幻灯片 1
advertisement

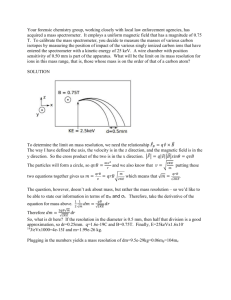
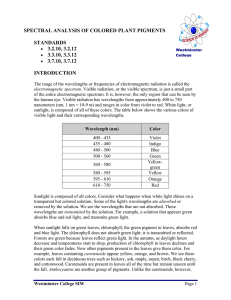
Experiment 1 Introduction to basic technique of biochemistry experiment (glass pipette, centrifuge, spectrophotometry) Rule of biochemistry’s lab 1.Wear your write gowns. 2.Each group has a set of apparatus. 3.If you break your apparatus, you must pay for it. 4.Four students will be on duty each time. 5. Hand in your report after experiment. 6.Signature your names after experiment . How to write the report? ⑴Title of experiment, Name, Date of experiment. ⑵Objectives ⑶Principle ⑷Material and reagents ⑸Procedure ⑹Results and calculations: The section will contain the calculation formula and results. ⑺Discussion Discussion is very important a. Conclusions b. Significance of your results c. Was the original objective achieved? (if not, why not?) d. Discussion of possible errors Part Ⅰ:Pipette A pipette is used to measure small amounts of solution very accurately, A pipet bulb is used to draw solution into the pipet。 ⑴Recognition ⑵Select ⑶Draw solution ⑷Release Exercise Practice this with water until you are able to use the pipette and bulb consistently and accurately. Part Ⅱ:Centrifugation The theoretical basis of this technique is the effect of gravity on particles (including macromolecules) in suspension. Two particles of different masses will settle in a tube at different rates in response to gravity. Centrifugal force (measured as as xg, gravity) is used to increase this settling rate in an instrument called a centrifuge. Simple centrifuges are used in biology and biochemistry for isolating and separating biocompounds on the basis of molecular weight。 Balance the load in the opposite slot with the same volume of fluid。 centrifuge caution ①The machine should be placed on a stable and firm desk. ②Keep the centrifugation tube balanced and symmetric. ③After turning on machine, increase the rotation speed little by little. ④Regulate the speed you wanted and record the time. ⑤At the end of centrifugation,turn down the speed little by little. Exercise Take 1ml blood serum into a tube,then add 10 drops of 10% acetate chlorine. Centrifuge the sample at the speed of 3000r/min for 5 minutes. Observe the liquid in the tube and record the change happened after centrifugation. Part Ⅲ:Spectrophotometry (spectrometer) A spectrometer measures the amount of visible light absorbed by a colored solution. This can be read as Absorbance or % Transmittance. Principle: According to Lambert-Beer's Law, the concentration of a sample could be calculated: A(Absorbance)=K × C (concentration) A(sample)/A(standard) = C(sample)/C(standard) Fingure 1. The main elements of spectrometer 1. 721 spectrometer Procedure: ① Setting up the spectrometer,Turn on the power of the instrument , Allow 10 minutes for warming up. Set the wavelength to the desired value using the knob on the left. ② Calibrating the spectrometer Add solution to the cuvette. Wiping the cuvette with a dry Kimwipe to remove drops of solution or finger prints. with empty, opened sample compartment, turn the knob to obtain a reading of 0% T,Close the cover. Adjust the knob to obtain a reading of T= 100% (A=0) ③Using the spectrometer Lining up the mark on the cuvette in the sample compartment. Insert sample cuvette and record the value of A on the scale. 2.722 spectrometer ( digital spectrometer) Procedure: ①Setting up the spectrometer Turn on the power of the instrument . Allow 10 minutes for warming up. Set the wavelength to the desired value using the nm and nm buttons. The wavelength is displayed on the LCD.. ②Calibrating the spectrometer. Fill a cuvette with your blank solvent and dry the outside of the cuvette carefully. Insert your blank, opened cuvette in the sample compartment. Press the A/T/C button to select Transmittance mode and set the transmittance of the blank to zero. Close the cover. Press 0 ABS/100% T to set the absorbance of the blank to zero. ③Using the spectrometer Lining up the mark on the cuvette in the sample compartment. Insert sample cuvette and the absorbance value of the sample will now be shown on the LCD. Exercise Use the spectrometer to measure A500nm of 0.5%, 1.0%, 1.5%, 2.0% cobalt sulphate, as well as an unknown concentration of it. Adjust the wavelength to 500nm. Draw a standard curve with the X axis representing respectively the given concentration of the liquid and the Y axis the absorbance of the four tubes. Obtain the concentration of the unknown cobalt sulphate with two methods.