Hess's Law: Enthalpy Change Calculations in Chemistry
advertisement

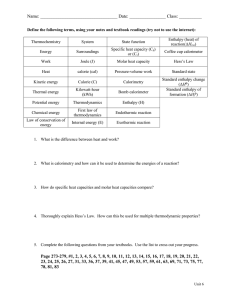
5.2 Hess’s Law From the Data Booklet • Section 11: Bond enthalpies and average bond enthalpies at 298K • Section 12: Selected compounds – Thermodynamic data • Section 13: Enthalpies of combustion 5.3 CH4(g) + 2 O2(g) CO2(g) + 2 H2O(l) ΔHθ = ? CH4(g) + 2 O2(g) CO2(g) + 2 H2O(l) ΔHθ = -808 kJ mol-1 5.3 Bond enthalpies ΔHrxn = ΣE (Bonds broken) – ΣE (Bonds formed) Combustion reactions: reacting with oxygen O2 Enthalpy of combustion ΔHco (kJ mol-1) – Energy released when one mole of a particular substance reacts with O2 5.1 Comparing Measured experimental values to Literature value HL stuff ΔHfo = Enthalpy of formation (SL) HL stuff 5.2 Hess’s Law • Understandings: – The enthalpy change for a reaction that is carried out in a series of steps is equal to the sum of the enthalpy changes for the individual steps. Law of thermodynamics: Conservation of energy 5.2 Hess’s Law Applications and skills • Application of Hess’s Law to calculate enthalpy changes. • Calculation of ΔH reactions using ΔHf data. • Determination of the enthalpy change of a reaction that is the sum of multiple reactions with known enthalpy changes. Using the equations below: C(s) + O2(g) → CO2(g) Mn(s) + O2(g) → MnO2(s) ∆Hο = –394 kJ mol–1 ∆Hο = –520 kJ mol–1 What is ∆H, in kJ, for the following reaction? MnO2(s) + C(s) → Mn(s) + CO2(g) A. 914 B. 126 C. –126 Determination of the enthalpy change of D. –914 a reaction that is the sum of multiple reactions with known enthalpy changes. Main reaction: MnO2(s) + C(s) → Mn(s) + CO2(g) Equations given: a. C(s) + O2(g) → CO2(g) ∆Hο = –394 kJ mol–1 b. Mn(s) + O2(g) → MnO2(s) ∆Hο = –520 kJ mol–1 Main reaction: MnO2(s) + C(s) → Mn(s) + CO2(g) Equations given: a. C(s) + O2(g) → CO2(g) b. Mn(s) + O2(g) → MnO2(s) ∆Hο = –394 kJ mol–1 ∆Hο = –520 kJ mol–1 Main reaction: MnO2(s) + C(s) → Mn(s) + CO2(g) a. C(s) + O2(g) → CO2(g) ∆Hο = –394 kJ mol–1 b. Mn(s) + O2(g) → MnO2(s) ∆Hο = –520 kJ mol–1 • If the direction of b is reversed, the sign of ΔH is also reversed (exo becomes endo). Main reaction: MnO2(s) + C(s) → Mn(s) + CO2(g) a. C(s) + O2(g) → CO2(g) ∆Hο = –394 kJ mol–1 b. Mn(s) + O2(g) → MnO2(s) ∆Hο = –520 kJ mol–1 • If the direction of b is reversed, the sign of ΔH is also reversed (exo becomes endo). b reversed. MnO2(s) → Mn(s) + O2(g) ∆Hο = +520 kJ mol–1 Main reaction: MnO2(s) + C(s) → Mn(s) + CO2(g) a. C(s) + O2(g) → CO2(g) b reversed. MnO2(s) → Mn(s) + O2(g) ∆Hο = –394 kJ mol–1 ∆Hο = +520 kJ mol–1 Main reaction: MnO2(s) + C(s) → Mn(s) + CO2(g) a. C(s) + O2(g) → CO2(g) b reversed. MnO2(s) → Mn(s) + O2(g) ∆Hο = –394 kJ mol–1 ∆Hο = +520 kJ mol–1 ____________________________________________________________________ Combining the two reactions C(s) + O2(g) + MnO2(s) → CO2(g) + Mn(s) + O2(g) Main reaction: MnO2(s) + C(s) → Mn(s) + CO2(g) a. C(s) + O2(g) → CO2(g) b reversed. MnO2(s) → Mn(s) + O2(g) ∆Hο = –394 kJ mol–1 ∆Hο = +520 kJ mol–1 ____________________________________________________________________ Combining the two reactions C(s) + O2(g) + MnO2(s) → CO2(g) + Mn(s) + O2(g) ∆Hο = ∆Hοa + ∆Hοb = (-394) + (+520) = +126kJ mol-1 Using the equations below: C(s) + O2(g) → CO2(g) Mn(s) + O2(g) → MnO2(s) ∆Hο = –394 kJ mol–1 ∆Hο = –520 kJ mol–1 What is ∆H, in kJ, for the following reaction? MnO2(s) + C(s) → Mn(s) + CO2(g) A. 914 B. 126 C. –126 D. –914 Hess’ Law • A Swiss-born Russian chemist Germain Henri Hess (1802 – 1850), proposed a precursor of the 1st Law of Thermodynamics (the Law of Conservation of Energy) • Hess’ 1840 statement referred to a ‘Conservation Law for Heat of Reactions’ (but he did not explicitly relate it to exchanges of heat and work) Hess’s Law • The enthalpy change of a reaction depends only on the identities of the reactants and products. • It is independent of the reaction pathway (how you get there doesn't matter). • This means enthalpy is a "state function." • *Hess' Law is essentially the Law of Conservation of Energy applied to chemical reactions. 5.2 Hess’s Law • Understandings: – The enthalpy change for a reaction that is carried out in a series of steps is equal to the sum of the enthalpy changes for the individual steps. Law of thermodynamics: Conservation of energy 5.2 Hess’s Law Applications and skills • Application of Hess’s Law to calculate enthalpy changes. • Calculation of ΔH reactions using ΔHf data. • Determination of the enthalpy change of a reaction that is the sum of multiple reactions with known enthalpy changes. 1. Linear representation 2. Energy diagram representation 3. Energy cycle representation (All main reactions are going from A to C) Solving Hess’s Law problems using “simultaneous equation method” Solving Hess’s Law problems using “simultaneous equation method” Solving Hess’s Law problems using “simultaneous equation method” • If the equations can be manipulated to "add up" to a specific reaction, then the enthalpy change for that reaction can be easily calculated by adding up the component enthalpy changes. Hess’ Law: Details • You can always reverse the direction of a reaction when making a combined reaction. • When you do this, the sign of DH changes. N2(g) + 2O2(g) 2NO2(g) DH = 68 kJ 2NO2(g) N2(g) + 2O2(g) DH = -68 kJ Hess’ Law: Details • The magnitude of DH is directly proportional to the quantities involved • E.g. if you need to multiply the number of moles, you also multiply the magnitude of DH N2(g) + 2O2(g) 2NO2(g) 2N2(g) + 4O2(g) 4NO2(g) DH = 68 kJ DH = 136 kJ 5.2 Hess’s Law • Understandings: – The enthalpy change for a reaction that is carried out in a series of steps is equal to the sum of the enthalpy changes for the individual steps. Law of thermodynamics: Conservation of energy Hess’s Law – What’s the point? • Allows us to calculate enthalpy changes theoretically that are impossible, or near impossible, to carry out experimentally. • Allows us to confirm/ compare how experimental enthalpy changes and theoretically calculated ones compare. – Provides information about how accurate experimental methods and theoretical models are. Example: The following reaction is difficult for its enthalpy change to be measured experimentally C (s) + ½ O2 (g) CO (g) ΔH = ? Meanwhile, these two other reactions are easier to carry out and have ΔH measured accurately. C (s) + O2 (g) CO2 (g) ΔH1 = -394 kJ mol-1 CO (g) + ½ O2 (g) CO2 (g) ΔH2 = -283 kJ mol-1 C (s) + ½ O2 (g) CO (g) C (s) + O2 (g) CO2 (g) ΔH = ? ΔH1 = -394 kJ mol-1 CO (g) + ½ O2 (g) CO2 (g) ΔH2 = -283 kJ mol-1 C (s) + ½ O2 (g) CO (g) C (s) + O2 (g) CO2 (g) ΔH = ? ΔH1 = -394 kJ mol-1 CO (g) + ½ O2 (g) CO2 (g) ΔH2 = -283 kJ mol-1 ...reverse… CO2 (g) CO (g) + ½ O2 (g) ΔH2 = +283 kJ mol-1 C (s) + ½ O2 (g) CO (g) C (s) + O2 (g) CO2 (g) ΔH = ? ΔH1 = -394 kJ mol-1 CO2 (g) CO (g) + ½ O2 (g) ΔH2 = +283 kJ mol-1 _______________________________________ Combine, subtract CO2 and ½ O2 from both sides C (s) + ½ O2 (g) CO (g) C (s) + ½ O2 (g) CO (g) ΔH = ? Combine, subtract CO2 and ½ O2 from both sides C (s) + O2 (g) + CO2 (g) CO2 (g) + ½ O2 (g) + CO (g) ΔH1 = -394 kJ mol-1 ΔH2 = +283 kJ mol-1 ΔH = ΔH1 + ΔH2 = (-394) + (+283) = -111 kJ 5.2 Hess’s Law Applications and skills • Application of Hess’s Law to calculate enthalpy changes. • Calculation of ΔH reactions using ΔHf data. • Determination of the enthalpy change of a reaction that is the sum of multiple reactions with known enthalpy changes. • Two classics reactions to determine enthalpy changes for reaction are formation and combustion • Formation – forming compounds from their elements • Combustion – reacting substances completely with oxygen Combustion reactions: reacting with oxygen O2 Enthalpy of combustion ΔHco (kJ mol-1) – Energy released when one mole of a particular substance reacts with O2 HL stuff ΔHfo = Enthalpy of formation (SL) HL stuff Standard enthalpy of formation • ΔHfo, the enthalpy change when 1 mole of a compound is formed from its elements in their standard states at 298K and 100kPa. • Elements have a ΔHfo value of zero! Standard enthalpy of formation • C (s) + 2H2 (g) CH4 (g) ΔHfo = -74 kJ mol-1 • 2C(s) + 3H2(g) + ½ O2(g) C2H5OH ΔHfo = -278 kJ mol-1 Enthalpy of Formation and Hess’s Law • The enthalpy change for a reaction can be calculated using Hess’s Law and enthalpy of formations. ΔHo = Σ(ΔHfo products) - Σ(ΔHfo reactants) Hess’ Law: Details • You can always reverse the direction of a reaction when making a combined reaction. • When you do this, the sign of DH changes. N2(g) + 2O2(g) 2NO2(g) DH = 68 kJ 2NO2(g) N2(g) + 2O2(g) DH = -68 kJ Hess’ Law: Details • The magnitude of DH is directly proportional to the quantities involved • E.g. if you need to multiply the number of moles, you also multiply the magnitude of DH N2(g) + 2O2(g) 2NO2(g) 2N2(g) + 4O2(g) 4NO2(g) DH = 68 kJ DH = 136 kJ - ΔHf CH4 ΔHf CO2 - ΔHf O2 2 x ΔHf H2O ΔHo = Σ(ΔHfo products) - Σ(ΔHfo reactants) CH4 (g) + O2 (g) CO2 (g) + 2 H2O (l) ΔHo = Σ(ΔHfo products) - Σ(ΔHfo reactants) ΔHo = (ΔHfo CO2 + 2 x ΔHfo H2O) - (ΔHfo CH4 + ΔHfo O2) = [(-394) + 2 x (-286)] – [+75 + 0] = -966 – 75 = -1041 kJ Do not mix these two up!! • ΔHo through formation (Using Table 12) ΔHo = Σ(ΔHfo products) - Σ(ΔHfo reactants) • ΔHo through bond enthalpy (Using Table 13) ΔHo = Σ(BE reactants) – Σ(BE products) CH4(g) + 2 O2(g) CO2(g) + 2 H2O(l) ΔHθ = ? CH4(g) + 2 O2(g) CO2(g) + 2 H2O(l) ΔHθ = -808 kJ mol-1 Discrepancies in the two methods CH4 (g) + O2 (g) CO2 (g) + 2 H2O (l) • Through enthalpy of formation – ΔHθ = -1041 kJ mol-1 • Through bond enthalpy – ΔHθ = -808 kJ mol-1 • Bond enthalpy assumes all substance in gaseous state, does not take into account energy needed to change state. Do not mix these two up!! • ΔHo through formation (Using Table 12) ΔHo = Σ(ΔHfo products) - Σ(ΔHfo reactants) • ΔHo through bond enthalpy (Using Table 13) ΔHo = Σ(BE reactants) – Σ(BE products) Read and practice: page 179 - 184