Exempt 21 CFR 812 (FDA) Investigational Device Exemptions (IDE) 3 Types of Device Studies
advertisement

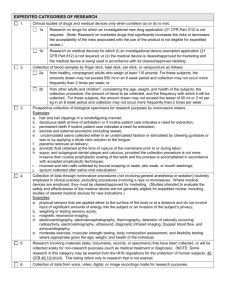
21CFR812(FDA) InvestigationalDeviceExemptions(IDE) 3TypesofDeviceStudies NOTE:Whenusingadevice aspartofresearch, Researchersmustcomplete AppendixK. Version08-23-11 SignificantRisk (SR) SRdevicesrequirefullcommittee reviewuponinitialreviewandat continuingreview(atleast annually). UseofanSRdevicerequiresan InvestigationalDeviceExemption (IDE)andmustfollowalloftheIDE regulationsat21CFR812. TheFDAisthefinalarbiterof whetherdeviceisSR.The Researchermayinclude documentationfromtheFDAofthe riskassessment.Whenthisoccurs, theIRBdoesnotneedtomakea formaldetermination. TheIRBmustdocumenttheSR assessmentinthemeetingminutes uponinitialreview. SRassessmentbasedsolelyon seriousnessofharmthatmayresult fromtheuseofthedeviceinan investigation,notonthedevice alone. ReassessmentofSRmayoccur basedonmodificationsthatinvolve ariskorotherchangesthatmay affectriskdetermination. IRBfurtherconsidersifstudy shouldbeapprovedornotasper criteriaforIRBapprovaland applicableUC/UCIpolicies. EvidenceofIDEneededbeforeIRB approvalmaybegranted(e.g.FDA determinationletter,IDE#listed onSponsorMasterProtocol). SRExamples: Surgicallasersforuseinmedical specialties Tissueadhesivesforneurosurgery, gastroenterology,ophthalmology, general&plasticsurgeryand cardiology Cardiacbypass&assistdevices NonsignificantRisk (NSR) NSRdevicesrequirefullcommittee reviewuponinitialreview.TheIRB mustdocumenttheNSRassessmentin themeetingminutesuponinitial review. UseofanNSRdeviceDOESNOTrequire anInvestigationalDeviceExemption (IDE). TheFDAisthefinalarbiterofwhether deviceisNSR.TheResearchermay includedocumentationfromtheFDAof theriskassessment.Whenthisoccurs, theIRBdoesnotneedtomakeaformal determination. NSRassessmentbasedsolelyon seriousnessofharmthatmayresult fromtheuseofthedeviceinan investigation,notonthedevicealone. ReassessmentofNSRmayoccurbased onmodificationsthatinvolveariskor otherchangesthatmayaffectrisk determination. IfdeterminedNSR,futurereviewsmay beexpeditedviacategory9ifcriteriais met.Thisshouldbedocumentedinthe minutesattimeofreview.Maybe category1bif sponsorprovidedaFDANSR determinationletteratinitialsubmission. IRBfurtherconsidersifstudyshouldbe approvedornotaspercriteriaforIRB approvalandapplicableUC/UCI policies. ResearchersthatuseNSRdevicesas partoftheirIRBapprovedresearch studymustfollowABBREVIATED requirementsper21CFR812.2(b). NSRExamples: Lowpowerlaserforpaintreatment ExternallywornmonitorforinsulinRXN Dentalfillingmaterialmadefrom traditionalmaterials Dailywearcontactlenses Exempt Mayqualifyforexpeditedreview, category#1b. Exempt=exemptfromIDE requirements(21CFR812)-with someexceptions. 7exemptioncategoriesin21CFR 812.2(C) Thefirsttwocategoriesfor exemptionpertaintodevicesthat wereeithermanufacturedbefore 1976orsimilarproducts manufacturedafter1976(referred toasa510Kdevice). A510Kdeviceismanufacturedafter 1976andisdeterminedas substantiallyequivalenttoadevice incommercialdistribution. Includesdiagnosticdevicestudies (e.g.invitrodiagnosticstudies)as longassponsorcomplieswith21 CFR809.10(C). Categories3(diagnosticdevicethat meetsspecificcriteria)and4 (consumerpreferencetesting, testingofamodificationor combination)arethemost commonlyappliedforexemptions. Anexemptstudybeingconductedto collectdatatosupporteithera clinicalinvestigationormarketing applicationmustcomplywith21 CFR50(protectionofhuman subjects)andshouldcomplywith21 CFR56(IRB). ExemptExamples: Consumerpreferencetesting Testingofadevicemodification Testingof2ormoredevicesin commercialdistributionifthe testingdoesnotcollectsafetyand efficacydataorputsubjectsatrisk