Density Lab Packet
advertisement

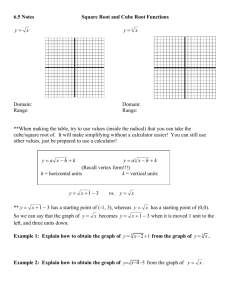
Name Per Density and Percent Error Lab Purpose: Density is the amount of mass in a given volume. It can be found by finding the mass of an object and dividing it by its volume. In this lab, the units for mass should be in grams and the units for volume should be in cubic centimeters. Using these units will give you a density in g/cm3 which is the same as g/mL. The purpose of this lab is to find the mass and volume for different materials and identify the materials’ density. The measured density will be compared to the material’s accepted density and a percent error will be found. Procedure: 1) Obtain a density kit and identify the aluminum, copper, iron, and brass cubes. 2) Using a ruler, measure the three dimensions of the aluminum cube. Make sure to measure to the nearest hundredth of a centimeter. Record your data. 3) Find the volume of the aluminum cube using the L x W x H formula and record your data using the correct number of significant digits. 4) Using the electronic balances, record the mass of the aluminum cube. The mass should have the same number of significant digits as displayed on the balance. 5) Repeat steps 2-4 for the copper, iron, and brass cubes. 6) Finally, find the percent error of you measurements based on the accepted values given in the data section of the lab. Data: Aluminum Cube L=___________ W=_____________ H=_______________ Calculation of volume: Volume of cube ____________ Mass of cube__________ Calculation of density: Density of cube____________ Copper Cube L=___________ W=_____________ H=_______________ Calculation of volume: Volume of cube ____________ Mass of cube__________ Calculation of density: Density of cube____________ Iron Cube L=___________ W=_____________ H=_______________ Calculation of volume: Volume of cube ____________ Mass of cube__________ Calculation of density: Density of cube____________ Brass Cube L=___________ W=_____________ H=_______________ Calculation of volume: Volume of cube ____________ Mass of cube__________ Calculation of density: Density of cube____________ The accepted densities for the metals are as follows: Aluminum= 2.70 g/cm3 Copper= 8.96 g/cm3 Iron= 7.87 g/cm3 Brass=8.40 g/cm3 Percent Error calculation for density of aluminum: Percent Error__________ Percent Error calculation for density of copper: Percent Error__________ Percent Error calculation for density of iron: Percent Error__________ Percent Error calculation for density of brass: Percent Error__________