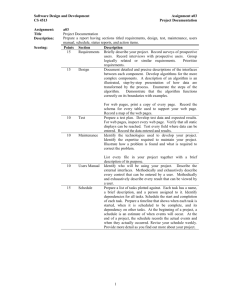
, , 7-9. n 0 1 2

7-9. (a) For n = 3, l = 0 1 2
(b) For l =
0 , m
=
0 . For l =
1 , m
= !
1 0 1 . For l =
2 , m
=
2 1 0 1 2 .
(c) There are nine different m -states, each with two spin states, for a total of 18 states for n = 3.
7-10. (a) For l = 4
L = ( + 1 ) h = h = 20 h m l
= 4 h
!
min
= cos " 1
4
20
# !
min
(b) For l =
2
L = 6 h
!
min
= cos " 1
2
6 m l
= 2 h
# !
min
=
7-12. (a) l = 1
L = 2 h
+1
0
(b)
− 1
+2
+1
=
°
°
0
l = 2
L = 6 h
(c)
+4
+3
+2 l = 4
L = 20 h
+1
0
− 1
− 2
− 3
− 4
(d) L = ( + )
7-13. L 2
= L 2 x
+ L 2 y
+ L 2 z
!
L 2 x
+ L 2 y
= L 2
" L 2 z
= ( + 1 ) h 2
" ( )
2
=
( 6 " m 2 ) h 2
(a) ( L 2 x
+ L 2 y
) min
=
(
!
2 ) h 2
= 2 h 2
(b) ( L 2 x
+ L 2 y
) max
=
(
!
2 ) h 2
= 6 h 2
(c) L 2 x
+ L 2 y
= ( 6 1 ) h 2
= h L L
(d) n = 3
7-15. L = r ? p d L dt
= d dt r
!
p + r !
d p dt d r dt
!
= !
m m
! =
0 and r
!
d p dt r F .
Since for V =V ( r ), i.e., central forces,
F is parallel to r , then 0 and d L dt
=
0
7-16. (a) For l = 3 , n = 4, 5, 6, … and m = − 3, − 2, − 1, 0, 1, 2, 3
(b) For l = 4 , n = 5, 6, 7, … and m = − 4, − 3, − 2, − 1, 0, 1, 2, ,3 ,4
(c) For l =
0 , n = 1 and m = 0
(d) The energy depends only on n . The minimum in each case is:
E
4
= !
2
= !
.
eV / 4 2
= !
eV
E
5
= !
.
eV / 5 2
= !
eV
E
1
= !
eV
7-17. (a) f n
=
6 , l =
3
(b) E
6
= !
13 6 eV n 2
= !
13 6 eV / 6 2
= !
eV
(c) L
= ( +
1 ) h = ( ) h =
12 h = "
!
34 J s
(d) L z
= m h L z
= !
3 h , !
2 h , !
h h h h
7-20. (a) For the ground state, ( ) $ r
= !
2 ( 4
" r 2 )
$ r
=
4 r 2 a
0
3 e # 2 /
0 $ r
" r =
For " r = 0 03 a
0
, at r = a
0
we have ( ) " r =
4 a 2
0 a
0
3 e !
2 ( a
0
) =
(b) For
.
a
0
, at r = a
0
( ) " r =
( a
0
)
2 a
0
3 e !
4 ( a
0
) =
7-21. ( ) = Cr e !
2 /
0
For P ( r ) to be a maximum, dP dt
= (
.
2
$
*
,
!
2 Z a
0
%
+
e !
2 /
0 + 2 re !
2 /
0
#
) = 0 & C '
2 a
Zr
0
$
* a
Z
0
!
!
2 /
0 = 0
This condition is satisfied with r = 0 or r = a
0
/Z.
For r = 0, P ( r ) = 0 so the maximum
P ( r ) occurs for r = a
0
/Z.
7-22. ' "
2 d # =
&
' ' '
0 0 0
" sin $ $ % = 1
=
#
4 ! "
0
+ = 4 !
C 2
210
+
0
# % Zr &
'
) a
0
(
*
2
$ /
0 dr = 1
=
4 !
C 2
210
"
*
0
$
&
(
Letting a
0
2
%
'
) e # /
0 dr
=
1
=
0
we have that r
=
/ substituting these above,
&
"
2 d # =
4 !
a C 2
210
Z 3
&
0
$ x
Integrating on the right side
and dr
=
/
#
0
!
x
=
6
and
Solving for C 2
210
yields: C 2
210
=
Z 3
24 !
a 3
0
$ C
210
=
"
%
'
Z 3
24 !
a
0
3
#
&
(
7-26. For the most likely value of r , P ( r ) is a maximum, which requires that (see
Problem 7-24) dP dr
= A cos 2
!
#
' r 4
%
)
+
"
Z a
0
&
*
, e " /
0 + 4 " /
0
$
( = 0
For hydrogen Z = 1 and A cos 2
!
( /
0
) ( 4 a
0
" ) "
0 = 0 . This is satisfied for r = 0 and r = 4 a
0
. For r = 0, P ( r ) = 0 so the maximum P ( r ) occurs for r = 4 a
0
.
7-33. (a) There should be four lines corresponding to the four m
J values − 3/2, − 1/2,
+1/2, +3/2.
(b) There should be three lines corresponding to the three m l
values − 1, 0, +1.
7-68.
=
4 Z 3 a 3
0
!
2 /
0
(See Problem 7-63)
For hydrogen, Z = 1 and at the edge of the proton r = R
0
= 10 !
15 m .
At that point, the exponential factor in P (r) has decreased to: nucleus, e !
2 /
0 = e !
( !
15 ) (
"
!
10 m )
= e !
(
"
!
5 )
# !
"
!
5
#
1
Thus, the probability of the electron in the hydrogen ground state being inside the to better than four figures, is:
=
4 r 2 a 3
0 r
0
P = ( )
0
!
= !
0
R
0 4 r 2 4 a 3
0
= a
0
3
!
0
R
0
=
4 a
0
3 r 3
3
R
0
0
=
4 a
0
3
" R
0
3
#
%
'
3
&
(
=
(
( !
15 m )
3
$
!
10 m )
3
= $
!
15
7-70. (a) Substituting !
( ) into Equation 7-9 and carrying out the indicated operations
yields (eventually):
% h 2
2 µ
!
( ) 2 / r 2
% a
0
2
% h 2
2 µ
!
( ) (
%
2 / r 2 )
+
V
!
( ) =
E
!
( )
Canceling !
( ) and recalling that r 2
= 4 a
0
2 (because !
given is for n = 2) we
have
!
h 2
2 µ
(
!
1 4 a 2
0
)
+ =
E
The circumference of the n = 2 orbit is:
C = 2 !
( 4 a
0
) = 2 " # a
0
= "
/ 4 !
= 1 2 k .
Thus, # h 2
2 µ
!
'
#
1
4 4 k 2
"
(
+ V = E $ h
2 µ
+ V = E p 2
(b) or
2 m v E and Equation 7-9 is satisfied.
$
,
0
!
2 dx
= , A 2
& r '
2
(
* a
0
)
+ e % /
0 cos 2
" r 2 sin " drd d
A 2
,
0
$ & r '
2
(
* a
0
)
+ e % /
0 r dr
!
,
0
2 !
cos 2
" sin d d #
0
, = 1
Integrating (see Problem 7-22),
=
1
A 2 ( ) ( / )( ) =
1
A 2
= a
0
3
!
" A = a
0
3
!