bireactant reaction: Varying substrate A Inhibitor Complexes formed in Reaction Pathway El EAI EBI EABI N Intercept m terms Slope terms m I KB K, [I] [B] K, [B] KB 1+B] [B] I KB 1+ [B] KB [I] KB [B] OK, [B] KB K_ KB [I] [B] [B] oc3yK, + KB [I] [B] .K, B 1+ KB [I] +K,[B] [I] + K, [B] K [I] 1K +B 1+ [B] K, [B] [B] 1+ KB + [I] [B] PK, 1aK, 1+B+ yK, [B] [B] [I] cpyK, aK, [I] yK, [B] [I] 1+ aK, + OaK B [I] + [B] yK, [B] pyK, 1+ K , [B] 1+ CaK B + [I] KB + [I] 1+KB PK , KB KB [I] [I] [B] K, [B] OK, K, + KB [I] [B] KB [I] 1 KB [B] PK, 1 KB [B] + [B] K, [B] [B] KB aKB [I] yK, [B] B [B] [B] 1+K B [B] ByK, K [I] yK, [B] [I] [I] + 1+ KB + KB K, [B] [B] yK, [B] ocpyK, KB KB [I] [I] K+ [I] [B] K, [B] 1K, [B] c3pyK, KB [B] KB [B] + [I] + 1+ BK, B± [B] KB [I] [I] K, [B] PK, 153 K 1+ K, [B] KB [I] + yK,[B] + [I] ocpyK, KB [I] [I] yK, [B] al3yK, Table 8B-3. Michaelis-Menton slope and intercept terms for random sequential bireactant reaction: Varying substrate B Inhibitor Complexes formed in Reaction Pathway EABI EBI EAI El Intercept terms Slope terms 1 KA [I] 1+ K A [A] K, [A] [I] KA 1 1+ KA+ [A] SKA 1+ [A] aK, [I] [I] K, [A] yK, A 1++KA + KA [I] KA + KA PK, [A] [I] aP1K, 1+ KA [I] 1+ KA + [I] PK, [A] yK, [A] [I] [I] KA 1+ + 1+ yK, [A] acyK 1+ KA 1+ 1+ KA + KA [I] + [I] [A] K, [A] yK, KA KA [I] [I] 1+ K, [A] KA + [A] [I] [I] ocoyK, 1+ K, [A] [I] yK, [I] 1+ KA + KA [I] + [A] K, [A] yK, 154 acpyK, 13K [A] yK, 1+--+ [A] [I] + 1+ KA [I] KA [I] 1+ K+ + + [A] KA PK, [A] [A] 1+ [I [ 1+ K, [A] [A] [I] +KA 1+ K, [A] [A] 1+ K, [A] 1+ [A] K KA KA [I] 1+ [A] 1+ 1+ yK1 1+ KA [I] + [I] PK, [A] xocyK, KA [I] PK, [A] + [I] aloK, [I] + [I] 1+ KA 3K,[A] upyK, Appendix C: Rate equations for non-essential activation of a random sequential bireactant reaction A sequential bireactant reaction with substrate A and B binding randomly, with the binding of one substrate changing the dissociation constant for the other substrate by a factor a, is described by the equilibria shown in Scheme 3-1. In the equilibria scheme shown in Scheme 3-5, M is a non-essential activator that binds to all forms of the enzyme E to form 4 unique activator complexes. Using the rapid equilibrium assumption, the rate equations for the random sequential bireactant reaction can be obtained by writing the reaction velocity as follows: v = kp[EAB] + Okp[MEAB] (8C-1) where [EAB] is the concentration of the species which yields product, kp is the catalytic rate constant and 0 is the activator rate enhancement factor. Dividing equation 8C-1 by the total concentration of enzyme, [E]t, yields an equation of following form: v [E]t kp[EAB] + 0kp[MEAB] [E] + [EA] + [EB]+... (8C-2) ,and so on. where [E]t is expressed as the sum total of all species: [E] + [EA] + [EB] + .... Finally, the concentration of each species is expressed in terms of [E] to yield an equation of the following form [A][B] [A][B] v _= [E]t KAKB r yKMKAKB 1+ [A] +[B] KA (8C-3) KB All the species in Scheme 3-5 can be expressed in terms of [E] as shown below in Table 8C-1: 155 Table 8C-1. Expression of Scheme 3-5 species in terms of [E] Multiplier of [E [A] Species [EA] KA [B] [EB] KB [EM] [M] KM [EAB] [A][B] [EAM] cKAKB [A][M] YKAKM [B][M] 3KBKM [EBM] [A][B][M] [EABM] a(OYKAKBKM 156 The rate equations non-essential activation of the random sequential bireactant reaction can be written in the Michaelis forms described in Equations 8C-4 and 8C-5 for varying substrate A and varying substrate B, respectively. (8C-4) v= aKA [B] + PKMJ +[A](+ [B] + B yKM[B] + [M yKM) Vmax[B](1+ O1M v ( KA 1+ K [M] [A] yKm [ +[B 1K + [B] 1+ [A] 157 + [ PK,[A] yKm (8C-5) Appendix D: Rate equations for non-essential activation of an ordered sequential bireactant reaction A sequential bireactant reaction with substrate A and B binding in an ordered fashion, with substrate A binding first, is described by the equilibria shown in Scheme 3-2. In the equilibria scheme shown in Scheme 3-6, M is a non-essential activator that binds to all forms of the enzyme E to form 4 unique activator complexes. Using the rapid equilibrium assumption, the rate equations for the ordered sequential bireactant reaction can be obtained as described in Appendix 8C for a random sequential bireactant reaction All the species in Scheme 3-6 can be expressed in terms of [E] as shown below in Table 8D-1: Table 8D-1. Expression of Scheme 3-6 species in terms of [E] Multiplier of [E] [A] Species [EA] KA [EM] [M] KM [EAB] [A][B] KB [A][M] [EAM] YKAKM [A][B][M] [EABM] c/PyKAKBKM 158 The rate equations for non-essential activation of the ordered sequential bireactant reaction can be written in the Michaelis forms described in Equations 8D-4 and 8D-5 for varying substrate A and varying substrate B, respectively. 0[M] Vmax [A] 1+ KA(\K[B] +K O[M] L(Jy OYKAK ra +[A] 1+ [M]KB KA[B] [B]KAK, ) , (8D-4) KB+MI M yKAKm[B] OyKAKm V f±O[M] (8D-5) S 1 KA [M] 1+ -A]K+ [+ [A] [A]K, + [B]i [M] yKA,K +[B] 1+ M 159 [M] yKAK,