Measurement Notes
advertisement

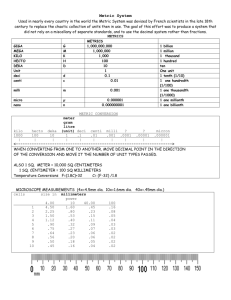
It is important that scientists can communicate with each other. Measurements Measurement: comparison of some aspect (dimensions, quantity, capacity) of an objector phenomenon with a . The makes the measurement All measurements need a and . for comparison. Use in measurements: kilo = 1000 deci = 1/10 hecto = 100 centi = 1/100 deka = 10 milli = 1/1000 Scientists use metric units to measure , density, and , volume, mass, . It is easier to convert meters to centimeters or millimeters than to go from yards to feet or inches. All you have to do with the metric system is move decimal places. Precision: gives a better representation of the value. 2.1cm is more precise it is measured to the nearest tenth of a cm 2cm is only measured to the nearest cm Length Metric standard for length is the (m) 1 m = 100 cm = 1000 mm (1) m (1/100) m (1/1000) m Whereas: 1000m = 1 km What if you wanted to measure something the size of an atom? Angstrom = one ten billionth of a meter Sometimes we are given a unit that we don’t want; we need to use a to get the unit we want. For example: Given 5m want the measurement in cm Using conversions We know that 100cm = 1 m Therefore we can set up a conversion 2km = m 5mm = m 6.3m = cm