The Fundamental Interactions of 2D Nanomaterial Powders and Surfactants
advertisement

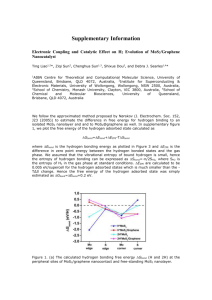
The Fundamental Interactions of 2D Nanomaterial Powders and Surfactants Curtis Heishman, Richard Livingston, Dale Brown, and David Estrada Abstract Techniques High yield and affordable production of graphene and other two-dimensional (2D) crystals has been a topic of considerable research in multiple disciplines. Liquid exfoliation utilizing ionic surfactants has proven to be an effective way to create 2D materials with potential uses such as lithium ion battery electrodes, printable electronics, and thermal interface materials. The focus of this research is the examination of the relationship between surfactants used in the exfoliation process and the quality of the resultant 2D nanoflakes of graphene and transition metal dicalchogenides (TMDs). To do this we first create MoSe2, MoS2, and graphene suspended flakes via liquid exfoliation in an aqueous solution of ionic surfactant. We then create dry powders of randomly stacked 2D crystals for further use in device applications by freeze drying the solutions. Finally we perform DOSY NMR Spectroscopy, IR Spectroscopy, Raman Spectroscopy, Thermogravimetric Analysis, and Inductively Coupled Plasma Atomic Emission Spectroscopy to elucidate the fundamental interactions of surfactants with our 2D crystals. Characterization Single Layer MoS2, MoSe2, Graphene Solutions Preparation •80mg Bulk Powders •40mL Soldium Cholate Solution (2% w/v) •Tip Sonication in Ice Bath 30 min •Centrifuge 25 min @ 4500 rpm •Creates 2D Materials in Surfactant Micelles e.g. Jonathan Coleman 5 Lyophilization to Create 2D Powders •0.110 mBar, -53 C, 24 hours IR Spectroscopy Sodium Cholate Ionic Surfactant Fig 4 MoSe2 + NaCh overlayed w/ Sodium Cholate 1H Lyophilization Data Research Relevance Create a pellet of mixed 2D nanomaterials • Cheap, Efficient, High Reproducibility, Easy To Work With • MoSe2, MoS2, Graphene, WS2, WSe2 • Achieve a high quality ZT Value Material MoSe2 MoS2 Graphene Graphene + MoS2 Volume of Solution 15mL 15mL 15mL 14mL Mass Yield 0.1820 g 0.2930 g 0.2550 g 0.2677 g Density of Solution 12.133 g/L 19.533 g/L 17.000 g/L 19.121 g/L ZT = s2 σ T λ-1 (Thermoelectric Figure of Merit) s = Seebeck coefficient (converting temperature to current) σ = Electrical Conductivity λ = Thermal Conductivity MoS2/Graphene Heterostructured Nonvolatile Memory T = Temperature NaCh MoSe2 06/23/14 NaCh MoSe2 06/13/14 NaCh 06/13/14 Future Work Van der Waals Heterostructures Labconco FreeZone 4.5L Graphene & Surfactant Flakes MoSe2 Flakes Heated to 575 C Characterization Thermogravimetric Analysis 6 Fig 2 Funded by: National Science Foundation, Office of Special Programs, Division of Materials Research •Grant Number DMR 1359344 Fig 3 NMR Spectroscopy Evidence of Hydrolysis Fig 1 Common TMDs MoSe2 after heating to 575 C To Remove Surfactant • • • • • MoS2 & MoSe2 + Surfactant Powders Heated to 1100 C, 50 C per minute, 21 minutes ~ 71% of Powder is NaCh Surfactant Surfactant Digests at Min 390 ̊C - Max 550 ̊C Yield @ ~20%, but does Sodium remain? Preliminary ICP MS anaylsis suggests yes TGA of MoS2 + Sodium Cholate Surfactant Powder • • • • • ICP MS Analysis to determine levels of sodium doping Raman Spectroscopy to determine thickness of flakes Investigate potential to bind molecules to 2D flakes Find non-ionic surfactants that can be lyophilized Heat Flakes in Inert Atmosphere of Tube Furnace and analyze TGA of MoS2 + Sodium Cholate Surfactant Powder Acknowledgements 1. A. K. Geim and I. V. Grigorieva, Nature 499 (7459), 419425 (2013). 2. Q. H. Wang, K. Kalantar-Zadeh, A. Kis, J. N. Coleman and M. S. Strano, Nature Nanotechnology 7 (11), 699-712 (2012). 3. A. K. Geim and I. V. Grigorieva, Nature 499 (7459), 419425 (2013). 4. Seong, Maen-Je, Journal of Korean Physical Society Vol 58, No 4 5. Science 331, February 4, 2011, DOI: 10.1126 6. Dr. Jerry Harris, NNU