P. PETROLOGY NEW MEXICO GEOCHEMISTRY
advertisement

.
.
PETROLOGY AND GEOCHEMISTRY .OF THE PALISADES SILL, NEW MEXICO
OPEN FILE REPORT 81
by
Robert P. Cannon
...
,
A t h e s i s submitted to the faculty.of the University of North
C&oline i n p a r t i a l f d f i l l m e n t o f t h e
fequirements for the
degree of Master of Science i n the Department of Geology.
Chapel Hill
1976
Reader
Reader
I
.
.
....
. ..
I
..
"
.
.
..
, .
.
ROBERT PAUi CANNON. Petrology, and geochemistry of the Palisades sill,
S i x t y five
saaales were collected alongtwo profiles from the
"
Paliaal=s SA.,
eastwarl dtpping daciti-
:he lowest of Sour to sin
thick sills cropping out in Cimarron Canyon, Colfax County, Northern
New Mexico.. Both profiles contein samples from tka t=
uppar
ths
lo%zr ccntect of the sill; one profile is near-horizontal
the and
cther is sear-vezticsl.
All samples were analyzed for major elements,
Rb, atis Sr by x-ray fluorescence
Microscopic
and
atonic
petrographywza'perfomed on thin
absorption
sections
spectrozztzyc
prepared
from
twenty repzesantative samples.
The
excapti
.
sill
exhibits
little
mineralogical
or chenical
Variati9n
frtz K q 3 , Rb, and CaO concentrations. Sod6m d Rb sho-; a
8erong nege:ive correlation with
greater
CaO.. Elemental variation is much
the
insmples frciii the hsrizoniai profile than from verticd
profile, with samples from the horizontal profile showing an indistix?:
cyclic variation. Approximately 40 percent of the sill is phenocrysts
of plzgioclase, quartz, and biotite; the remaining
60 percent is a
groundmass of orthoclase, plagioclase, quartz, biotite, and magnetite.
Quartz phenocrysts are embayed and rounded. Some biotite
phewcryats
are altered to chlorite or to sericite, magnetite, and 1eiicoxer.e.
Voids in all types
of phenocrysts exceptqurtz and in the groundmass
occupy approximately1 percent of the
rock,volume.
Chemical and.mineralogica1 data suggest thatmagma
the underwent
its present
equilibrium crystallization and that the magma intruded
to
position in subvertical isochemical and isomineralogic "fronts".
The
phenocrysts apparently crystallized ain
deep magma chamber; the ground-
..
..
....
3
...
i.
Page
...............................................
vi
List of figures.
List of
I""d""3"
L'il"
.......................................
tahles
..Cftrc.Lt.*..-.*=..
... --.&-Xm- ...... . . .
1
Y
C
L
I
"
.
'
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
Geologic.SE:;-mg
...............................................
..............................................
Previous Mrk
Field relaEionships
.......................................
.........................
Geoche%ist ry .............................
Sampling procedures
...............
........................
Sample preparations .......................................
................
..........
Major elements
....................
Normative amlyses................ ...... ................
. Chemical comparison with "typical"
' igneous rocks ..........
Rubidium andstrontiE....................................
Petrography ....................................................
Plagioclase
phenocrysts
....................................
Quartz phenocrysts; .......................................
Biotite, chlorite. and magnetite phenocrysts
..............
Croundmass.................................................
.
.........................................
Crystallization and alteration
.................................
Intrusion..................................................
Relation between inzgna and phenocrysts
...............
Dissolution
voids
..........
........................................
.......................................
................................
Effects of pressure change during intrusion
Chill zone. magma convection. and temperature
distribution
Deuteric alteration
Phenocryst alteration
Phenocryst and groundmass dissolution
Comparison with experimental results
................
......................
............................................
Conclusion......................................................
Mode
of
emplacement
AcknowleJ&ments
.................................................
. .
..
.
.
....
3
3
3
6
6
8
9
13
16
16
21
23
24
29
33
34
40
40
40
41
44
48
48
49
50
56
63
66
...
. .
V
P"fF
References cited.
..............................................
67
.......................
71
Appendix A: Whole-rock chemicalanalyses
AT"t &JF.I" 72..
L.
E: **Stratigraphic"sample poaitior?....................
Appendix C: Analyticalprocedures,precision,
and accuracy.....
73
74
.
\I\
..
..
. . .
__
... . . . . . .
....
.
.
..
:
. .
...
j
LIST OF FIGURES
Figure
.
2.
1
3
.
paw
.................................
P a l i s a d e s s i l l and v i c i n i t y ................
LocationofPalisades
Geologic map of
sill
SchematicdiagramofPalisaden
profiles
.
9.
10
.
.................
i n p l a g i o c l a s e ...........................
17
.
7
11
12
11
17
Rb F, S r d i s t r i b u t i o n i n 11- and V- p r o f i l e s
18
B i o t i t ei n c l u s i o n
22
S o l i d s o l u t i o n o f plagioclase phenocryst in system
An-Ab-Or
...................................................
11. Highly embayed quartzphenocryst ...........................
1 2. S l i g h t l y emhayed quartzphenocryst .........................
13. Partially resorbed quartz phenocryst with
rim ..............
14 . Brown b i o t i t e surrounded by g r e e n b i o t i t e ..................
15. P a r t i a l l y andcompletelyreactedbiotite
...................
16;
4
s i l l nnd c o l l e c t i o n
...................................................
4 . Oxide d i s t r i b u t i o n i n H-prof.ile ............................
5 . Oxide d i s t r i b u t i o n i n V - p r o f i l e ............................
6 . CaO versus Na20 - a l l samples ..............................
7 . S i l i c a / d i f f e r e n t i a t i o n inde.x - P a l i s a d e s s i l l versus
"typical"igneousrocks
....................................
8
2
B i o t i t e .s e r i c i t e .
relationships
Rnd magnettte showing r e a c t i o n
..............................................
Dissolution void in plagioclase phenocryst
groundmass
26
27
28
30
31
32
and
..................................................
18 . Dissolutionvoids ...........................................
19. Dissolutionvoids
1-5
...........................................
35
36
37
vii
Figure
Page
20.
Dissolution voids filled withquartz......................
21.
Pressure temperature diagram demonstrating
quartz crystallization- resorption history...............
39
. .
-
..................................
43.
22.
N a p s convection diagram
47
23.
Phase diagramfor crystallization of the
Palisades s i l l magma....
51
Projection of samples H13 and H23 on the system
An-Ab-Or-Q
54
24.
..................................
................................................
25.
Relationships of whole-rock composition with phenocryst
and groor.2i;ass coiiiposition in the systemAn-Ab-Q
55
26.
CaO & Rb versus sample'location in the
27.
Element distributionin the sill prior to convection...... 5 8
28.
p "..
25.
Location of Palisades sillto possible subductionzone....
62
30.
Phases and crystallizationand alteration
processes ve temperature of crystallization
64
Oxide concentrations in the magma
vs temperature
of crystnllization
65
31.
..........
€I-profile
..........
57
chamber and element segregation prior to intrusion..60
...............
........................................
..
LIST OF TABLES
Table
page
for major elements,
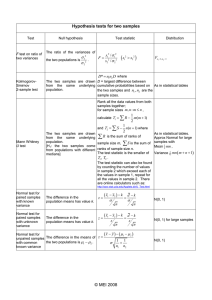
1. Hean and'stendard deviation
Rb, and Sr
............... ;.................................
I. P. W. n o r m s . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .
10
15
2.
C.
3.
Oxide distribution "normal" igneous rocks
versus Falisades sill samples
19
Modal data from twenty thin sections.......................
23
4.
-
..............................
...
.....
.
INTRODUCPION
The P a l i s a d e s s i l l is t h e ' l o w e s t of four t o s i x t h i c k , l o b a t e
d a c i t e p o r p h y j sills' cropping out i n and near Cimarron Canyon, Colfax
County, New Mexico.
The sill is v i s i b l e as c l i f f s j u s t w e s t of Phil-
mont Scout Ranch alolig U. E. 64 betweenEagle
Nest andCimerron;
New
Mexico (Fig. 1). The sill is a p p r o x i m t e l y 370 m t h i c k and outcrops
w i t h i n an a r e a ' o f 3 km by 3 km.
It i s roughly concordant with the
c o n t a c t s between .underlying Precambrisn
overlying Permian
to
Jurassic sedimentery sequence.
The purpose o f this resairrch
mediate composition
metamorphic rocks and an
is
t o y:u:s
8
large.s:ll
Gf inter-.
by petrographic and geochemical m e t k d s a n d , t o
model i:s c r y s t a l l i z a t i o n h i s t o r y . S i l l -
or. l o p o l i t h - l i k e m a f i c
bodies such as' the' famous Skaergaard intrusion (Wager and Deer, 1939)
and Bushveld cosplcx <Halli 1532) h m e been studied in g r e a t detail,
Few l a r g e sills o f f e l s i c or intermediate composition have.been
systematically studied, primarily because of their relative scarcity.
The Palisades s i l l , New Mexico is i d e a l f o r s u c h
a study because
sill dips aPPrunrulacslr
1__-_"l2O t o t h e northeast, allowing collection
from the lower to upper contacts.
the
2
..
. D
..
SAMPLE LOCALITY p!,
L\9
1
Espanok
. . '
Appox. malt
Figure 1. General location of the Palisades sill (after Northrop and
Read, 1966. p. 10).
..
. ,
. . .
.....
. .
..........
. .
. . .
. . .
. . . . . . . . . . .
.
..
.
. . . . . . . . . .
...........
............
.
.
.
.
. . . . .
-
. . . ' .
., ..........
2'
..
.
.
-.
.' .
'.)
...L ....
..
.
' : .)
'.
,
I
;i
. .
.
GWILOGiC SETING
'
'
Previous Work
Thc; E=st:'recent mapping of t h e P a l i s a d e s s i l l area was performed
by Robinsonand
o t h e r s (1964).Previous
a d F+y (1941, 1943).Smithend
mapping was done by Smith
Ray (1941,p.192)
s i l l material as quartz mmzonite porphyry
as p o r p h y r i t i c monzonite(Smithand
c l a s s i f i e dt h e
and lakr r e c l a s s i f i e d i t
b y . . l 9 4 3 ,p .
904).Robinsonand
i n the s i l i e as
o t h e z s (2,954, p. 5 0 ) remmed t h e dominantrocktype
d a c i t e porphyry.Thesepapers
related'sills :=:rdd&
s t a t e t h a tt h eP a l i s a d e s
during the Tertiary,
sill and
b u t no other canclusions
concerning petrogenesis are given.
. .
Field Relationships
The l i g h t c o l o r e d P a l i s a d e s
sill contains approximately, 40 per-
cent phenccrjrsts of plagioclase, quartz,
groundEse.
and b i o t i t e i n a t a n
The only vis'ible difference in the rock throughout
s i l l is t h e amount 6f quartz phenecrysts, varying in
1 t o 10 percent.
the
abundancefrom
'
Mstir;ct columnar j o i n t i n g i s found i n most p a r t s of t h e sill.
The s i l l rests conformably on the top
(Fig. 23.
of Precambrian amphibolite
The contact is d i s t i n c t and is locatedwithin
than 0 . 3 m wide.
a zone less
No contact metamorphism i s v i s i b l e i n t h e
amphibo-
l i t e and no xenoliths or evidence of a s s i m i l a t i o n are present i n
the sill.
Tha upper c o n t a c t , o b e a r v a b l r i n t h e v i c i n i t y
of Cimarron Canyon,
.
. . . . ...
. .
.. . .
.
. .
3
.
.-.
.: 1
..
4
. I
. .,.
. .
0
.
2 Km
I'
8
Figure 2.
Geologic map of the Palisades sill (containing pror'ilesj and
country rocks (after Robinson and others, 1964, Plate 3).
area is Tertiary Palisades.sil1 and related sills.
White
Dotted area is
undifferentiated Trias,sic, Jurassic and Cretaceous sedimentary
rocks.
Diagonally crosshatched area is Precambrian metamorphic
and igneous rocks.
Area with circles is Quaternarystream and
landslide deposits.
. .
. ..
'
. .
.
. .
-__
.. .
..:.:I
.
is morevaguebecause
.
. 5
i t i s in a n a r e a o f l e s s s t e e p
topography w i t h
. ,
a s i g n i f i c a n t soil p r o f i l e and vegetation. .The .contact
can only b e
..
locatedwithin 2 to, 4 m of"stratigraphic"
distance(distance
measured
..
. .
..
.
..
I n addition,theuppercontact
perpandicelor t o contzct-a),
is not
since it c u t s c o n t a c t s
..
. . . .
between Triassic Dockum,Group and Jurassic Entrada 'Sandstone as w e l l
s t r i c t l y concordant with overlying strata,
a s between Jurassic Entrada Sandstone and T r i a s s i c Morrison Formation
(Robinson and o t h e r s , 1964, Plate 3).
.
sedi-
.
from t h e lowermost of t h e o t h e r
mentary rocks separates this-sill
sills.
A 60 t o $0 m s e c t i o n o f
The s i l l varies I n thickness by about 30 m, having a d i s t i n c t
lobate' formandrounding
1964, Plate 3 ) .
o f f a t i t s extremities (Robinson and o t h e r s ,
Along thethickcentralportion
which t h e a u t h o r c c l k c t e d h i s
. NlOW and dip 1
2'
of tii+ s i l l , from
samples,contacts
.
s t r i k e roughly
.
. ..
NE.
. .
. .
.
.
.
. .
.. .
.. .
..
. .
.. .
.
..
.....
GEOCHEMISTRY
. S i l i c a , Ti02, Al203, Fe2O3*
.
-'
( t o t a l Fe as Fe203).
K20, Rb, and S r were analyzed for a l l samples.
a l l analyses.
.eo,
.
CaO, Na20,
.
'
Appendix A t a b u l a t e s
Loss on ignition procedure was performed on f i v e s a m p l e s
to check how c l o s e l y tlie analyses totaled 100 percent.
The mean and
s t a n d a r d d e v i a t i o n for the t o t a h o f . t h e f i v e zEdyses 2re 100.08
-+
0.65.
Sampling Procedures
Samples were collected along two p r o f i l e s which &end
bottom t o thetopofthe
sill.
One p r o f i l e , r e f e r r e d
are labeled H1, H2,
t r e n d ss o u t h
.... H40.
.
;'53
andplunges
are l a b e l e d V 1 , V2,
.
.... V25.
t o as t h e H-pro-
N75E. approximatelyperpendicular
f i l e , i s roughly horizonfal.and trends
t o s t r i k e of t h e s i l l (Fi'g. 2).
from t h e
.Samples c o l l e c t e da l o n gt h i sp r o f i l e
.
:
-
The o t h e rp r o f i l e ,c a l l e dt h eV - p r o f i l e ,
.
5
.
Samples c o l l e c t e da l o n gt h i sp r o f i l e
Samples w i t h l a r g e r
.
numbers are progres-
s i v e l y nearer the upper contact i n each profile.
. .
. . -. ... .. . .
These two p r o f i l e s were chosen in hope ' t h a t any c o E i . s t e n t l a r g e -
...
scale chemical o r p e t r o l o g i c a l v a r i a t i o n e i t h e r . v e r t i c a l l y o r
Any large-scale,
h o r i z o n t a l l y would beobserved.
through the sill should be observable except'
non-random v a r i a t i o n
if the isopleths (surfaces
are p a r a l l e l t o a plane contain-
of similar mineralogyandchemistry)
ing tho two p r o f i l e s , t h a t p l a n e s t r i k i n g a p p r o x i r e a t e l y
N75E and
dlpping 36O SE (Fig. 3 ) .
It would seem p l a u s i b l e t h a t i s o p l e t h s on i n t r u s i o n would be
,.
..
. .
.
.
,.
.
.
_ .
.
.
.. .. . . . . . . .
..
.
.
j .
. .
.
.
. .~
. .
.
7
. ...
Palisades
Slll
FiRure 3.
Schematic diagram of. the Palisades s i l l , c o i l e c t i o n pro-
f i l e s , and planescontainingtheprofiles.
. .
..
1
. . _..
..
.
.,
.
..
5
8
of (1) layering of s e p a r a t e s i l l - l i k e
essentially horizontal because
i n j e c t i o n s , (2) c r y s t a l s e t t l i n g a n d l o r f l o a t i n g ,
or (3) layerixzg due
t o c h i l l zones of different composition than material near t h e c e n t e r
..
. .
. ..
of the s i l l . . Conversely, isopleths could be near-vettigal.
and p k r a l k X
I f t b isopleths are near-horizontal, dete
t o a feederdike.
..
from
both p r o f i r e s should Blioi~i t since both a r e c o l l e c t e d from top to
bostciii.
Tkc E-prcffleshould
show any large-scalev2riatLoort.unless
t h e i m p l n - t h s trend near N75E.. V-profileshould
show v a r i a t i o n i f
From the work of Robinson and
the isopleths are not near east-west.
.
o t h e r s (1964, Plates 3 and f ) , the feeders
that m
g iztruded
~
easwrd.
.
seem t o b e t o t h e
west so
If t h i s i s thecase,yariation
from
iliject-lotr "fronts" would be easily recognizable in the H-profile.
Samples from t h e H- and V- profilez werecoZ1,ected
et appproxi-
mately15 m zad 9 m'"stral$.graphic"intervals,respectively.Reasons
for inzccuracies and ' i r r e g u l a r i t i e s i n t h e l o c a t i o n s
. .
of sample sites
.
are finding fresh samples in'place (i.a*, collection site depends
outcrops available),and iaharent inaccuracies
is 383 m and 345 m respectively.
In order t o make c o r r e l a t i o n s alczg
i n t h e sill, the scale for the V-profile
changed t o be 383 m thick, equal
t o the thickness
sites from the bottom of the
was
of H-profile.
and, f o i '+profile, the
AppenGix B g i v e s t h e o r i g i o a l
d i s t a n c e s of sample
when locating each site.
of the s i l l for the H- and V- p r o f i l e s
The calculsted thickness
"stratigrsphic" levels
on
recdc-datd
Sill.
Sample Preparation
and trimmed so .that no weathered surfaces
Samples were checked
. .
wereused
foranalysis.
The fresh material was subjectedtothe.
following:
.- .
...
,
.
..
. ..
.
_.
..
^_",
....I
~
..
. _.
'
c
"
:
,.?I...
9
Jaw crusher
sample
splitter
J .
...
small jaw crusher
with
alumina
ceramic
\
.
,
part of sample
to be kept
. .
plates
. . . .
. . . .
k
sample splitter
-'
J.
part of sample
to be .kept
semi-micro pulverizer with
a1umir.a ceramic prates
. .
Spex
shatterbox
-&
hydraulic
press
dissolved
for HF-H3B03
by
pelletizing
(Bland.
1972)
decomposition
technique
I
"
Norelco
universal
vacuwpath
X-ray
spectrometer
atomic
absorption
unit
(for S i , Ti, Ca, Rb,
Sr)
Appendix C' gives
methods
used
(Wooden, 1975,p. 154-155)
Perkin-Elmer 303
(for Na, K, Al, Mg, Fe)
.for
the
analyses.
.
'
Maj.or Elements
..
. .
Prom Teble1, Appendix A;mnd
Figures'4 and 5, it is obvious that
oxide concentrations vary little over the entire sill, except for
samples
V23,
VZ4,
and"$25
whose
anomalous
values
will
be
discussed
stands-rd deviation, and
in a later section. Table 1 gives the mean,
coefficient of variation for all samples (v23, V24, and V25 excluded
from this and subsequent tables unless otherwise noted) and for
. . .
samples from8- and V- profiles individually. Variation for most
. .
elements i s greater.in.theH-profile than in the V-profile; therefore, correlations among elemental concentrations are more apparent
in the H-profile.
...
. . . . . . . . . . . . .
, .
. . . . . .
- ...
~.
..
"-.........
. .
- 9.
TABLE 1.
.-
element
mean
conc
or oxide
a l l samples
MEAN AND STANDARD
menn conc.H-profile
V*
0-
DEVIATION FOR MAJOR ELEMENTS, Rb, AND Sr.
.
.-
V
0-
mcan
conc
eprofile
.
V
0-
'
... ....
5
si02
57.75
2.81
1.90
67.67 41 ,67.62
1.55
,1.05
15;71
1.97
0.31
15.71
Al 2O3
.
. .
2.50Pe203
0.1116.18
CaO
2.60
0.78 .JO.OO
4.66
.
0.55
CaOtNa20
7.28
0.32
4.40
K20
3.29
0.12
*
16.67
0.11
11-80
.
0.6313.48
4.68
"
3.65
.
3-31
0.28
700 ppm 30.43213
"
59
4 7.59
coefficient of variation = 100 u/x
.
59
0.30
1.91
'
7.14 0.02
4.63
. . . .
0.40
'!
'
"
"
3.25
.2.46
0.08
0.29
0.03
10.34'
. .
2
3.45
"
'
8.47
18
"
.
8-64',
24.20
175 723
5
.
0.21'8.20
25.48
0.66
2.59
',
3.93
0.13
34.02
230 676
1.36
12.680.D90.71
31.58
2.66
. 0.84
7.14 0.02 0.28 Ti02
Sr
. .
0.'66
0..68
Rb
.
,
.2.56 4.84 0.12
0.16
2;'48 6.40
xgo
Na20
L5.71
2.04
0.32
.
0.92
. .... ')
:'W>
.,.
1.:
.......
.
....
/,
.
.
.
..
...
.
.
. .
. . . . . .
- ...
.. .. _. .
. . . . . . . . .. .. . . .
........
. .
.......
a
0
.
. .
. .
. .
.
.
Figure 5.
Oxide distribution i n theV-profile.
vertical axes represent the
Longer tic markn on the
mean values for each oxide.
.
*.
I
." i:)
1:
13
..I
The following observations with regard to elemental variations
Si02
-
4 and 5 :
and V25 excluded) using Figures
can be made(V23,V24,
Variation is q u i t e small.
Soda and S i 0e x h i b i t
2.
c o r r e l a t i o n ; CaO and SiOi, a ' n e g a t i v e c o r r e l a t i o n .
a positive
Samples V5 and V6
show a minor c o n t r a d i c t i o n t o t h i s t r e n d .
Ti02 - There i s i n s i g n i f i c a n t v a r i a t i o n e x c e p t
in samples V18 to.V22.
(- Ti02 mean) f o r V17 t o 0 . 3 6 . f o r
Percent Ti02 increases from0.28
v22.
A1203
-
Pep3*
Alumina shows a poor positive correlatio!? with Si02.
- T o t a l iron e x h i b i t s minor v a r h t i o n e x c e p t f o r
samples V 1 8
*
Fe 0
is highwithvalues.rangingfrom
2 3.
2.75 t o 3.02 i n comparisonwith a mean of 2.50 and w o f 0.16.
t o V22.
'..
MgO
For thesesamples,
- Magnesia
is random.
shows more variation. than Ti02 o r PeZ03*, b u t v a r i a t i o n
Samples V18 t o V22 have highvaluesrenging
0.90 in conparison with e Eeen cf 0,68 end r o o f
Q:Uc
from 0.76 t o
Note,thppe high
values are from t h e samples t h. a t. have
.
high
of T i 0 2 and
. . . .percentages
.
Fe203*.
Sample H33.has a low value of 0 . 2 4 ; sample H11. a highvalue
of 0.96.
CaO and Na20
r
-
K20
-0.95
- These oxides
(Pig. 6).
- Potashexhibits
cotrtactj
shcw a strong negetivo correlation, with
l i t t l e variation.
Sample H&O (nearesttheupper
iits a c i i g b t i y h i & K2G Vaiee of 3.56 coEp&id ~ i t h
~
~
6
of 3.29 and u o f 0.12.
Normative Analyses
Becausechemicalcompositions
out t h e s i l l (except V23,V24,
and V25), n o m c a l c u l a t e d from oxide
analysesarealsoverysimilar.Table
..
_ .
..... .
.
are very similar f o r asmples through-
2 gives C. I. P. W. norms of
.......
. . _.
.. . . . . . .
..
~-
. . . . .....
......
. . .
5
-.,
.. .
j::
:)
14
6
5
..
..
*.
.
3
2
I
0
2
3
4
.
Figure 6.
.
.
..
..
.
. ....
CaO
VR
Na20 for.all samples.
..
...
.
.
.
.
., .
,
,
.
. ..
. . . ...
.. ..
.-.
.
..
. .
. ..
,
. ..
..
_.. . ..
. V23,V24,
and V25 and t h e mean, standard deviation and c o e f f i c i e n t o f
variationfor16other
randomly, selected.samples.
The noms are
calculated from d a t a g i v e n - i n Appendix
Awith total oxides recdlculated.
. .
t o t o t a l 100 percent.
.
.
.
TABLE 2.
C. I . P.
.
W; NORMS.
.
.
"_
Norcative
.%nerals
[anhydrous)
Mean Values
(16 samples)
'
Q
.
V24
V23
V
".
Quartz
21.88
1.63
7.45
29.50
27.94 28.24
Orthoclase
0.82
4.04 24.16
24.39
19.68.
20.28
Albite
40.78
4.32
10.59
36.34 40.46 39.10
Anorthite
11.47
2.77 24.15
8.11
3.18
8.40
Diopside*
0.39
0.00
0.65'166.67
0.00
0.00
Hypersthene
1.61
.0.35
.21.74.
0.41
0.62
Hemtite
7.450.192.55
1.05
1.06
1.56
Titanite
9.85
o.i8 0.73 0.74
0.71
0.07
Corundum*
0.35
106.06
0.20 . 1.54 . 1.65
-_
0.33
Average contains both diopside and corundum, although in6:vidual
noms do' not
~~
*
"
"
.
"
_
i
_.-A
.
,.
.
. .
Since C. I. P. W. norms'calculate only anhydrousminerals,
diopsiZs
s z x . k , ~ e r s t h e n e a p p e a r i n t h e norm a l t h o u g h n e i t : l r r a r e . o p t i c a i i y
Fe203 .: F.eO r a t i o was assumed t o b e i n f i n i t e .
tectedintherock.
and quartz contents w i l l b e r e l a t i v e l y a c c u r a t e .
Nevertheless; feldspar
In 10 of t h e 1 6 c a l c u l a t i o n s ,
, .
iccl*&ee dinpai2e.
de-
.
h1203 is . n o t i n excess and. t h e norm'.
.
From normative analyses, the
s i l l i s composed primarily of d a c i t e ;
some samples, whose normative q u a r t z , i s . l e s s t h a n 20 percent, are com-
posedof
quartz latite-andesite (classification based on
liyndrnan; 1972,
p. 35).
Using d a t a fromAppendix A, normative a l b i t e and a n o r t h i t e were
c a l c u l a t e d and An (AnxlOO/ArrtAb) f o r t h e whole rock.wasdetermined
. ..
. .
. .. . ... .
..
. ..
....
-
..
,:;:.i
..
16
,._.
.I
for each sample. The An range is extreme, 9.4 for sampleV24 to 41.0
for
sampleH20, with all samples giving
a mean of 24.3 and
o o f 7.9.
Chemical Comparison with "Typical" Igneous .. Rocks . .
. .
. .. .
. .
.
'
. .
Tha n a a oxide
percentages
for
the
Palisades.sil1~ qwere
a r e d
against contoured frequency diagrams' (oxide percentage versus differ.
..
. ...
(1960) usisg.the
entiatin= idex! prepared by Thornton and Tuttle
.
.
5000 analyses in Washington's (1917) tabies. Differentiation indexis
the sum of three normative'minerals, quartz, orthoclase, and albite,
and is 80
t3
85 for w s t Palisades
sill samples.
For Si02 versus D. I. (differentiation index), Palisades sill
samples plot at the contour
maxim-a in the Si0 oversaturaeed ragis= .
..
.
.'
2
but ina higher SiOz snd D. I. range than typic& dacite, approximately
in the rhyodacite-rhyolite area (Fig.7).
r e s d t of
the
T h i s discrepancy is
(1
Palisades
sill's having 'a lower percentage of femic
mir?ezels th=n typic-1. &cite;
In edditiz-,.its high NazO content
.
. . . ,
.. .
name, dacite, to'be somewhat misleading from the
causes the rock
rock's real composition.
Potash,
NO, Fe203*,
and A l 203 plot near the center
of the contour
maximum on their respective.oxideversus.D. I. diagram (Teble 3).
Na20 and CxO are slightly highD. for
I. = 80 to 85, but bath are
.
within
the
.
..
.
.
maximum.
Rubidim a3d Strontium
Rubidium
varias
1080 ppm (Fig. 8 ) .
greatly
aver sili,.
the with a range of251 to
The mean for all samples
is 694 ppm and c i s 210.
Variation is nan-random and the only correlation that can be made with
other
(r
-
oxidesis a poor
"0.65).
positive
correlation
betwaen
Rb and Na0
2
No correlation can be drawn between
Rb and K20 (r
. ..
.
-
-O.lO),
. .. .
:..T)
..
.
..
.
,
17
.
. .
. .
..
,
..
1
10
..
0
100 .
.
80
40
.
20
1.
'.I1
DIFFERENTIATION INDEX
Figure 7.
Silica / differentiation index plot for PnliandeR sill ond
"typical" igneous rocks (after Thornton and.Tuttle. 1960, p. 675).
..
.
.
.
. .
.
.. , ... . .
. .
.
.
.
._...
.
. ..
..
.
0
i
\
i
r;
b
i
3
OS€
r(221)
2
Y
which i s contrary to general observations of igneous rocks since
Rb are chemically'very s b i l a r (Taylor,1965,
Potassium is extremely .invariant,
K and
p. '229).
andTauson,1965,
and i t is obvious t h a t Rb end K20
direct>; te:a:ed.
concen_traricns cenrat be
-
TABLE 3.
OXIDE DISTRIBUTION
" N O W ' IGNEOtiS K W K S VEKsLrS
PALISADES SILL SAMPLES
~-
_.."
__._"
"
.
"
Approximate meximum range from
Thornton and T u t t l e (1960)-(1)*
-- 70.0
16.9
i.0 - 5.0
0.0 - 1.3
.3G3x
1.0 - 3.2
NaO
3.5 - 4.7
2.5 - 6.3
K,O
"
-*differentiatieri h & x 62.5
si0
66.7
12.5
Ai2iI3
"
"
"
The amount of Rb is .qui;e unusual, especially
amount of K 2 0 present.
52, generally
The K/Rb r a t i o s f o r
with t h e moderate
t h e Palisades sill (12 t o
less than 30) a r e extremely low, a Rb enrichmentwhich
musthavebeenproducedfroman
p. 144) notes that
anomalous sourcerock.Taylor(1965,
"norteal"'K/Rb
r a t i o s are typically 150
Rb v a r i a t i o n i s much greater i n t h e €I-profile than
f i l e (Pig. 8 ) .
".
67.7
15.7
2.5
. 0.7
2.6
4.7
3.3
CaO
"
.
ban for Palieedes
s i l l samples (%)
eo 300.
i n t h e V-pro-
The mean and standard deviation for each are given
as
follows:
Rb (ppm)
676
744
230
H-profile
V-.profile
0
-
7
174
.Strontium varies l i t t l e over the sill, from52
t o 75 ppm with a
mean of 58.6 ppm and crof 4.4 (excluding sample8 V23 and V24, which
I
. ..
.
,
..
.
23-
20
are 122 and 108 ppm respectively).
between Sr and CaO (r
$0.37),
A slight positive correlation exists
.as expected for "granitic" rocks
(Turekian and Kulp,1956, p. 2 4 5 ) .
St
The
variationfor the H-profile is greater thsn the
for
V-pzfifile.
mean
and
standard
deviation
for
each
Sr (ppm)
mean
E-profiie
V-profile
59.2
57.6
profile
U
'
5.1
2.5
are
gSven
as
follows
. .
..
. .
PETRCGFJ2VV
All samples.contain phenocrysts
of plagiocleee (An lass than
-
501, quartz, + b i o t i t e , + c h l o r i t e , and +magnetite i n a submicroscspic
tomicroscopic
groundmass of c r y s t a l l i n e m a t e r i a l .
The groundmss
far 0nl.j two eemplas (V19 and V22) is coarse enough so t h a t i n d i v i d u a l
c r y s t a l 2 sz= d i c t i z g u i s h a b l e w i t h a petrographic microscope.
All types of phesxryste i\icluJe t%rdier c r y s t a l s of a l i ' o t h e r
types of phenocrysts except
may beobscured
for the absence of quartz
by b i o t i t e a l t e r a t i o n .
i n b i o t i t e , which
The most conrmOn i n c l u s i o n
recognized is b i o t i t e i n p l a g i o c l a s e ( F i g . 9 ) , s u g g e s t i n g t h a t
clase hasnuclentedaround
suggest that
small b i o t i
o,
plagis-
crystals.Theserelationships
a l l the phenocrysts began to crystalli'ze at about the
same t i m e and at approximatelythe
that magnetite crystallized at
same temperature.
It is assumed
a l l s t a g e s of magma c r y s t e l l i z a t i o n
i n quartz,
because 1) several primary magnetite inclusions are observed
plagioclase, and b i o t i t e p h e n o c r y s t s ,
and 2) m a g n e t i t e c i y s t a l s are
of vazhble eizee i n t h e groundmass.
K e d d e z d y s e e were performedontwenty
thinsections.
i n Table 4 , t h e v a r i a t i o n i n p e r c e n t a g i s of groundmassand
(except quartz) for
kri shown
phenocrysts
a l l samples is smail enough t h a t t h e s t a n d a r d
d e v i a t i o n Fs less t h a n p e r c e n t r e l i a b i l i t y
Der P l a s andTobi(1965).
other phenocrysts (from
as determined from Van
Modal quartzpercentagesvary
1 t o 10 percent) and
made w i t h " s t r a t i g r a p h i c " p o s i t i o n . V a r i a t i o n
r.0
more than
c o r r e l.a t i o n can be
I n percent S i 0 2 i n t h e
..
22
..
Figure 9.
.
Biotiteinclusioninplagioclase.
Crossed n i c o l s ;f i e l d
of view, 2.1 mm w i d e .
..
. J 5
whole rock cannot be.correlated with percent quartz phenocrysts.
Phenocryst percentages have no apparent relationship. t o. whole rock
chemical variations.
.
TABLE 4 .
_..
MODAL DATA FROM TWtW THIN SECTIOFIS
P1,enocrysts
Quartz
-
Plagioclase
Biotite
..
..
. .
6.0
28.9
2.4
2.7
4.0
1.1
2.0
4.0
1.5
.
..
,Of t h e twenty t h i n s e c t i o n s examined {every fourth sample
i n the.V-profile), two c o n t a i n x e n o l i t h s .
H-profile'iindeverythirdsample
A small amphibolitic xenolith approximately 19
conteinshornblendewith
m wide i n sample V19
some magnetit. and plagioclase.
ap?zsr to have reacted with the
xenolith or autolith
in the
magma.
It does
nnt
Sample V16 has a 16 rmn wide
of a s i n g l e q u a r t z crystal containing a euhedrai
brrt highly alterecl feldspar inclusion
and a e u h e d r a l b i o t i t e i n c l u s i o n .
T'ne q u a r t z c r y s t a l c o n t a i n s and is surrounded by s e r i c i t e .
Plagioclase Phenacrpts
are equant and range
Plagioclase phenocrysts
t o .3.0
mm. Most of
thelargercrystalsareeuhedraltosubhedral,
c o n t a i n i n ga l b i t e ,p e r i c l i n e
sectionshave
i n width from 0 . 5
andCarlsbadtwinning.Severalthin
zoned phenocrysts.
from s m a l l b i o t i t e c r y s t a l s
nzay
Some plagioclase apparently nucleated
as demonstrated by numerous b i o t i t e
inclusions (Fig.. 9) .
Plagioclase crystals vary
fromhavingnarrow"cloudy"
d i s t i n c t twinning t o being completely disrupted
rims and
in which no twinning
. . . .. .. .. .
.
..
... . .
.
..
..7,
.?..
;-.
.. i.1
-\
.. ..'
24
L
:
is v i s i b l e and i n t e r f e r e n c e f i g u r e s are i n d i s t i n c t o r n o t o b t a i n a b l e .
Within a s i n g l et h i ns e c t i o n ,
t h e ?+me degree.
a l l plagioclase crystals a r e altered t o ,
No correlationcanbe
a t i o n and samplelocation.
. .
alter-
p a d e between degreeof
.
.
.'
.was
Determination of the,An-content of plagioclase
. . phenocrysts
. .
attempted by two methods, by Michel-Levy's method and by chemical
analyses of drilled out plagioclase phenocrysts.'
tzethod is of no value for Palisades
Michel-Levy's
s i l l samples.
twinned c r y s t a l s were of s u i t a b l e o r i e n t a t i o n
t h e method s t a t i s t i c a l l y v a l i d . P h e n o c r y s t s
Too few a l b i t e -
(less t h a n f i v e ) t o
ofsamples
make
H17 and H25
have An values of '35 and 4 4 , respectively, 'as determined by atomic
absorption analyses.
The amount of K20 in plagioclase phenocrysts
of a relatively,high temperature
of c r y s t a l l i z a t i o n ( T u t t l e
1958, p . 131). 'On a ternary.diagram(system
p l o t on the curve that Tuttle
Ab-Or-An),
and Bowen,
thephenocrysts
.
,
and Bowen (1958, p. 135) d e s c r i b e a s
crystallizing at temperntures for rhyolites
high H20 content(Fig.
is h i g h , i n d i c a t i v e
and phonolites w i t h
R
10).
Quartz Phenocrysts
Quartz crystals
invar:&-Jy
have equantdimensions,
form.
andhaveanhedraltos&hedral
are rounded,
All c r y s t a l s a r e embayed and
rrxnded by r e s c r p t i o n from f o n e r l y e u h e d r a l c r y s t a l s ( P i g s .
vary considerably but predodnantly
and 13).Pheoccrystwidths
in the range
0.2 t o 0.5
mm..Contacts with the
groundmass.
rims o f f i n e c r y s t a l s o f . q u a r t z , a p p a r e n t l y
r e n c t i o n and partialresorption(Pip.13).Crystalshnving
.
..
are
Several.
a result of
no rims
..
.
..
.
.
,. .
.
..
groundmass a r e commonly
very d i s t i n c t and t h e embayments a r e f i l l e d w i t h
sampleshave
il, 12,
.. .
. ..
.
. . .,_
87.
.
.
.,
. ..
i .
25
,
. .
Ternary diagram illustrating solid solution of plagioclase
phenocrysts from sample H17 and €325 in the system An
LIne C
.
Or
Ab
Figure 10.
.. .
- C'
-
All
-
Or.
represents solid solution expected for feldspars crystal-
lizing from rhyolite and phonolites with high water contents
(temperature dependant) .Curve from Tuttle and Bowen (1958, p. 135).
.
*
.....:;>
26
:...'
. ..
Figur'e 1
.
;hly et&aye d quartzphenocryst.Crosse
of view, 2.1
mm wide,
.
..
. .
.
.
d nilccAs; field
..
._.
,
Figure13.Partially.resorbedquartzphenocrysts(largewhitearea
i n lower right andround
grained quartz rims.
white area
i n upper l e f t ) w i t h f i n e -
Crossed n i c o l s ; f i e l d o f
view, 2 . 1 mm w i d e .
31
29
must be a r e s u l t of complete di.ssemination
of fine quartz crystals
of Si02 in the
magma af.ter resorption.
. . diamond. .
o r square- shaped form o f many quar.tz phenocrysts
The
i n d i c a t e st h a tt h e yc r y s t a l l i z e d
as beta-quartz(highquartz).
is prednminantly hexzgond dipyr=!Fdel
habit of beta-quartz
,
Thc
e ~ d
prism
. .
are predominant and subinedrai o r
f a c e s are subordinate.Prismfaces
euhedral crystals are elongate if the quartz crystallizes
as alpha-
q u a r t z (low quartz).
Biotite,Chlorite,
Biotite, chlorite,
and MagnetitePhenocrysts
and magnetite phenocrysts are discussed togeth-
er hscaiise t k t r genes:r:
beingalterationproducts
mxi be r e l a t e d , ' w i t h c h l o r i t e
of b i o t € t e .
and mngnctlte-
Not all magnetite i s r e l a t e d
- ' t o b i o t i t e and chlorite: therefore, the following discussion
will begin
..
. . .
. . .
w i t h m a g n e t i t e . n o t r e s u l t i n g from a l t e r a t i o n .
Amaunt, s i z e , and form of magnetite phenocrysts vary greatly.
Crystals appear subhedral
and anhedral, generally ranging
from less than 0.01 t o 0.75 m.
Larger crystals,
i n diameter
up t o 2.0 m i n d i a -
meter, are found i n s e v e r a l samples.
Biotite phenocrysts show r e a c t i o n r e l a t i o n s h i p s s u c h t h a t
biotite has altered.to olive green biotite,
of various shades of green,
and magnetite
opaque o x y b i o t i t e ; c h l . o r % t e
+ sericite +
leucoxena.
Both g r e e n b i o t i t e and chlorite appear to be'alteration products
brown b i o t i t e because brown b i o t i t e i s seen a t t h e center of
g r e e n b i o t i t e and chlorite phenocrysts (Fig.
brown
Of
some
14), never the opposite.
Magnetite and s e r i c i t e a r e more common around b i o t i t e rims t h a n w i t h i n
thecrystals(Fig.
15).
When s e r i c i t e is presentwithin
a biotite
c r y s t a l , . m a g n e t i t e is within or adjacent
. to. 'the sericite (Fig. 16).
.. .
. ..
..
.
,
.
32-
30
.
Figure 1 4 .
.
Brown b i o t i t e surrounded b y . g r e e nb i o t i t e .
netite.. .Uncro.ssed n i c o l s ; f i e l d of view, 2.1 mm *de:
i
Black i s mag-
31
..
.
32
. .
---
S e r i c i t e h a s a creamy yellow color due to submicroscopic leucoxene.
All d e g r e e s o f b i o t i t e a l t e r a t i o n
exist within the s i l l and com-
.. Alteration generally disrupts the
monly w i t h i n s i n g l e t h i n s e c t i o n s .
crystal structure such that the different varieties of biotite
and
c h l o r i t e aze i n d i s e i n g u i s h a b l e , e s p e c i a l l y g r e e n b i o t i t e v e r s u s c h l o r i t e .
Separate phenocrysts within one thin section
brown b i o t i t e t o b i o t i t e
15).
leucoxene(Fig.
may range from normal
pseudomorphs of s e r i c i t e
+ magnetite
According t o Schwartz(1958,p.176),
(i.e.
equilibrium alteration of biotite
f
t h i s non-
,. varying degrees of alteration)
is notunusual.
Schwartz (1958), in'agreement with Winchell
p. 376), suggeststhefollowingsequence
'
.
'
as being typical for biotite
alteration,
consistent
with
observations
presented
i n c i p i e n t s t a g e o f a l t e r a t ...
ion,
and Winchell (1951,
above.
I nt h e
brown b i o t i t e r e c r y s t a l l i z e s w i t h
a
change i n c o l o r t o g r e e n b i o t i t e ( v e r y l i k e l y d e u t e r i c ) . N e x t , b i o t i t e
c o n v e r t st oc h l o r i t e .
releaseof
Alterationtosericite
Ti.
a t any time.
Very f i n e leucoxene c r y s t a l l i z e s from t h e e a r l y
Inthinsectioa,
a t i o n ofprimary
and magnetitecouldhaveoccurred
i t is indetermineblewhetherthealter-
biotite to green biotite
and c h l o r i t e p r e c e d e s ,
or is syngenetic with the ~ ~ ~ P - T Z ~tc
L CmFz .g n e t i t e , s e r i c i t e ,
.
leucoxene.
The formsof
follows,
and
.
b i o t i t e and i t s pseodomorphs a r e g e n e r a l l y e u h e d r a l
and subhedrelsix-sidedcrystals
of t a b u l a r h a b i t .
The widedimension
is' t y p i c a l l y 0.2 t o 1.0 mm across.
Groundmass
The groundmass i s composed of plagioclase, K-feldspar, quartz,
.+biotite,
and +magnetite (feldspars distinguished by staining tech"
.F q
._....._.
..
niques). Finely crystalline magnetite
34
and l i t t l e o r no b i o t i t e are
v i s i b l e i n the relatively coarse groundmass.of samples
'
'V19 and V23.
Rough c a l c u l a t i o n s of An for p l a g i o c l a s e ' i n the groundmassof
samples H17 and HZ5 were made using modal d a t a and chemical data from
whole rock and phenocryst analyses..
.Groundmass plagioclase f o r
sampie H17 has hn near 0 ; for sample H25, approximately 18.
Dissolution Voids
Voids are found i n all t h i n s e c t i o n s .
percent of the rock
They occupy l e s s t h s n
volume (except for sample
2
V23 which is approxi-
mately 4 percant ~ o i d j . It is.obvious from thinsectionexamination
thhct these voids are caused by d i s s o l u t i o n of c r y s t a l l i n e p h e n o c r y s t s
andgroundmass
2nd are notsimplevesicles(Figs.
Otherwise,thevoids
would befoundonly
1 7 , 1s. and 19).
i n t h e groundmass.
Percen-
tage and d i s t r i b u t i o n of these voids, which.will'be referred to as
dissolution voids, are.inconsistent
2nd no correlation can be
made
t o sample l o c a t i o n .
DissGhtion voide are i n groundmassand
exceptquartz.
phenocrysts;
in a l l types of phenocrysts
Only sample V23 containsdissolutionvoids
In most t h i n s e c t i o n s , v o i d s a r e
more common i n p l a g i o -
clese enZ.biotite.phenocry6tsthan in the groundmass.
material adjacent to voids does not appear to
i n quartz
Crystalline
b e a l t e r e d or disrupted.
Samples V23 and V24 are unusual for t h r e e r e a s o n s . F i r s t ,
d i s s o l u t i o n v o i d s are most abundant i n t h e s e two samples.
Secod,
very l i t t l e o r no b i o t i t e or magnetite is v i s i b l e , and it is unclear
whether these minerals
or whetherthe
were leached out during formation of the voids
samples never contained them p r i o r t o l e a c h i n g .
t h a edgecl of some disoolution voids.
o very irregular
Along
opaque " i r o n
37
... i
35
'
.
..
.
I
1
Figure 17. Dissolution void (black) i n a plagioclase phenocryst and
.
groundmass.
Crossed nicols; field of view; 2..l ma wide.
.
.-.
1 . :,..1
36
. .
. .
.
.
..
Figure 18.
.
.
Dissolution voids (largerblackareas)
phenocryst,'a biotite phenocryst,
andgroundmass:
i n a plagioclase
Crossed n i c o l s ;
f i e l d of view, 2.1 uan wide.
.37
'..
~
'. i
37
<
Figure 19.
Dissolutionvoid(black)
Crossed n i c o l s ; f i e l d
i n coreofplagioclasephenocryst.
of view, 2 . 1 w wide.
,:?I
..
:"
38
oxide" is seen, p o s s i b l y r e s u l t i n g from r e p r e c i p i t a t i o n o f l e a c h e d
iron.
Third, some of t h e d i s s o l u t i o n v o i d s are p a r t i a l l y f i l l e d
w i t h q u a r t z which. grew r a d i .a l l .y from the gutside toward t h e c e n t e r ,
l e a v i n g a void i n the middle.
Under a petrographicmicroscope w i t h .
crossed nicols, these radial growths
around the void (Fig.
produce an "isogyre" effect
2 0 ) , a r e s u l t of r a d i a l o r i e n t a t i o n o f q u a r t z ' s
c-crystallographic.axis.
-7..
:..::j
'
39
I
Figure 20.
. causing
growth).
. .
Dissolution voids filled with radially orientedquartz
the "isogyre"effect (round voids at center of quartz
Crossed nicols; field of view, 2.1 nun wide.
.
.
CRYSTALLIZATION AND ALTERATION
Chemicaland
ir?csr?jwction w:th
petrologic information used
experimental data are.used to develop
a c&eren_t 2nd consister.t
c r y s t a l l i z a t i o n and a l t e r a t i o n model f o r t h e P a l i s a d e s ' s i l l .
observations presented above a r e t h e b a s i s f o r t h i s
The
model.
Intrusion
R e l i i t i ~ nbetween Magma and Phenocrysts . .
. ....
Quartz, b i o t i t e , and plagioclase phenocrysts.apparently
..
crystal-
.
l i z e d from t h e magma i n a deep chamber b e f o r ei n t r u s i o nt ot h ep r e s e n t
p o s i t i o n of the s i l l .
Modal data(Table
4 ) ir?dicatethatthe
eEorrnt
of b i o t i t e and plagioclase phenocrysts for different parts of the
magna
chamber was constant.
The percentage of quartz phenocrysts
ail or' t h e magma before embayment.
was probably constant for
Inthinsection,10to
enbbayec! PES - r i l l
modal q u a r t z is found when the phenocrysts are least
d i s p l a yc r y e t df a c e s .
phesocrysts contain
Samples withextremely
11 percent
embayedand
small quartz-
less than 6 percent modal quartz phenocrysts.
Intermediate degrees of
embayment and intermediate quantities of
quartz also exist. Before intrusion,
modal
a l l p a r t s of t h e magma probably
contained 10 to'12 percent euhedral quartz crystals.
C r y s t a l s e t t l i n g and floating can be ruled out, either before
o r after intrusion,, based
on phenocryst distribution, chemical data,
andcomparisonwithexperimental
and theoretical data. Table
that distribution of phenocrysts throughout
4 shows
t h e s i l l is c o n s t a n t ,
.
.
..
93 ..
'.
1
41
i n d i c a t i n g that crystal s e t t l i n g and f l o a t i n g d i d n o t o c c u r d u r i n g
c r y s t a l l i z a t i o n of phenocrystsand/or
As a result,
groundmass.
e q u i l i b r i u m c r y s t a l l i z a t i o n is t h e dominantprocess,.
No r o r r e l a z i o n
= 5.2) 2nd p o s i t i o n i n t h e sill;
canbe drawn between n s g n e t i t e (
therefore, crystal s e t t l i n g f o r o t h e r minerals would be very unlikely
(Shasr, 1965, p. 128).
For magma as viscous 'as t h e P a l i s a d e s s i l l
magma was during intrusion (approaching the system
(1P53> has demonstratedthatphenocrysts
E:r:.l=tt
in t h e
sill
codd not have settled.,
Ab-0r-Si02-H20),
as small as t h o s e
They A u l d f o l l o w , c ~ n v e c l ; i v e o r
forced r'low without "slippage" of the crystals in the fluid.
.
.
No r e a s o n s e x i s t t o s u s p
..e c. t.
i n a t i o n from either country rock
of xenoliths, apparent constant
andgroundmaes,and
any type of metasomatism or contam-
or mixing with other
magmas.
Absence
abundance and composition of phenocrysts
p r e d i c t a b i l i t y ofsamplechemistry
are evidence
f o r a . s i m p l e model i n v o l v i n g e q u i l i b r i u m c r y s t a l l i z a t i o n .
"r
5 ~ r r c f sof Pressure Change during Intrusion
'
The m i n i m u m d e p t h o f i n t r u s i o n
estimated from maps .=dc r o s s
sections of Robinson and o t h e r s (1964) is probably 1,5000.to 3,000 m.
The approximate l i t h e s t a t i c p r e s s u r e f o r t h e
900 bars, assuming a densit? of
(Nash2nd
Wilkinson,1970,
substantial pressure drop
of phenocrysts
l a t t e r depth would b e
2.3 gm/cc for overlying xztsrial
p. 256).
The magma c l e a r l y underwent
a
. .
. ,
on i n t r u s i o n , and p r e s s u r e s e n s i t i v e r e a c t i o n s
and magma are l i k e l y t o have occurred.
P a r t i a l r e s o r p t i o n of quartz
ing pressure, a n e f f e c t n o t
i s t h e meet obvious effect of lower-
uncommon involcanicrocks.Resorption
of q u a r t z w i t h pressure decrease is dependent on water.content
magma.
If the magma is dry o r less than 150 s a t u r a t e d , t h e
oE the
phase
'boundary of a PT diagram for quartz has
a positive slope (Boyd and
at a constant
England, 1960,-p.752) so that a drop in total pressure
temperature could.cause quartzto resorb (Fig. 21).
If the magma
1s water saturated,a drop in pressure will enhance crystallization.
Fyfe .(1970) suggests that most "gr.enitic" magmas are water-undersaturated, and it is generally accepted that more mafic igneous rocks
..
are very dry.
Other reasons to suspect that the Palisades sill magma
was water-undersaturated are as follows. First, P
H20
is buffered by
reactions such as
.
.
KFe2-3(AlSi3010) (OH) 2(biotite) $
KAlSi308(K-feldspar)
..
+ Fe304(magnetite)' + H
P
'
.
(Fyfe. 1970, p. 205)
and
2KFe2_3(AlSi~O10)(OH) Z(hiotite)
K$14(OH)4Si6A12020(~ericite)
+ Z N F 3 + 1120 %
+ Fe304(magnetite).
The mineralogic relationships for the latter reaction are observed in
the Palisades sill, Second, i f PEz0 Ls high it will lower the phase
so that it will near
the K-feldspar phase boundary
field of quartz
1970).
(Eggler, 1974, and Piwinskii and Wyllie,
Since no K-feldspar
phenocrysts are foundin the sill, it is most likely thatPH was not
2
equal to Ptotal so that theqsrtz phase boundary was aathigher
ragza nzst
temperature relative to the K-feldspar boundary. Third, the
have.been H20 undersaturated before intrusion. Otherwise, the magma
would have reached saturation, "second boiling",
on intrusion and
quartz would not have been resorbed (Burnham and Jahns, 1962).
of quartz. An
One other possibility exists for the resorption
such
increase inPH 0 could'lower the quartz melting temperature that
2
quartz phenocrysts could resorb, but there
is no renson to suspect
:4
-. .
...
h3
1
1
+saturated melting boundary
for
Figure 21.
Schematic pressure-temperature.diagram showing p o s s i b l e
quartz 'crystallization
- resorption history in the Palisades
s s s a d n gi n s t a n t a n e o u si n t r u s i o n
and Pg
2
less than P,,,_,
Brown, 1970, p. 356, and Harris and'others,1970,
A
"
.
p. 191).
l i n e is a quartzmelting boundary f o r a constant PH
is less than Ptotal.
Field
Point
Field
Point
Point
A
B
C
D
X
Flold F
Polnt C
-- ql iuqauritdz b e g i n s t o c r y s t a l l i z e
- quartz crqrstallizea
- i n t r u s i o n of ma!:ma
- Quartz begins t o melt
- quartz resorb6
- quartz crystalllzntian
resumes
2
sill
(after
Dashed
where PH
2
.
such a change.
.,j
44
Ifwater
would haveabsorbed
were added, t h e H20 buf€ersdescribedabove
It is ,unlikely that added water would
much of it:
be homoganously mixed throughout the
since quartz phenocrysts in all
s i l l . a s it would have had t o be
samples show resorption.
Different degrees of quartz resorption arerelated to silica
mobility.. Least affected cryetsls
fine grained quartz, clearly
13) without effective
have a narrow rim of extrezely
a r e s u l t of reaction 2nd resorption (Fig.
mixing of resorbedSi02
and magma.
The most
111, i n d i c a t i n g t h a t t h e s i l i c a
embayed crystals never
have a r i m (Fig.
was more mobile.That
i s , more SiOz was a b l e t o r e a c t
and d i s p e r s e i n t o
t h e l i q u i d due t o l o c a l v a r i a t i o n s i n v i s c o s i t y .
It i s - u n c e r t a i n whether plagioclase phenocrysts
by th,e pressuredroponifitruaion.
edges are s l i g h t l y "corroded."This
They 2re genernllysubhedrai
and
s l i g h t r e s o r p t i o n phenomenon- i's
possibly due to the pressure.drop with plagioclase
quartz because of
were a l s o a f f e c t e d
i t s bigher temperature
less a f f e c t e d t h a n
and p r e s s u r e s t a b i l i t y f i e l d .
Because b i o t i t e is-a hydrous mineral,
i t s phase boundary has the
opposite slope from t h a t of plagioclase 2nd q u a r t z ; . t h e r e f o r e , b i o t i t e
should be more s t a b l e upon lowering a€ to.tal pressure.
C h i l l Zone, Magma Convection, and Temperature D i s t r i b u t i o n
It i s notclearwhetherthePalisades
d e f i n i t i v ee v i d e n c e
i s a v a i i a b l e ,t e x t u r a i i y
groundmass i s s u f f i c i e n t l y f i n e g r a i n e d t h a t
significantly finer..
s i l l has a c h i l l zone.
o r chemicaily.
A
i
No
'
a c h i l l zone may not be
Chemically, t h e s i l l is homogeneous enough such
t h a t a c h i l l zone can not be distinguished.
A possiblechill
fromwhich
zone is found a t t h e lower contact.
Of samples
t h i n s e c t i o n s were made, the c l o s e s t sample t o the lower
contact in the H-profile,
111, and t h e c l o s e s t
two samples i n t h e V-
p r o f i l e , V 1 and V4, have t h e f i n e s t groundmass.
are less than 1.0
m from the contact;
m from the contact.
H1.
Samples H 1 and V 1
sample V4, approximately 30.5
Another observation was made withregardtosample
The t h i n s e c t i o n
for t h i s samplecontainsseveralkinkedchlorite
crystals, possibly indicative
of a s o l i d i f y i n g groundmass during
forceful intrusion.
Samples a t t h e upper contact
showno
evidence of being chilled,
but textural features indicate that the highest
30 t o 90 m of t h e
th.Ls is the coarse groundma,es i n earnple V23, 4 coarserthanaverage
groundmass i n V19, and theconcentration
voids in their vicinity..
and nature o € d i s s o l u t i o n
The coarser groundmass ofsamples
.
V23 and
V19 is i n t e r p r e t e d a s a r e s u l t of higher fluid content during crystallization.
It is presumed that watercontent
would increase i n t h e
last s t a g e s of c r y s t a l l i z a t i o n and would m i g r a t e t o t h e h i g h e s t l e v e l
.. .
of magma remainiag i n t h e sill. I f t h e m a t e r i a l
abovesample V23 were
crys-talline, fluid
causethe
would c o l l e c t i n t h e v i c i n i t y o f
developmentof
l a r g e rc r y s t a l s .
Sample V19 probablycrystal-
l i z e d a t a l a t e r time a f t e r t h e samplesabove
crystallization, again creating
Dissolution voids in samples
a capunder
V23 and thus
had completed
which f l u i d couldaccumulate.
V23 and V24 a r e numercus, e s p e c i a l l y
V23, i n which dissolution voids occupy 3 t o 4 percent of the rock.
This may be taken
as a result of concentration of the dissolving
v o l a t i l e s below a w e l l c r y s t a l l i z e d and cooler capping material.
The “corrosive” volatiles apparently concentrated near
samples V23
and V24 and became o v e r s a t u r a t e d t o t h e p o i n t t h a t q u a r t z r e - p r e c i p i -
x
.:. . I
46
t a t e d i n some of t h e d i s s o l u t i o n v o i d s .
Magma.
convection is a prcicess which probably.occurred after intru-
s i o n and p r i o r t o c r y s t a l l i z a t i o n , k e e p i n g . t h e t e n p e r a t u r e g r a d i e n t t o
a mininm.
Convection alsoconfusesinterpretatio?.
of chemicol ond
Bartlett (1969, p. 1069-1071)
mineralogicdistributiononintrusion.
has demonstrated'thst z granitic ~ + p ~ is
a expzcted t o have n a t u r a l
convection for a s i l l the thickness
The v i s c c s i t y o f t h e P a l i s a d e s
of the Palisades
s i l l (Fig. 22).
s i l l on intrusion should be rorrghly
;with magmas containing as much as 50 percent phenocrysts.
An order-of-magcitudeapproximation
f o r t h e cryatallizati.cn
time f o r t h e - P a l i s a d e s s i l l is 600 t o 1400ye2.r~ (Tsraer, 1968, p . 20;
Carmichael,1974,
p. 446).Although
t h e rate ofconvection
unkco-m,' i t is c e r t a i n l y p o s s i b l e f o r t h e
numerous times.
Individualconvection
is
magma to have overturned
cells are generallyhexagonal
i n f c x with width approximately equal to height (thickness
oE the
s i l l ) i n which t h e magma flows up t h e center. o f t h e c e l l and down
t h es i d e s
(Bartlett, 1969, p. 1069).
Convective overturn kept temperature differences
r e l a t i v e l y small.
by cooling of the
If thetemperaturegradient
in t h e c e l l
becomes l a r g e e i t h e r
magma through the ceiling or through both the
c e i l i n g and t h e f l o o r , d e n s i t y i n v e r s i o n i n p a r t o f t h e
magma w i l l
cause convective overturn, decreasing the temperature gradient
(Bartlett, 1969).
Convectiontendstopromoteequilibriumcrystallization.
If no
47
heat fluxes
.. .
y a p p r o x : range f o r
Palisodes
CHAMBER HEIGHT
Figure 22.
sill magma
- meters
S t a b i l i t y range of the Palisades
s i l l magma a f t e r intru-
s i o n based on magma convection s t a b i l i t y limits from B a r t l e t t .
(1969, p. 1071)'.
convectionoccurred,
two factorsshouldbeobservable.First,textures
r e s u l t i n g from different lengths of
.time f o r c r y s t a l l i z a t i o n s h o u l d
be Systematic i n r e l a t i o n t o " s t r a t i g r a p h i c " p o s i t i o n
Variations in crystal size of t h e groundmassand
tion voids should
showsome
abundance o f dissolu-
systematic pattern from.top doh, neither
of whichcanbedemonstrated.
variations should be
i n the sill.
Second, chemicalandminerelogical
more systematic i n r e l a t i o n t o " s t r a t i g r s p h i c "
position as a result of fractional crystallization;
a process which
would'be more l i k e l y i f t h ec r y s t e l l i z e dw i t h
a largetemperature
gradient. There is-no evidence that in s i t u
fractiqnal
arystallization
"
and its subsequent elemental differentiation occurred either
top down,, from t h e bottom.up, or toward the center;
significant variation but not
from t h e
Oxides do show
i n a "stratigraphic" pattern.
Deuteric Alteration
Two possibletypesofdeutericorlate
magmatic a l t e r a t i o n exist.
One i s t h e a l t e r a t i o n of b i o t i t e and plagioclase phenocrysts.
o t h e r is t h e d i s s o l u t i o n of groundmassand
The
phenocrysts.
Phenocryst Alreraeicn
It'is' unclear howmuch
.is deuteric.
of t h e b i o t i t e and p l a g i o c l a s e a l t e r a t i o n
Some of t h e b i o t i t e r e a c t i o n r e l a t i o n s h i p s
clase p h e n o c r p t a ~ l o ~ d i ~ g disrrrption
.&d
cf cr;ztallinity
todeutericalteration.Plagioclasealteration
and plagio-
may be due
c c u l d becaused
by
pressurechangesduringintrusion,
by s l i g h t 'pH & i a t i o n s i n t h e
2
magma, o r by deuteric reactions which cannot be defined optically.
Many a u t h o r s c o n s i d e r t h e r e a c t i o n s , b i o t i t e t o c h l o r i t e
and plagio-
clase .to scricite, deuteric alteration.
Since biotite. has been a l t e r e d t o a . l a r z e d e g r e e , t h e a u t h o r
..
m
j
..
49
..
considers only biotite to chlorite
and brown b i o t i t e t o g r e e n b i o t i t e
It hasbeendemonstrated
as'deutericeffects.
that the biotite t o
s e r i c i t e 4- magnetite reaction may be H20buffered and ,was.probably a
c r p t a l l i z a t i o u of b i o t i t e phe%cTstq;.
continuousprocessafter
..
Phenocryst and Groundmass Dissolution
Dissolution to prodiice voids is a d e u t e r i c p r o c e s s s i n c e t h e .
the source of the corrosive fluid
groundmss was c r y s t a l l i n e , a i n c e
was probably i n t e r n a l , and since t h e minerals were probably a t nearliqtriduatemperature
di~;;>;:%cz
when dissolved.
n f cryeta&
Only two references of
; z . i g x r x z rockscouldbefound.
Matthews (1969) reported quartz leaching
S s l o t t i and
from wa'll zone graphic
g r a n i t e in a zoned pegmatite.Barkerand
Burmester (3970) r e p o r t a d
..
complete removal of q u a r t z and a l b i t e w i t h decomposition of
.,
,leaving only microcline and.a red iron
hypabyssalrhyoliteporphyry.
biotite,
s t a i n i n two outcrops of
a
They a t t r i b u t e t h i s l e a c h i n g t o c o n t a c t
of the sanqles with a h i g h l y a l k a l i n e b r i n e
(pH greater than 9) long
deer crystallization.
Dissolution probably occurred
removal of
medium.
a t near-solidus temperature with
m a t e r i a l i n a low v i s c o s i t y and highly corrosive 112 0 r i c h
was necessary so t h a t the minerals
Near-solidustemperature
dissolved were r e l a t i v e l y n e a r t h e i r i n s t a b i l i t y f i e l d s
and t h e
concentration and amount of the corrosive constituent
was not high.
Experimental r e s u l t s o f W y l l i e
and T u t t l e (1961and
1964) on t h e
e f f e c t s of SO3, P205, HC1, Li20, MI3, and HF in a d d i t i o n t o H 2 0 on
melting temperatures of granite demonstrate that
most likely corros:ve
V O h t i h 5
HF and L i 2 0 a r e t h e
t h a t produced d i s s o l u t i o n of plagio-
clase in preference t o quartz, the relationship observed
i n tho
..
..
Palisades sill.
..:..I
Thesestudies
stabilityfieldofplagioclase
50
show t h a t HF and L i 2 0 i n H20 lowerthe
Wyllie
relative t o t h e q u a r t z f i e l d .
and T u t t l e (1961, p. 141) state, "The feldspar is less s t a b l e i n ' t h e
presence of
HF solutions than
i n the presence of €120, as would b e
expected, and t h e q u a r t z a p p e a r s t o b e
more stable."Althoughthese
of t h e f e l d s p a r s t a b i l i t y
studies involve melting of minerals, lowering
f i e l d relative to t h e q u a r t z f i e l d
may be the explanation
clase i s more s u s c e p t i b l e t o d i s s c l u t i o n t h a n q u a r t z . P u r e
why plagioIi 0 would
2
not be responsible for lowering the plagioclase stability field
to the quartz field
RS
relative
is shown from phase dhgrams of "granitic" rocks
( T u t t l e and Bowen, 1958; Piwinskii, 1968).
T h e amount of HF o r L i i O required tb' lower s i g n i f i c a n t l y t h e
s t a b i l i t y f i e l d s of p l a g i o c l a s e relative to quartz is not p r o h i b i t i v e l y
high.
According toWyllie
and T u t t l e (1964,
p. 9 3 7 ) ,
temperature of beginning of melting only requires
t o lower t h e
"a few t e n t h s o f
1'
(of HF o r Li20) i f a smaller proportion of t o t a l v o l a t i l e
weight percent
components is employed".thantheyused
.
i n experimentation.
Comparison with
Experimeatal
Results
Figure 23 i s a p,ressure-temperattire diagram
of melting relations
i n n a t u r a l r o c k s (PH 0 = Ptotal ) t h a t shouldbe a best approximation
2
f o r t h e P a l i s a d e s s i l l basedon mean oxide values, phenocryst mineralo g y , and .experimental data
Wyllie (1970).
Figure 23 i s .a.synthesisof
PT p r o j e c t i o n s of phase
boundaries for eight igneous rocks including granite
( 3 ) , q u a r t z monzonite(2),and
. .
from Piwinskii (1968)'and Piwinskii and
(21,
granodiorite
t o n a l i t e (1). Sincethechemistry
mineralogy of none of these samples can be directly correlated with
P a l i s a d e s sill samples, the following reasoning.has been used to
and
51
,
I!
\
I I I
\
I
6 00
700
T
.Figure 23.
Estimatedphaseboundaries
sill assuming Pito.
2
Wyllie, 1970).
Ptoral(after
8 00
900
(OC)
predicted for the Palisades
Piwinskli, 1.968: Piwinskii and
c o n s t r u c t t h e diagram: 1) The plagioclase phzse boundary i s basedon
rocksnotcontaininghornblende.
plagioclase phase houndary
pressurm.
boundary.2)
When hornblendeis:present,the
is approximately 100' C higher a t comparable
Whole rock CaO:NaZO r a t i o s have l i t t i e a f f e c t on the
The b i o t i t e phasehoundary
g r z n i t i c ssmplethatcontains
studies have cxtrenely
is based on theonly
no hornblende.Granites
low Fe203* and B O , andwhen
pfesei-it h ~ t h e rsa=Fles, the temperature
non-
used i n t h e i r
hornblende is
o f the biotite phase
boundary
is r e l a t i v e l y h i g h .
The experimental work .demonstrated above (Fig. 23) agrees w e l l
.
with the crystallization sequence determined 'petrographically for the
A t pressuresabove
Palisades s i l l .
t oc r y s t a l l i z e ,f o l l o w e d
1.5 Kb, b i o t i t e i s t h e f i r s t m i n e r a l
by plagioclase.andquartz.Intrilsion
o f the
s i l l occurred before the temperatu.re reached the K-feldspar phase
boundary.The
. 5.5
K
m
, but
are basedon
pressurerequiredneedsto
beabove
no upper limit of pressure con be mode.
PH
2
-
Ptotal,
1.5 Kb, deeperthan
Theseassumptions
and althoughtheyarenotstrictlycorrect,
are probably reasonable estimates.
'
System Q-Ab-An-Or-H20
(saturated) 2 s described by Winkler (1974)
and Winkler and o t h e r s (1975) also demonstrates that the Palisades
magma u n d e w s n t e q u i l i b r i u m c r y s t a l l i z a t i o n
same mineralogicsequenceasdescribed
and c r y s t a l l i z a t i o n
above.
sill
i1.i
Figure 24 is a p l o t o f
two samplesusingwhole-rocknormatfvedata.
These samples were
chosen torepresentextremesincomposition.
Most othersamplesplot
i n o r nearthespacebetweenthese
position of these points with rexpect to
ropresontthecomposition
two points.
C k
As shown from the
phase f i e l d s (assumi.nR thev
of themelt).plogioctaso
LH the f f m t m l n -
.
.I
. ,':.
.:'
. .. .J
,
53
era1 expected to crystallize. As plagioclase crystallizes and teiaperCoriposition of the magma will move away tlfrom
lc
ature lovers, the
point along a nearly straight line which intersects that and
point
a point on the Ab-An edge which is determined by the composition
of the
plagioclase crystals. When the~ s g s aco!!!p:osition
reaches the surface
E1-E2-E3, quartz wili begin to crystallize. Magma composition wiil
move along or near the plagioclase-quartz boundary toward the cotectic
line, P-E5, at which temperature K-feldspar begins to crystallize.
of crystallization at point
E is approximately
Since the temperature
5
50' C higher than at point
P (Winkler and others,.1975; p. 267), the
P until the
liquid composition will move along the cotectic line-toward
remaining magma is crystallized.
System Ab-An-Q-t120 i
3 useful i n demonstrating equilibrium crystallization of plagioclase. Figure 25 gives the plot in the system Ab-AnA s stated previously,
Q-H20 (saturated) of 20 samples using modal data.
plagioclase phenocrysts of samples €117 HZ5
andhave an An-cont.ent of.
approrimetely 35 2nd 4 4 , respectively; 'An-content of plagioclase in
0 for H17 and 18 €or H25.
the groundmass, nearly
Whole-rock CaO
composition is low for sample
H17 and is high for sample H25 compared
wi-h all other samples. These data are consistent with equilibrium
crystzllizction of plagioclase as shown.in Figure 25.
. .'
.. .)
..._
.~
::
..
54
A
. .n
..
A
Figure 24.
An
Plot of samples H13 and H23 using nonnative date on system
- Ab - O r - Q
) (Winkler and others,1975,
1
p . 246).
...:.:.
.... ,
1
.....
!
An
Figure 25. Relationships of sample whole rock composition
(') (from
normative
sition
in
data)
the
others, 1975).
with
approximate
system
An-Ab-Q
Small x
-
(PH20
- whole
H23 (see Fig. 24). Dotted line
phenocryst
and.
compogroundmass
Ptotal) (after Winklerand
rock compositionof samples 1113 and
- approximate
.
magma compositiona s
.
plagioclase and quartz phenocrysts crystallized for sample
H13 and €123.
..
.
.
. .
:
. .,..
.i ..I
MODE OF EMPMCEMEXT
A model
for the mode of intrusion, emplacement, and possible
convective history before final crystallization cannot be precisely
defined. Chemical data are far more complete and accurate
as well as
'show more variation than does petrographic information; therefore,
To make such a modal, the
they willbe the basis for the model.
topographic natureof the collection profilesfor the two profiles
is compared with chemical data both between the two profiles and
within the V-profile.
4 , 5, and's demonstrate that
Table 1 and comparison of Figures
no "stratigraphic" correlation
of chemistry
canbe
made
between
the
two profiles, ruling out a strictly horizontally layered intrusive
model. Furthermore, V-profile, which has a limited areal extent
(less than 450 m) shows
less oxiJe vatlation than if-profile,
w1~il.h
has an areal extentof approximately 2,000 m (Table 1).
The magma
must have intruded in isochemical and isomineralogic "fronts" (isopleths) that were apparently altered and mixed by convectLon before
complete crystallization. Figure 26 shows the cyclic chemical pattern
CaO and Rb. Several
for H-profile using two highly variable elements,
large
scale.and
corresponding
cycles
r both
are f oshown
sleiitents.
Figure 27 is'the author's conception of the intrusion and.chemica1
distribution of the sill before convection.
The isopleths couldbe
a result of continuous cl1onp.e in the chemistry the
of incom.Lnp, rnngm:,or
Figure 26.
Cyclic CaO and Rb variation vs sample location i n the H-profile.
Sample location projected
contacts.
to a vertical plane containing dip of
sill's
.'
3
: .,.. .
57
"_
. .....:
,i
:'
F l p r e 27.
Schematic cross.section of the'palisades sill showins
cl.cment distrthution prior to convection.
c,Iwmical and
isomineralogic "fronts".
Dashed 1.incs - {.so-
5R
.. .,
'...
..I
.:
.
"
.. .'.j
........
. ..
59
a r e s u l t . of "pulses" of differentchemicalcomposition.
ed,previouslyintruded
magma was-pushed i n ' f r o n t .
As magma i n t r u d -
The exact
o r i e n t a t i o n and d i r e c t i o n of movement of the isopleths cannot be
determined.
During the time i n t e r v a l when magma was beingtapped
chamber, the following
from t h e
two p o s s i b i l i t i e s c a n e x p l a i n t h e c y c l i c
compo-
s i t i o n a l changes:
1 ) The variation could be
result of
caused by d i f f e r e n t i a l i o n i c d i f f u s i o n a s
a temperature gradient in the top of the
the differentiated part
magma chamber with
of the magma being removed i n "pulses" repre:
sented by a completecycle.Although
no experimentalevidence
a v a i l a b l e , i t may be reasonable..to postulate that
Rb,Na,
most variable elements i n c o n c e n t r a t i o n ) d i f f u s e d i n t h e
t o a temperaturegradientnearthetopofthe
is
and C a ( t h e
magma r e l a t i v e
magma chamber.
berger (1975) demonstrates t h a t a s i n g l e magma body have
can
~
a
Eichelcoexisting
f e l s i c and intermediate composition.segments with
a nixedzonebetween.
Since Na and Rb c o n c e n t r a t i o n s a r e t h e a n t i t h e s i s
of Ca concentration
i n samples from t h e P a l i s a d e s ' s i l l , N a and Rb may have been enriched
a higher l e v e l than Ca o r , v i c e v e r s a .
Each compositionalcycle
i n F i g u r e 26 represents one pulse i n which both Na
and Ca enriched magma, both near the top
(Fig. 28).
A periodof
shown
Kb enriched magna
of the chamber,were
removed
time must have elapsedbeforethenextpulse
of magma vas intruded, during
'
+
at
which diffusion again produced a
concentration
gradient.
2) Another explanation involves partial
from a suhductjng plate
which moved a t
bcinp, continually removed and intruded.
m e l t f n g at a constant depth
an
i n c o n s i s t c n t r a t e w i t h mwma
Lipman and o t h e r s (1972)have
L73
hn
"feeder
dike
8 R b enriched
""""
Ca enriched magma
boundary o f
magmaremoved in
one
II
puIse"
magma
I
\
Figure 28.
Schematic dfagram demonstrating possible segregation fn
magma chambcr immediately before intrusion of one pulse.
.-.
:.. :,2
..'..)
:
..:.
.,...
61
proposed subduction of an imbricate zone during early and middle
Cenozoic giving rise to predominantly intermediate composition volcanic
sill m y be due to'this magmatic
and intrusive roc,ks. The Palisades
activity since the postulated subduction zone correlates both temporally and spatially (Fig.29).
Additional evidence for the Palisades
sill being related to the subduction zone is that the magma source
km approximated from Figure'29 is close to the depth
depth of 280
K20 values for the
calculated using cheaicel data. Mean SiOz and
for
Palisades sill in connection with data of Lipman and(1972)
others
the nearby Spanish Peaks igneous center
is used to calculate the
250 km. If the subducted
magma source depth, found to be approximately
plate were almost stationary for a period of time, partial
in melting
a
limited
area
would
produce
a
magma
with
ratio
a+ Rb:Ca
high Na
first,
If ,the plate were to .move far
changing to a lower ratio with. time.
melting andslow down
enough to bring fresh material into theof zone
or stop again, the melting'sequence would occur for a second time.
The
Palisadessill magma could be a result of partial melting of differ-
an inconsistently moving plate.
ent parts of
..
. .i
.?'
62
slli
500 KILOMETRES
0
Figure29.Spatialrelationships
Olipocene
+Eocene
of thePalisades
imbricatemiddleCenozoicsubduction
others,1972,p.
t i o n zone.
235).
s i l l to p o s s i b l e
zone (after Lipmanand
.Contoureddepths
(km)are tothe
Hachured l i n e i s a possible breakbetween
subductionzones.
two
subduc-
CONCLUSION
Figures 30 and 31 g i v e a summary of mineralogic and chemical
changes i n t h e magma as a r e s u l t of e q u i l i b r i u m c r y s t a l l i z a t i o n and
i n t r u s i o n phenomenon of the Palisades sill.
Assuming a model t h a t
primarily involves equilibrium crystallization, Figure
v a r i a t i o n irr a l l chemical constituents in the
31 shows t h e
magma with decreasing
t e m p e r a u . . T h e sequence of events that occurred
w i t h decreasing
temperaturehas follows: 1) p h e n o c r y s t c r y s t a l l i z a t i o n ( b i o t i t e ,
p l a g i o c l a s e , and quartz), 2) i n t r u s i o n , 3) quartzresorption
and
groundmass c r y s t a Z l i z a t i o n , 4 ) q u a r t z c r y s t a l l i z a t i o n , . and 5) p a r t i a l
d i s s o l u t i o n( F i g .
30).
As shown i n Figure
31, t h e r e is considerable
overlap o € temperaturerangefortheseprocesses.Finally,Figure
slaous -change
31
i n mineralogy of t h e s i l l with decreasing temperature.
:i
... ..
-Figure 30.
Schematic diagram of phases and crystallization
and alteration processes vs temperature
of crystallization.
..
Magnetite (less than-Y, percent) is excluded.. Temperature
is very approximate - based on Figure 23 using data a 3 Kb
:
:
.)
.
.
I
partialdissdlutlon
3
groundmass c r y s t a l l i z a t i o n
Crystallization
cnci Alterction
quartz
- . (groundmass) crystallization
quortzresorption
Precesses
P s i o n
phenocryst
crystallization
75
magma
Volume
50
(YO;
25
0
600
800
Figure 31.
Schemaeic diagram of oxide concentrations in the
magma vs temperature ok crystallization.
See Figure .30 for
respective
alteration processes..
Ti02 is excluded.
phases
and c~Std.liZa:iO~ and
ACKNOWLEDGMENTS
The author wishes to express his appreciationDr.toPaui C.
of this study, especially
Ragland for his assistance at all stages
for his
assistance in sample collection and his invaluable discussions
after critically reading an early draft of this thesis.
..
Paul Drez of the'university of North Carolina is thanked for
his invaluable help concerning analytic.procedures and for helpful
of this study. Thanks are dueto
discussions during the course
Sieve #i.sh of
Carolha for meaningful dis. . the University of Ncrth
4
.
cuszi=~r;. John Psgland, Caroline McLaughlin, and Susan Cannon are
A
gratefully thanked for their assistance in sample.collection.
special thanks goesto Susan Cannon for typing this thesis.
,_.. .. I
....
68
Harris, P. G., Kennedy, W. Q., and Carfe,
U., 1970,Volcanism
verpus plutanism
t h e e f f e c t of chemice'l composition, i n
Newall, G., and - s t , N., eds., Uechanisms of 1gne.ous I n t r u s i o n s :
Liverpool,. S2el Hause Press, p. 187-200.
--
Hyndmar;, D. W., 1972,Petrology of igneousandmetamorphk
New York, McGrm-Hill Book Co., 533 p.
reeks:
H.
Lipssin, F. U., Prostka,
J . , and Christiansen, R. L., 1972,
Cenozoic v o l c a n i s a and plate-tGctciidc evolution cf the Western
I. Earlyandmiddle.Cenozoic:
UnitedStates.Pt.
R. SOC.Lond. A., v. 271,p.
217-248.
Phil.Trsn5.
N m h , W. P., and'Wilkinson, J . F. G., 1970,Shonkin Sag l a c c o l i t h ,
K~ioutr*~Pe I. U f i c minerals and estimates of temperature,
pressure, oxygen fugacity, and silica a c t i v i t y : Contr.Mineralogy
andPetrology,
V.
25, p. 241-269.
Northrop, S. A . , and Read, C. B., 1966, Guidebook. of
. Taos-RatonSpsiiish Peak@ country, New Mexico and,Colorado:Socorro, New
Mexico Geologiczl Society, 128
p.
.
P i w l e s k i i , A. J.., 1968,Experimental s t u d i e s of igneousrock
series: Central Sierrr. Nevada Batholith, California: Jour.
76, p. 548-570.
E"cl~gy,.
..
V.
, aadWylliP-, P.'J., 1968, Experimental studies
of igneous
rock series: a zoned pluton i n t h e Wallowa Batholith, Oregon:
Jour. Geology,v.76,
p. 205-234.
-1970, Experimental studies
of igneous rock series: f e l s i c
body s u i t e from the Needle Point Pluton, Wallowa B a t h o l i t h ,
Oregon, Jour. Geology, v. 78, p. 52-76.
Robinson, G. D;, Wenek,-A. A., Hays, W. H., and McCallum, M. C.,
1964,Philmontcoilotry:
u. s. Geol. SurveyProf.Paper 505,
152 p.
S a l c t t i , C. A . , er?d &+-thews, V., 1969,Quartz-leachedgraphicGeol., V. IC,
g r a n i t e from Fanticello, Georgia: Southeastera
p . 185-188.
Schwartz, G. M., 1958, Alteration of b i o t i t e undermesothermal
p . 164-177.
conditlons: Econ. Geology,v.53,
and
Shaw, H. R., 1965, Comments on v i s c o s i t y , c r y s t a l s e t t l i n g ,
convection i n g r a n i t i c magmas: Am. Sour. Sci.. v. 263, p. 120-152,
Smith, J. F., and Ray, L. L., 1941, GP,OlOgy of tha Moreno Vailey,
Nev Mexico: Geol. SOC. Americn Bull., V. 52, p. 177-210.
.. .’?.I
...... . 3
.. . .:I
, :”
REFERENCES CITED
1
Barker, D. S., and Burmester, R. F., 1970,Leaching of quartzfrom
Precllmbrianhypabyssal rhyolite porphyry, Llano
County. Texas:
Contr. MineralogyandPetrology,
V. 28, p. 1-8.
Bartlett, R. W., 1969, Magma convection. temperature distribution,
Jour. Sci., V. 267, p. 1067-1082.
and d i f f e r e n t i a t i o n :
&.
the Meadow F l a t s .complex, O r m g e
Bland, A. E., 1972, Geochmistry of
County,North Carolina [M. S . ] . t h e s i s : Chapel Hill, Univ. of
North Carolina, 49 p.
Boyd, F. R., and England, J. L., 1960, The q u a r t z - c o e s i t e t r a n s i t i o n :
Jour. Gophys.Research,
v. 65, no. 2, p. 749-756.
Brown, 6. C., 1970, A comment on t h e Pole of water in t h e p a r t i a l
and Planetary Sci. Letters,
fusion of crustal rocks: Earth
VI
9 , ’ ~ .,355-358,
..
,
.
.
Burnham, C. W., andJahns, R. H., 1962, A method for’ d e t e d n i n g t h e
s o l u b i l i t y of vater t n s i l i c e t e melts: h. Jour. Sci.,
v. 260,
.. .
p.
721-745.
.i..*
Carmichael, I. S., Turner, F.
andVerhoogen,
J., 1974,Igneous
Petrology: New York, EcGraw-Hill BO& Co., 739 p.
Eichelberger, J. C., 1975,Origin of a n d e s i t e and dacite: evideoce
of-mixing a t Glaes Mountain i n C e l i f o r n i a and a t o t h e r circumPacificvolcanoes: Geol. SOC. h e r i c a bull.,,^. 86, p. 1381-1391.
,
1974, A$piication of ti p ~ r t i s z iof the syatem
C & . . 1 2 S f Z C S - X ~ S f , 0 -Si02-Y4C-Fe””
-2-E 2G C O 2 t o genesis of the
calc-a1 a i n e s u i e:
Jour. S c i . , V. 10, p. 297-315.
Eggler, D. H.,
28A
m
.
Flanagan, P. J., 1973,1972values
for international geochemical
samples: Geochim. e t Cosmochim. Acta, V. 37, p. 1189-1200.
Fyfe. W. S., 1970, Some thoughts on g r a n i t i c magmas,
Newall, G.,
and Past, N., eds., Mechanisms of IgneousIntrusions:Liverpool,
S e e i House Press, p. 201-216.
Hall, A. t., 1932, The Bushveldigneous complexof
Transvaal:Gaol.Surv.
S. Africo Hem. 28.
t h e Central
..<
: .i
69
1943, .Geology of t h e CimarronRange,
America Bull.,
54, p. 891-924.
New Mexico: Geol. SOC.
V.
V.,
1965, Factors i n t h e d i s t r i b u t i o n o f t h e
trace
Tauson, L.
elements during the crystallization of
magmas, & Ahrens,
L. H., Press, F., Runcorn. S. K., andUrey, H. C., eds.,
' Physicsand
chemist6 of t h e e a r t h , V. 6: Oxford, Pergamon
Press, p. 215-259.
Taylor, S. R., 1965, The a p p l i c a t i o n o f trace element d a t a t o
problems i n p e t r o l o g y , & Ahrens, L. H . , Press, R., Runcorn,
S. K., and Urey, H. C., eds.,Physicsandchemistryof
:ha
e a r t h , V. 6: Oxford, PergawnPress, p. 133-213.
Thornton, C. P., 2nd T u t t l e . 0. F., 1960.Chemistry
Jour.Sci.,
rocks. I. Differentiationindex:
p. 664-684.
A
m
.
of igneous
v. 258,
Turekian, K. K., and Kulp, J. L., 1956, The geochemistry of s t r o n tium: Geochim. e t Cosmochim. Acta, v. 1 0 , p. 245-296.
Turner, P. J . , 1968, Metamorphic Petrology: N e v York, McGraw-Hill
Book Co., 4U3 p.
T u t t l e , 0. F., and Bowen, X. L., 1958,Origin of g r a n i t e i n t h e
l i g h t of e q ~ e r i m a n t a l s t u d i e s i n t h e s y s t e n N a A l S i 3 0 8 - W S i 3 ~ Si02-H20: Geol. SOC. America Hem. 74,153 p.
Van Der Plas, L., and Tobi, A . . C . , 1965, A c h a r t f o r j u d g i n g t h e
Am. Jour. Sci., v. 263,
reliabilityofpointcountingresults:
p. 87-90.
in
Wager, L. R., and Deer, k'. A . , 1939,Ckologicalinvestigations
East Greenland.Pt. III. The petrology of the Skaergaard
i n t r u s i o n , iiangerdiugssuag. East Greenland: Medd. Greenland;
v. 105, no:4,
p. 1-352.
Washington, H. S . , 1917, C h d c a l a n a l y s e s o f igzeom rocks publ i s h e d from1884 t o 1913: U. S , Genl. SurveyProf. Paper 99,
12Oi p.
Winchell, A. N., andWinchell,
P t . 11: New York,JohnWiley
H . , Elements of Optical Mineralogy.
b Sons, Inc.,551 p.
Winkler, H. G. F., 1974, Petrogenesis of metamorphic rocks: New
York, Springer-Verlag, 320 p.
- - , Boose,
M., and Marcopoulos, T.. 1975, L c w t e q e r a t u r a grani t i c melts: N. Jb. Miner. Mh., p. 245-268.
-..
...:;.... .....
::::j
i
..
70
-
Wooden, J. L., 1975,Geochemistryand
Rb
Srgeochronology of Pre- .
cautbrian mafic dikes from.the Beartooth, Ruby Range, andTobacco
Root Mauntains, Montana [Ph.D.] dissert.: Chapel Hill, University
of North Carolina, 194 p.
Wyllie, P. J., and T u t t l e , 0. F., 1961,Experimentalinvestigation
of s i l i c a t e systemscontaining two volatile components. P t . 11.
The e f f e c t s o f Nli and HF, i n a d d i t i o n t o H20 on the melting
Jour. Sci.; v. 259,
temperatures of a b i t e and g r a n i t e :
p. 128-143.
Am.
?
1964, Experimental investigation of s i i i c a t e systems
containing two v o l a t i l e components. P t . 111. The e f f e c t s o f
SO3, P205, HC1, and L i 0, i n a d d i t i o n t o H20, on t h e melting temperatures of a1 i t e and g r a n i t e :
Jour. Sci.,
v.
i
262, p . 930-939.
Am.
"'
..
. ..,..!,.
i
H-prof i l e
.
sample
number
HI
HZ
H3
H4
H5
86
H7
H8
H9
.
.
HlG
H11
HI2
H13
H14
HI5
H16
B17
H18
H19
HZ0
HZ1
H22
823
H24
H25
H26
H27
H28
HZ9
H30
H31
832
833
H34
H35
H36
n37
1138
H39
H40
S i 0 2 A1203 Fe203 MgO CaO
(%)
67.9316.14
68.08 15.33
67.2816.64
67.59 1 s s a
66.8115.93
67.7615.54
68.1015.71
'68.33 15.68
68.28 15.77
68.77 15.80
67.14.15.65
68.32 15.99
68.84 16.05
68.97 15.92
'68.84 15.67
68.74 15.65
69.00 15.75
68.60 15.91
65.90 15.67
65.48 15.00
67.66 15i61
65.4315.33
68.2215.63
68.66 16.25
67.74 15.67
66.18 15.73
68.16 16.33
68.04 15.92
68.52 15.75
68.51 15.74
69.10 15.84
67.57 15.33
66.68 15.40
66.15 15.11
66.14 15.43
67.0515.38
66.48 15.89
66.36 15.41
67.04'15.49
64.76 15.75
2.88
2.32
2.10
2.58
2.54.
2.50
2.47
2.52
2.48
2.42
2.57
2.56
2.55
2.48
2.38
2.60
2.50
2.48
2.46
2.58
2.48
2.38
2.35
2.53
2.48
2.48
2.62
2.56
2.45
2.52
2.58
2.48
2.48
2.46
2.42
2.22
2.42
2.38
.2.36
2.47
0.751.72
0.64 3.11
0.64 2.90
0.74 2.42
0.86 2.66
0.781.96
0.63 2.24
0.73 1.84
0.76 2.18
0.62 2.38
0.96 3.05
0.61 1.86
0.54 1.63
0.621.73
0.54 1.16
0.66 1.52
0.70 1.33
0.68 2.11
0.72 2.42
0.754.23
0.68 1.73
0.64 4.39
0.66 3.06
0.641.63
0.53 3.57
0.78
3.62
0.64 2.45
0.72 1.72
0.64
2.98
0.721.77
0.71 2.07
0.50 3.33
0.24
3.43
0.64 3.56
0.59
3.40
4.23
0.54 3.35
0.68 3..11
0.54 3.16
0.56 3.46
0.70 3.80
.
Na20 K20
Ti02
5.50 2.96 0:30
4.50 3.20 0.28
4.38 3.44 0.28
4.88. 3.22 0.24
4.40 3.52 0.28
5.14 3.42 0.30
5.38 3.18 0.27
4.98 3.42 0.30
5.14
3.32
0.28
4.80 3.26 0.27
4.62 3.40 0.26
5.32 3.40 0.28
5.38 3.400.28
5.22 3.34 0.26
5.39 3.43 0.26
5.43 3.38 0.30
5.29 3.40 0.30
4.92 3.34 0.26
4.83 3.40 0.27
3.57 3.09 0.27
5.48 3.27 0.28
3.42 3.42 0.26
4.36 3.350.26
5.50 3.300.29
4.43 3.16 0.27
3.53 3.36 0.28
4.92 3.34 0.30
5.06 3.44 0.30
4.54 3.18 0.24
5.30 3.40 0.30
5.01 3.180.27
4.22 3.22 0.31
3.88
3.54
0.27
4.13 3.10 0.29
3.12 0.28
4.07 3.26 0.28
4.54 3.24 0.29
4.19 3.15 0.30
3.91 3.34 0.31
3.26 3.56 0.30
total
oxides
98 I18
97.46
97.66
97.25
97.00
97.40
97.98
97.85
98.21
98.32
97.55
98.34
98.67
98.54
97.67
98.28
98.27
98.30
96.67
95.47
97.19
95.27
97.84
98.80
97.85
95.46
98.75
97.76
98.30
98.26
98.75,
96.96
95.92
95.44
95.61
96.15
96.65
95.49
96.47
94.60
Rb
Sr
(ppn;) (ppm)
693
588
467
662
434
615
983
659
767
944
775
966
905
1076
855'
1071
697
991
468
3iO
684
436
690
964
640
4ii
831
771
884
750
904
645
508
269
251
340
507
607
523
422
46
61
64
60
61
57
52
63
54
53
57
60
57
59
61
54
60
57
56.
54
=.a
68
62
54
60
60
64
63
58
59
56
58
71
61
54
58
61
58
63
75
"
77
72
V-profile
v1
v2
v3
v4
V5
V6
v7
V8
v9
v10
v11
v12
V I3
V14
V15
V16
V17
1118
v19
v20
v21
v22
V23
v24
v25
66.3315.41
66.62 15.32
68.43 15.56
68.5115.97
68.91 15.58
69.26 15.34
68.24 15.81
68.6515.86
68.36 15.58
68.36 15.81
68.2215.98
67.6815.88
68.6815.77
67.4115.30
66.08 15.61
67.18 15.51
66.4015.73
67.77 16.04
68.0416.17
67.37 15.41
67.5316.49
66.4815.50
71.96 14.33
71.83 14.65
69.27 15.31
2.38 0.68 3.52 3.95
2.40 0.69 3.45 3.80
2.46 0.74 3.01 4.51
2.57 0.64 2.66 4.61
2.49 0.68 2.66 4.43
2.46 0.68 2.50 4.42
2.44 0.55 2.63 4.64
2.41 0.56 1.38 3.14
2.40 0.72 2.78 4.48
4.36
2.58 0.731.78
2.46 0.70 2.76 4.54
2.46 0.70 2.28 5.00
5.36
2.48 0.601.73
2.31 0.64 3.14 4.40
2.44 0.78 3.37 4.4.4
2.42 0.72 2.48 4.33
2.46 0.66 3.17 4.50
2.75 0.82 2.03 4.91
3.02 0.85 3.00 4.79
2.98 0.90 3.00 4.91
2.81 0.76 1.00 5.46
2.99 '0.84 2.67 4.82
1.02 0.16 1.64 4.18
1.04 0.16 0.83
5.68
1.50 0.24 1.83 4.44
3.280.27
3.21 0.30
3.36 0.27
3.24 0.25
3.28 0.27
3.18 ,0.27
3.20 0.26
3.14 0,30
3.30 0.28
3.34 0.32
3.240.28
3.26 0.29
3.32 0.27
3.12 0.26
3.18 0.30
3.22 0.28
3.30 0.28
3.24 0.32
3.06 0.32
3.32 0.33
3.35 0.34
3.30 0.36
3.98 0.07
4.04 0.08
3.20 0.29
95.81
95 * 79
98.34
98.45
98.30
98.11
97.77
97.44
97.98
97.28
98.18
97.55
98.21
96.58
96.20
96.14
96.50
97.86
99.26
98.22
97.74
96.96
97.34
97.31
96.08
562
464
912
910
893
882
940
823
780
644
909
799
922
590
617
551
606
700
909
934
655
370
486
549
677
62
59
55
57
61
52
56
54
59
56
57
57
60
57
59
58
59
57
58
55
56
63
122
108
57
'
73
APPENDIX B: "STRATIGRAPHXC" DISTANCE OF
SAMPLES FROM LOWER CONTACT
H-profile
V-profile
,
actwl
actuel
distance
distanca
".
, _ _
"
"
"
,
H1
H2
H3
H4
HS
H6
H7
H8
H4
HI0
H11
H12
H13
H14
H15
H16
H17
Hi8
H19
HZ0
H21
H22
H23
H24
H25
0.5
27.7
77.1
93.9
83.8
80.8
86.9
85.6
99.4
120.4
139.0
149.0
164.0
176.8
188.1
202.4
211.8
218.2
224.3
232.0
245.1
242.3
250.9
262.7
259.4
recalculated distance*
(used in Thesis)
v1
v2
v3
v4
H26
H27
H28
H25
H30
Rfi
H32
H33
H34
H35
H36
H37
E38
H39
H40
,271.9
277.4
282.5
289.0
303.0
3ii. 8
324.3
332.5
346.3
353.0
357.2
366.7
378.9
381.9
382.8
V5
V6
V7
V8
v9
v10
v11
v12
V13
vl4
v15
Vl6
V17
V18
. v19
v20
v21
v22
V23
v24
V25
'
0.3
9.1
18.3
27.4
42.9
. 64.0
75.0
91.7
106.3
120.i
134.7
160.3
174.9
191.4
206.0
218.8
231.6
251.7
262.4
275.8
292.6
306.0
. 315.7
329.8
343.5
0.3
21.1
20.4
30.5
47.9
71.0
83.'5
102.1
118.3
133.5
149.7
178.3
194.5
212.8
228.9
243.2
257.6
279.8
291.7
306.6
325.2
340.2
355.4
366 4
381.6
APPENDIX C: ANALYTICAL PROCEDURES, PRECISION, AND ACCURACY
X-ray Fluorescence
Element
Tube
Crystal
Detector
Analytical
Line
Information
Comments
1 .
si
Cr
Cr
Ca
PET
Flolr proportional Kp1.2
(gas)
Fixed time count (10 sec.).
Background not counted.
Vacuum used.
PET
Flow proportional Kd1,2
Fixed time count (10 see.).
Background not counted.
(Pas)
Ti
Cr
PET
St
>so
LiF (220)
Rb
KO
LiF (220)
-.
Fixed time count (10 sec.) .
Background not counted.
Vacuum used.
Scintillation
Scintillation
K61,2
K41,2
Fixed
count
of 10,000 and
40,000 on 3 backgrounds
and 2 peaks, respectively.
. Fixed count of 10,000 and
40.000 on 3 backgrounds
and 2 peaks, respectively.
. ,. .,.. .
.”.....
'.. .
.
..... .
.:.;.. ...)
. ,
I
;
,
..)
,.
75
Atomic Absorptior. Analytical Information
Elenent
Line.
Na
Vis f 293.5
K
Vis 1 302.0
-.
1 309.4
Al
b"i
Mg
W 1 205.4
FP
W
1 249.3
1 'Oddent
,
.
.
.
Bead Out
Acetelene
I air
Null meter
Acetelene
I air
Null meter
Acetelene
1 NQ2
Null
Acetelene 1 air
Acetflene
f air
meter
Null
meter
N U I ~' m t e r
81
-..
...
..
76
AGV
-
A*
sio2
A
B
Alco..
A
B
Fe203*
A
B
MgO
A
B
CaO
A
B
Na2O
A
B**
~.
69.19
68.95
52.54
52.26
1 7 9c
*.r.
15.04
16.85
2.77
2.75
11.09
0.78
6.62
^....I
6.80
”
1.53
4.98
”
”
”
10.96
4.33
4.34
4.15
2.15
x20
2.’90
2.96
4.51
4.51
0.64
0.61
TiOl
1.08
0.53
0.52
1.0;
”
”
**
*I.
4
.
1.99
1.95.
5.00
-
3 E
0.79
”
A
B
WL
”
59.00
59.14
17.35
B
*
01
_
I
”
”
--
Accepted value from Flaneg’aan (1973).
This.8tudy.
Precision
si02
-
Coefficient of
Variation
0.32
K20
- -
Ma20
0.90
0.90
1.33
0.61
0.67