Document 10808063
advertisement

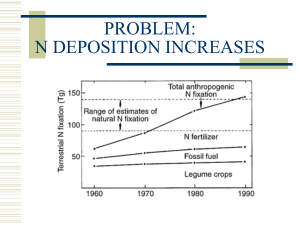
Wetland and Soil Chemistry for GHG Emissions Key Words: Wetland; Soil; Greenhouse gases; Dry and wet deposition; Sewage treatment; Life cycle assessment Description: Wetland breeds varied plants, animals and microorganisms. The crisscrossed roots of plants and soil colloid offer a perfect condition for sedimentation. Then organics, trace elements, nitrogenous substances, phosphorus-­‐containing compound and even heavy metals could be removed in certain ways with less ecological effects. Artificial wetland is generally constructed to treat wastewater. Wastewater treatment processes are identified as important sources of world GHGs (greenhouse gases) budgets. During the aerobic biological treatment processes, organic carbon is transferred into carbon dioxide through anabolism, catabolism and biomineralization by aerobic microorganisms. In the processes of anaerobic biological treatment, methanogenus would combine with acetogen and fermentative bacterium to generate methane. Nitrous oxide as an emerging GHG produced from biological nitrogen removal treatment is mainly due to incomplete nitrification and denitrification processes. To evaluate the environmental burdens associated with a product, process, or activity from cradle to grave, life cycle assessment (LCA) is applied by identifying, quantifying and assessing the impact of the utilized energy, materials and the wastes released to the environment. LCA could identify and evaluate opportunities for environmental improvements to guide government policy, strategic planning, marketing, consumer education, process improvement, product design, eco-­‐labeling and consumer education programs throughout the world. Pictures: Fig. 1. Gas sampling of lab-­‐scale subsurface flow constructed wetland Fig. 2 Photo of on-­‐site gas sampling in wastewater treatment plant Fig. 3 Lab-­‐scale aquaponic systems 2174 Environ Earth Sci (2013) 68:2171–2180 NH4+ Amo NH OH 2 N 2O NOH Hao NO3- Nar iNor N 2O NO2- Nor NO2·NHOH Fig. 2 Scheme denitrification NO3N2O NO 2- Nir of relevant NO N2O Nr N2 O production Nos pathways N2 in Fig. 1 Scheme of relevant N2O production pathways in nitrification the cell while N2O will be released using NO2- as Fig. 4 Scheme of relevant N2O production pathways in AOB’s nitrification an electron acceptor to avoid the accumulation of NO2- in the system. In nitrifier denitrification, N2O is an intermediate product during the reduction of NO2- to N2 under O2Nir Nos Nr asNthe - Nar N2 NOnitrifiers The of areNO regarded NOactivities 2 2O main nitrogen 3 limited conditions, which is also similar to heterotrophic removal pathway in CW systems. Among all species of denitrification (Wrage et al. 2001). Fig. 2 Scheme of relevant N2O production pathways in nitrifier to nitrification, Indaddition to nitrifiers, N2O can be produced by aerobic Fig. 5 Scontributing cheme of relevant N2O pammonia-oxidizing roduction pathways in enitrification denitrification bacteria (AOB) are ubiquitous in wetland ecosystems and denitrifiers, which contribute to the reduction of NO3- or dominant members of the rhizosphere microbial NO2- to N2O and N2, respectively. Blankmer et al. (1980) are community. found that heterotrophic bacteria aid in the release of N2O the AOB’s cell while N2O will be released using NO2- as Nitrification is a bacterial respiratory process with two under low DO, low sludge retention time, and acidic an electron acceptor to avoid the accumulation of NO2- in successive steps carried out by ubiquitous AOB and nitritecondition. the system. In nitrifier denitrification, N2O is an intermeoxidizing bacteria (Inamori et al.- to 2008). AOBO isdiate product during(NOB) the reduction of NO N under 2 2 2 responsible for nitrite (NO2is-)also formation, whereas NOB is N2O emission in denitrification limited conditions, which similar to heterotrophic responsible for (Wrage the conversion of NO2 to NO3 . The first denitrification et al. 2001). stepIncan be catalyzed by ammonia (Amo) Denitrification is the microbial-mediated process carried addition to nitrifiers, N2O canmonooxygennase be produced by aerobic -step and hydroxylamine oxidoreductase (Hao). The second out by facultative aerobic bacteria and aerobic microordenitrifiers, which contribute to the reduction-of NO3 - or of the- nitrification pathway, oxidation of NO2 to NO3 , is ganisms in hypoxic or anaerobic conditions, whereby NO 2 to N2O and N2, respectively. Blankmer et al. (1980) catalyzed by nitrite oxidoreductase (Nor). N O emission NO3- is converted into N2 via the intermediates NO2-, 2 found that heterotrophic bacteria aid in the release of N2O isunder affected by the activity and expression of these three nitric oxide (NO), and N2O. The generally recognized low DO, low sludge retention time, and acidic enzymes, which are influenced by many factors under pathway is shown in Fig. 2 (Tallec et al. 2008). condition. different environmental conditions (Hu et al. 2011). Fig. 1 N2O is produced as an intermediate of denitrification, shows the pathway of N2O emission in nitrification. which is different from nitrification. Microbial denitrificaN2O emission in denitrification As a nitrification byproduct, N2O is produced mainly in tion is a respiratory process that consists of four consecuEnviron Earth Sci (2013) 68:2171–2180 N 2O emission in nitrification ao iNor N 2O NO2- Nor NO2·NHOH NO3N2O on pathways in nitrification ed as the main nitrogen Among all species of on, ammonia-oxidizing wetland ecosystems and rhizosphere microbial atory process with two uitous AOB and nitritei et al. 2008). AOB is ation, whereas NOB is O2- to NO3-. The first - Human toxicity impact (cases/year) Human Air pollution activities PAHs Inhalation Plant DNA damage 400 Straw (Rice) Bituminous coal Indigenous coking Others Firewood Alumuninum Metallurgical coking 300 200 100 0 400 Huamn toxicity impact (Case/year) Sediment Mechanism analysis Water 2005 2006 2007 2008 2009 2010 2011 2012 Soil Ingestion BaP IcdP Chr Flu BbF Others DahA BghiP NaP BkF 300 200 100 0 2005 2006 2007 2008 2009 2010 2011 2012 1.0E-05 1.0E-06 <5 Inhalation 5 ~ 15 BghiP Chr BkF BbF IcdP Nap DahA Flu BaP 1.0E-08 BghiP Chr BkF BbF IcdP Nap DahA Flu BaP PAHs air emissions distribution (kg/km2) 4µmol/L 1.0E-07 700 120 y = 0.15x + 38.48 R² = 0.69 600 60 400 30 y = 1.84x - 100.85 R² = 0.98 300 0 200 Ingestion 24h 15 ~ 25 90 500 48h 72h PAHs emissions (kt) 1.0E-04 0.5µmol/L 1µmol/L iF (kginta ke/kgemission) 1.0E-03 HTP (cases,epidemiology ) Control 1.0E-02 240 280 320 360 400 HTP (cases, animal data) Sustainable management 25 ~ 40 > 40 Energy, raw material supply, waste disposal, labor, carbon emissions, infrastructure, and maintenance cost Indentifying key factors Economic assessment Inputs Process 1 Outputs Inputs Process 2 Outputs Inputs Process n Outputs Social information Technical information (Improvement potential, safety, human rights, worker initiative, etc) of each process (e.g., experiments, advancment, maneuverability, legality, feasibility) of each process Social assessment Technical assessment Life cycle multi-objective assessment Emissions and consumption Environmental impact assessment at mid-point and end-point level Indentifying key factors Environmental assessment Fig. 6 Life cycle assessment Field study Description: The Weishan Lake Wetland, located in Jining City of eastern China's Shandong Province, is one of the most famous national wetland parks in the country, thanks to its abundant lakes, flowers, plants and animals, as well as its breathtaking scenery. It was award “Top Ten Most Charming Wetlands” in 2013. Pictures: Fig.1 Photo of wetland at Lake “Weishan” Fig.2 Photo of wetland at Lake “Weishan” Fig.3 Photo of wetland at Lake “Weishan” Internet links: http://www.huanke.sdu.edu.cn/en/index.php http://en.sdu.edu.cn