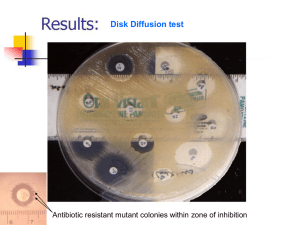
C difficile - Carolinas College of Health Sciences
advertisement

Unit VIII Anaerobes Anaerobe Terminology Terminology: • 1. Aerotolerant: ability of an anaerobic microorganism to grow in air; usually poorly, especially after initial anaerobic isolation. • 2. Glove/Gloveless Box: a device with flexible, semi-rigid or complete rigid plastic walls used for the cultivation of anaerobic bacteria. The bacteriologist must work by way of glove ports and materials that are brought into and out of the anaerobic work area via an interchange (air lock-port hole) that can be evacuated and refilled with an anaerobic atmosphere repeatedly until it is anaerobic. The interchange has one door leading into the glove box and another door to the outside. Some glove boxes have incubators within them . Also, called “anaerobic chamber” or “anaerobic cabinet”. • 3. Laked Blood: a.k.a. “hemolyzed blood” – hemolysis of red blood cells used in agar may be effected in various ways but alternate freezing and thawing is a simple method. • 4. Meconium: pasty, greenish mass in the intestines of fetuses; made up of mucus, desquamated cells and bile and such. • 5. Gas Liquid Chromatography (GLC): a method for separating substances by allowing their volatile phase to flow through a heated column with a carrier gas (usually helium) and measuring the time required to detect their presence at the distal end of the column. • 6. Pseudomembrane: necrosis of mucosal surface simulating a membrane • 7. Pseudomembranous Colitis (PMC): syndrome in the large bowel characterized by a layer of necrotic tissue and dead inflammatory cells often caused by the toxin of Clostridium difficile. • 8. Sulfur granules: small colonies of organisms with surrounding club-like material; yellowbrown; resembles grain of sulfur. ANAEROBES • Obligate anaerobe - an organism that can not grow in the presence of oxygen. The anaerobic bacteria are subdivided according to their requirement for O2. Obligate anaerobes are killed by exposure to atmospheric O2 for 10 minutes or longer. • Facultative anaerobe - an organism that can use aerobic or anaerobic respiration or anaerobic fermentation system, depending on the availability of oxygen. • Microaerophilic – organisms that will not grow in an aerobic incubator on solid media, and only minimally under anaerobic conditions. However, they will grow in minimal oxygen (5% O2). Superoxide dismutase (SOD) is produced by many anaerobes, which catalyzes the conversion of superoxide radicals to less toxic H2O2 and molecular O2. Eh POTENTIAL: (oxidation-reduction potential) • Eh is a measure of the tendency of a system to give up electrons • expressed as millivolts • as Eh potential is lowered by 60 mv, the pH increases by 1 • pH affects the oxidation-reduction potential (Eh) of media for anaerobic bacteria more than oxygen • ** Eh is most affected by pH and is expressed at pH 7.0. In cultivating anaerobic bacteria, reducing agents such as thioglycollate and L-cysteine are added to anaerobic transport and culture media in order to maintain a low Eh. Certain anaerobes will not grow in the media above a specific critical Eh level • most anaerobic media is at - 150 mv • actively growing cultures are Eh = -250 mv • if Eh is low enough, anaerobes may grow in the presence of oxygen concentrations which would ordinarily be inhibitory • when surfaces are damaged or necrosis occurs then the Eh potential is lowered CLINICAL SIGNS OF ANAEROBIC INFECTIONS: • foul smelling discharge • infection in proximity to a mucosal surface • necrotic tissue, gangrene; pseudomembrane formation • gas in tissues or discharge or presence of “sulfur granules” in discharges • endocarditis with negative blood cultures • infection associated with malignancy or tissue destruction • infection related to the use of aminoglycoside drug therapy (oral, parenteral or topical) • infection following human bites, septic abortion or gastrointestinal surgery Anaerobic Specimens UNACCEPTABLE Specimens for Anaerobic Culture • - throat, ear, NP swabs, sputum, tracheostomy • - feces or rectal swabs • - voided or cath urine • - vaginal or cervical swabs • - urethra • - superficial wounds/abscesses • - GI or abdominal wounds contaminated with feces ACCEPTABLE Specimens for Anaerobic Culture • - transtracheal aspirate or direct lung puncture • - feces only requested for Clostridium difficile • - suprapubic bladder tap • - culdocentesis aspirate, placenta, Bartholin’s gland • - prostatic or seminal fluid • - abscess aspirates or deep wounds collected to avoid skin flora Specimen Collection and Handling SPECIMENS • Proper Collection: • 1. Specimens for anaerobe culture must be sent in the proper anaerobic transport tubes which may include: “Port-a-Cult” semi-solid agar tubes, COPAN tubes, a closed syringe (best) or a gassed out tube. • 2. Tissue may be cultured anaerobically but it should be ground inside the chamber to minimize the amount of oxygen forced into the tissue during the grinding process. • 3. When anaerobic culture on CSF is requested, the culture may be set up from the regular CSF collection tube as long as it is done immediately upon receipt of the specimen. • 4. If a valid specimen for anaerobic culture is received in an inadequate transport device and cannot be recollected, consult with the laboratory supervisor or the Dept. Head. • 5. Certain specimens are unsuitable for anaerobic culture. These include specimens coming from body sites normally inhabited with normal anaerobic flora. (See acceptable versus unacceptable specimens). • 6. On vaginal and cervical cultures (from surgery which the doctor is persistent about culturing) which yield a light or moderate growth of anaerobes mixed with aerobic flora, note the presence of the anaerobe on the worksheet, but do not identify. Send the report out as "No Significant Anaerobes Isolated" or “Mixed Anaerobic Flora”. Identify (either by site or Rapid ID ANA) only a heavy or pure growth. NO SENSI. Specimen Collection and Handling: Specimens must be collected and transported anaerobically • • • • • ** Needle aspiration - is the best method of collection when possible. The specimen should not sit in the syringe for more than 30 minutes due to the passage of air through the plastic ** Gassed-Out tubes - are appropriate (gassed with CO2); Resazurin is often used as the indicator for anaerobic systems. colorless = Eh -43 mv pink = increased oxygen ** Inoculating loop should be platinum or plastic NOT nichrome due to agar surface tension breakdown Inoculation and Incubation: • • • • • • • • • • • • 1. CDC Anaerobic Blood Plate containing Vitamin K1 Allows for the growth of all facultative organisms (aerobic and anaerobic) including gram positive and gram negative rods, gram positive and gram negative cocci. 2. Laked Blood plate with Kanamycin and Vancomycin For selective isolation of obligate anaerobic gram negative bacteria (Bacteroides, Prevotella, Fusobacterium and Veillonella species). Essentially inhibits all other bacteria encountered in clinical specimens including anaerobic gram positive bacteria. 3. Anaerobic Blood plate with Phenyl Ethyl Alcohol For isolating obligate gram negative and gram positive anaerobic bacteria. The anaerobic formulation (CDC blood agar with PEA) inhibits facultative gram negative bacteria such as Proteus species and other members of the Family Enterobacteriaceae but will support gram positive aerobes and anaerobes and gram negative obligate anaerobes. 4. 5% TSA (tryptic soy agar) Sheep Blood Agar plate Supports the growth of most aerobic gram negative and gram positive organisms. This plate is incubated aerobically at 35OC and is used for comparison with the anaerobic BAP. 5. Gram stain slides (2) Usually performed and reported by the Exudate Bench technologist. All media are inoculated and incubated inside the anaerobe chamber at 35-37oC with the exception of the TSA blood plate. This is incubated aerobically in CO2 for comparison. Any organisms recovered from the aerobic plate that were not seen on the corresponding exudate culture are then sent back to the exudate bench for complete work up and reporting. All cultures are held for a minimum of 5 days. Any specimen with a request for isolation of Actinomyces species will be held for 7 days. Currently, specimens of vaginal or cervical swabs will be held for 3 days. Other Media: • 1. Egg Yolk Agar - recommended for determining the production of lecithinase and lipase that aids in the identification of the Clostridium species • Lecithinase + : colonies with opaque zone around colony (essentially a surface phenomenon); reaction occurs within the agar Lecithinase • Lipase www.asm.org Lipase + : colonies are covered with an iridescent, multicolored sheen, sometimes described as resembling the appearance of “gasoline on water” or “mother of pearl”. This sheen may also appear on the surface of the agar in a narrow zone around the colony ** Modified McClung or Neomycin egg yolk agar is used to determine the presence of lecithinase activity, which causes an insoluble, opaque, whitish precipitate within the agar. Lipase activity is indicated by an iridescent sheen or pearly layer on the surface of the colony or agar. • • • • • • 2. Bacteroides Bile Esculin Agar - used for the determination of Bacteroides fragilis which is tolerant of increased bile concentrations 3. Litmus milk - a differential medium on the basis of lactose fermentation caseolysis and casein coagulating properties. Skim milk and litmus are used in preparation. Litmus has the advantage of being readily reduced by certain bacteria Reactions: 1. Acid: pink color indicates an acid reaction caused by fermentation of lactose. 2. Alkaline: a purple/blue color indicates no fermentation of lactose. 3. Reduction: a white color results when litmus serves as an electron acceptor and is reduced to its leuco base. 4. Coagulation: Clotting is caused by precipitation of the casein by the acid produced from lactose. It may also be caused by the conversion of casein to paracasein by the enzyme rennin. 5. Peptonization: Dissolution of the clot indicates digestion of the curd or milk proteins by proteolytic enzymes. Anaerobic Respiration Anaerobic Respiration: • The mechanism of electron transport and oxidative phosphorylation is basically the same in anaerobes as in aerobes with minor variations: • The final electron acceptor is not oxygen but is some other inorganic molecule, such as nitrate (NO3), sulfate (SO4), nitrite (NO2), or an organic molecule, such as fumarate. • The terminal enzyme component in electron transport in anaerobes is not cytochrome oxidase but enzymes such as reductases that catalyze the reduction of nitrate to nitrite, sulfate to hydrogen sulfide and nitrite to molecular nitrogen. One of the most frequently used inorganic electron acceptors is nitrate, where as fumarate is a more common organic electron acceptor. The synthesis of terminal reductases is repressed and their activities inhibited by oxygen. This mechanism is especially important in facultative anaerobes that switch from anaerobic to aerobic growth when oxygen becomes available. • Less energy is realized during electron transport under anaerobic conditions. The difference in the oxidation-reduction potential between the donor and acceptor (for example, between NADH and nitrate) is not sufficient to generate three molecules of ATP. Depending on the donor and acceptor, the number of ATP molecules generated during electron transport under anaerobic conditions is usually between one and two. The rate of growth of anaerobes is therefore, considerably slower than that of aerobes. A facultative anaerobe, for example, under anaerobic conditions might require 48 hours of optimal growth conditions to produce a population of cells that under aerobic conditions should be established in 16 hours or less. Oxygen and Anaerobic Bacteria: • 1. Obligate anaerobes are not able to grow in the presence of air, but they do show different sensitivities to oxygen. Some anaerobic bacterial populations are almost completely killed when exposed to oxygen for 3 minutes, whereas others remain viable even though they cannot grow. Indifference to oxygen by some anaerobes is the result of their inability to reduce oxygen; therefore toxic intermediates are not produced. In other anaerobes the concentration of oxygen is the critical factor. Obligate anaerobes are often found associated with facultative anaerobes in areas where oxygen is present. Facultative anaerobes can remove oxygen from the environment by using it in aerobic respiration, thus making the environment suitable for the growth of obligate anaerobes. Studies with oxygen-tolerant anaerobes have demonstrated that a positive correlation exists between oxygen tolerance and the content of superoxide dismutase in the cell. It is now clear that microbial cells whether aerobes or anaerobes rely on scavenger systems such as catalase, peroxidase, and superoxide dismutase to remove toxic intermediates produced by the reduction of oxygen. Work Up Protocol WORK-UP PROTOCOL • A. Identification • Since the type of specimen accepted for anaerobic culture is carefully defined, any amount of any anaerobe should be identified. The technologist must remember that anaerobic infections are rarely caused by one organism; the usual situation is isolation of at least two or three different anaerobes. Blood cultures and CSF may however, yield only one organism. • B. Susceptibility testing: • Susceptibility testing will be performed on any number of pathogens in any amount from any normally sterile body site, bone specimens and brain abscesses. (See E Test Procedure). Occasionally, if a pure or predominant anaerobic organism is recovered from a non-sterile body site, then discretion is used by the MLS reading the plates. • C. Direct Gram Stains: • Gram stains prepared from the specimens will be performed on all cultures. The results will be reported for both the aerobic culture (sent to Exudate bench) and the anaerobic culture by the Exudate bench technologist. Occasionally there are instances when the gram stain shows organisms and the culture is negative. • D. Beta Lactamase Testing: • A beta-lactamase test will be performed on all anaerobic gram negative rods and anaerobic spore-forming gram positive rods. The results of the test will be reported. (Positive versus Negative). Gram Stain Procedure for Anaerobes GRAM STAIN PROCEDURE FOR ANAEROBES • Procedure: • 1. Fix the slide with methanol for about one minute. Drain. • 2. Flood slide with Crystal Violet for one minute. • 3. Using a freshly mixed bottle of Gram's Iodine (if using unstabilized iodine), wash off the Crystal Violet and flood the slide with Iodine for one minute. • 4. Decolorize quickly and gently with 95% alcohol. • 5. Wash with water. • 6. Counterstain with Basic-fuchsin for one minute. • 7. Wash with water. • 8. Air dry. • The cell wall of anaerobic organisms is much more fragile than that of aerobic organisms, and may be destroyed by heat fixing. Fixing the slide with methanol is more protective of the cell wall. The mordant ability of Gram's Iodine declines rapidly once the solution is prepared. The gram positive characteristic of anaerobes is often difficult to demonstrate. For these two reasons, the anaerobe Gram stain must be performed with a freshly mixed bottle of Gram's Iodine. The decolorizer used for routine gram stains is too harsh and easily over decolorizes most anaerobes thus using just 95% alcohol yields more accurate results. The interpretation of Gram stains on anaerobes is often difficult to interpret. As a general rule, an organism which shows even small areas which stain Gram positive should be considered to be gram positive, as it is unlikely that the smear has been under-decolorized. The Gram Positive, Spore-Forming Rods CLOSTRIDIUM • - large, gram positive rods • - obligate anaerobes • - spores cause cell walls to swell • - found mainly in soil and intestines of man and animals • - discovered in 1877 by Pasteur CLOSTRIDIUM PERFRINGENS - a. k. a. "WELCH'S BACILLUS" • isolated by Welch in the early 1900's • most common cause of gas gangrene (myonecrosis) • most common isolate of the clostridium • encapsulated and non-motile; box-car shaped bacilli seen on gram stain; may stain gram-negative • spore production is not usually seen; if present, centrally located • only Clostridium to have a double zone of hemolysis on anaerobic blood agar (an inner zone of complete beta hemolysis and an outer zone of partial hemolysis) • causes stormy fermentation of litmus milk: media changes from blue to pink, forms a clot, the clot explodes and is digested to a whey color • • “Reverse camp test" positive “Unknown” Clostridium Postive result shows “arrow-head” hemolysis www.microbelibrary.org www.asm.org Known ATCC Strep agalactiae www.microbelibrary.org Clostridium perfringens continued: • Nagler reaction positive which is recommended for presumptive rapid ID: This test employs an EYA plate (inoculated at set up of specimen) and a reagent known as C. perfringens type A antitoxin. This reagent inhibits the lecithinase reaction produced by C. perfringens thus presumptively identifying C. perfringens. (This test is also positive for 3 other Clostridia as well).. • on egg yolk agar it is lecithinase positive, lipase negative • Clostridium perfringens is normal in GI tract; gets in through perforation in GI tract or wound. Evidence of toxemia precedes gas development. Anemia occurs due to the hemolytic action of the toxin. Organism ferments sugars in muscle tissue to form gas. The enzyme hyaluronidase helps spread the gangrene. Jaundice results from RBC destruction. TREATMENT: • - antibiotics sprinkled directly into the wound • - hyperbaric oxygen chamber • - remove the necrotic tissue (amputation) CLOSTRIDIUM TETANI - causes "lock jaw" • grows well on anaerobic blood agar • colonies have a compact grayish center which flattens out to be a fine, gray filamentous network described as "maple leaf or fern-like" • small zone of hemolysis; they are motile organisms • litmus milk negative, lecithinase and lipase negative • spores are terminal and appear "racket-shaped" or "drumstick" shaped • the organisms are introduced by spore into deep, lacerated wounds in which there has been considerable tissue destruction (compound fractures, gunshot wounds, etc..) The organism has limited invasive power and there is only a localized infection with no systemic results until there has been sufficient time (5 days to a www.textbookofbacteriology.net week) for the vegetative forms to produce the exotoxin. The toxin "tetanospasmin" becomes bound to gangliosides within the central nervous system acting like strychnine, suppressing motor neuron activity (disrupts nerve impulses to muscles). This results in a spastic type of paralysis with continuous muscular spasms leading to trismus (stiffness of the muscles of the neck and jaw.a.k.a.lockjaw), and difficulty in swallowing. The damage to the tissues and nerve endings is irreversible resulting in brain damage. The prognosis depends upon incubation time with an 80% mortality rate. If left untreated, spasms continue to be triggered by even minor stimuli, leading to exhaustion and, eventually death due to asphyxiation (respiratory failure) during a muscle spasm. Clostridium tetani continued: TREATMENT: • antitoxin usually coupled with one or more antibiotics. The antitoxin is prepared from horses. Recovery from the disease does not give lasting immunity • excellent immunity is gained from the toxoid vaccine, though revaccination is recommended every 10 years. ** However, if one acquires a major wound infection with Clostridium tetani 5 years out from their vaccination, then they should receive a “booster” of the tetanus toxoid as a precautionary measure. • Penicillin is the drug of choice for the vegetative cell. CLOSTRIDIUM BOTULINUM - causes "food poisoning" • motile, forms subterminal spores • grows best at 25 C, slightly hemolytic • produces the most powerful toxin known to man • this exotoxin is not destroyed by the enzymes in the GI tract • this organism is most commonly found in home canned vegetables, home-cured meats such as ham, fermented fish and other preserved foods (protein). The exotoxin is heat labile. • Symptoms may occur as soon as 2 hours due to toxin absorbed in the small intestine. (There are 6 antigenic types with A and B being the most common. The tuna fish scare of the 1960's was type E). • The condition is caused by ingestion of the preformed toxin. • the botulinal toxin attaches to individual motor nerve terminals preventing acetylcholine release at the nerve endings . Death occurs due to a flaccid type of paralysis of the respiratory muscles. • ** persons who have eaten food suspected of containing botulinum toxin should be purged with cathartics (laxatives), have their stomach pumped AND be given enemas*** • The antitoxin is available from CDC. • on egg yolk agar it is lecithinase - and lipase + • recovery gives lasting immunity • the toxin is destroyed at 1000C for 10 minutes • confirmation of botulism is made by demonstration of the toxin in serum, gastric, or stool specimens • ** Infant botulism is the most frequent form occurring in the United States. Epidemiological studies have demonstrated that infant botulism results from the ingestion of spores via breastfeeding or exposure to honey. Preformed toxin has not been detected in food or liquids taken by the infants. C. botulinum spores germinate, colonize in the gut (colon) of the infant and produces the neurotoxin in situ. Clostridium species continued: CLOSTRIDIUM RAMOSUM • second most commonly encountered Clostridium in clinical infection • often appears gram negative in smears made from culture • very resistant to antibiotics CLOSTRIDIUM SEPTICUM • strong association between bacteremia with this organism and malignancy. Clostridium species continued: CLOSTRIDIUM DIFFICILE: • Clostridium difficile has become well recognized as a major nosocomial pathogen associated with antibiotic use within hospitals and communities. After treatment with antibiotics, many patients develop gastrointestinal problems ranging from mild diarrhea to severe pseudomembranous colitis. Many cases of the milder forms of GI illness and most cases of pseudomembranous colitis are caused by Clostridium difficile. This organism is an opportunistic anaerobic bacterium that grows in the intestine once the normal flora has been altered by the antibiotic. The disease results from the toxins that the organism produces. C. difficile disease is responsible for substantial strain on the hospital system by increasing patients’ length of stay and increasing costs. Diagnosis should be considered if patients have diarrhea and a history of antibiotic exposure. Toxin detection assays remain the first step in proving infection; however, sigmoidoscopy should be considered if patients have possible alternative diagnoses or if assay results are negative but clinical suspicion is high. Prevention of infection, by prudent restriction of antibiotic use, strict adherence to hand washing, disinfection, and infection control precautions for infected patients, is crucial. Antibiotic therapy for first-episode infections is well established; however, treatment of relapsed cases is less well defined and multiple relapses could require lengthy therapy. www.nlm.nih.gov Clostridium difficile continued: Risk Factors • Antibiotic exposure is the single most important risk factor, contributing to altered gut flora and allowing overgrowth of C. difficile. Disease can occur after a few days of antibiotic therapy or up to 2 months after use. Clindamycin has been identified as the main culprit. Other antibiotics commonly found to predispose patients to infection include the second- and third-generation cephalosporins and penicillins. Less common agents include the quinolones, sulfonamides, and metronidazole. In addition to antibiotics, cancer chemotherapy is also a risk factor as well as age and severity of underlying illness. • Research has shown the organism can be carried asymptomatically or acquired nosocomially. Patients treated with antibiotics are then at risk for developing Clostridium difficile associated diarrheal disease (CDAD) which, if left untreated may progress into pseudomembranous colitis (PMC), toxic megacolon and death. The rate of nosocomial infections will vary with the institution, ward, housekeeping procedures, recent epidemiology and patient population. Facilities with large in-patient population, chemotherapy wards or long term patient care facilities need to be particularly aware of the problems related to nosocomial acquisition of Clostridium difficile. Two distinct groups which can harbor toxigenic Clostridium difficile asymptomatically are infants and cystic fibrosis patients. A picture of pseudomembranes and lesions within the colon. www.cfpc.ca Clostridium species continued: Diagnosis • The clinical symptoms associated with the disease are believed to be primarily due to toxin A which is a tissue-damaging enterotoxin. Clostridium difficile also produces a second toxin, designated toxin B. Essentially all strains either produce both toxins or neither toxin. • Clostridium difficile strains that produce high levels of toxin A also produces high levels of toxin B. Likewise, strains that produce low levels of toxin A produce low levels of toxin B, indicating that the toxin production may be regulated similarly. Tests that detect either or both toxins are best used as diagnostic aids. There is some evidence suggesting that toxins A and B act synergistically and that the initial tissue damage caused by toxin A allows toxin B to exert its toxicity. Therefore, although toxin A is believed to cause most of the clinical signs, toxin B also may play an important role in the disease. Clostridium species continued: Tests for Detection • The C. difficile TOX A/B TEST that we use is an enzyme immunoassay (EIA) for the detection of toxins A and B produced by toxigenic strains of Clostridium difficile. It can be used to detect toxins A and B in fecal specimens from persons suspected of having C. difficile disease. The diagnosis of AAPMC is dependent upon the identification of the characteristic lesions by sigmoidoscopy or colonoscopy exam , recovery of the organism by culture or detection of its toxins present in the stool. This test that we perform is to be used as an aid in the diagnosis of C. difficile disease and results should be considered in conjunction with the patient history. • If test results are reported negative, sending one or two additional stools on subsequent days could improve sensitivity. In addition, if only toxin A alone is tested, diarrhea secondary to toxin A–negative—toxin B–positive strains will be missed. Principle of the Test: • In the assay, an aliquot of a fecal specimen is emulsified in the diluent and the diluted specimen is then transferred to the microtiter well containing the detecting antibody. If toxins A and B are present in the specimen, they will bind to the detecting antibody and the immobilized polyclonal antibody during the incubation phase. Any unbound material is removed during the washing steps. Following the addition of substrate, a color is detected due to the enzyme -antibody-antigen complexes that form in the presence of toxin. Any shade of yellow is a positive test result indicating toxin A and/or toxin B are present in the stool. Clostridium species continued: Other Tests for Detection: • 1. Cytotoxicity assay/Tissue Culture: This test is positive when stool filtrate inoculated onto cell culture monolayer demonstrates cytopathic effect due to the presence of C difficile toxin. It remains the • criterion standard of detection. Sensitivity and specificity are 67% to 100% and 85% to 100%, • respectively. The test, however, is relatively expensive; it requires 24 to 48 hours for completion; and • inactivation of toxins during transport, through dilution of sample, and through age of the cell line • gives rise to false-negative results. • 2. Latex agglutination: based assays recognize the enzyme glutamate dehydrogenase but lack sensitivity. These latex agglutination tests detect the antigens of C. difficile in the supernatant of stool specimens. • The antigens and toxins are soluble and are found in the supernatant solution. Agglutination is • observed for a positive test. • 3. Stool culture: for C difficile is sensitive, but will also detect nontoxigenic strains. A special media is used to grow C. difficile that contains cycloserie, cefoxitin, fructose and egg yolk (CCFYA). The • colonies have a “mustard-yellow” color and will fluoresce under a UV light. • 4. Genetic analysis: via polymerase chain reaction is available, but confined to research settings. • 5. Endoscopy: should be reserved for when a patient’s condition necessitates rapid diagnosis or when other diagnoses are being considered. Colonoscopy can detect cases not apparent during sigmoidoscopy because C difficile infection can involve the transverse colon or ascending colon cecum without evidence of involvement of the more distal segments of the colon. • False-negative results can occur in any of these assays, so if the clinical suspicion is high and the assay is negative, either the test should be repeated or a sigmoidoscopy or colonoscopy should be considered. This is critical, for we have seen several cases where the initial assay is negative and empiric C difficile therapy is stopped or the diagnostic possibility of C difficile is abandoned, resulting in delayed treatment and unfortunately serious adverse outcomes for some patients. Clostridium species continued: Treatment: • Once C. difficile diarrhea is diagnosed, inciting antibiotic therapy must be discontinued (if possible). Anti-diarrheal agents should be avoided and narcotics minimized in order to prevent colonic stasis and decreased toxin clearance. Volume resuscitation is necessary for dehydrated patients. Specific treatment involves antibiotic regimens directed at C. difficile. • Metronidazole. Metronidazole is recommended as first-line therapy for .C difficile diarrhea in non-pregnant adults. It is inexpensive and generally well tolerated; side effects may include a metallic taste, nausea, peripheral neuropathy, and disulfiram effect. • Vancomycin. Oral Vancomycin is not well absorbed from the gastrointestinal tract and has an intracolic effect. Vancomycin is second-line therapy and, because of its cost and the need to prevent development of vancomycin resistance in species such as Enterococcus, is reserved for patients whose treatment with metronidazole fails to provide benefit. Fluid Replacement: • • Electrolyte replacement such as Pedialyte (Each liter of Pedialyte © provides sodium 45 mEq, potassium 20 mEq, chloride 35 mEq, citrate 30 mEq, and dextrose 25 g) Complications: Some major complications of pseudomembranous colitis are perforation of the colon, dehydration with electrolyte imbalance, and toxic megacolon, which can be a life threatening complication of other intestinal conditions. It is characterized by a very inflated colon, abdominal distention, and sometimes fever, abdominal pain, or shock. Non-Spore Forming Gram Positive Rods ACTINOMYCES - causative agent of “Actinomycosis” • Actinomycosis: a chronic suppurative or granulomatous infection characterized by the • formation of abscesses, multiple draining sinuses, and the appearance of tangled mycelial masses, or • "granules" (sulfur granules: small colonies of bacteria) in the discharges (pus) or in tissue sections. • most common species is A. israelii • gram positive rods often branching and beaded • normal flora in the upper respiratory tract, oral cavity, bowel and the vagina; can be an opportunistic pathogen • can be isolated from carious teeth and tonsillar crypts in the absence of infection. Gram stain of branching, gram positive rods www.textbookofbacteriology.net Actinomyces continued: 1. Cervicofacial actinomycosis is an infection of the jaw and tissues of the face and neck, characterized by swelling and hardness (“lumpy jaw”) with the formation of multiple abscesses which eventually break down to form draining sinuses. The sanguinopurulent drainage from the sinuses usually contains macroscopic granules of A. israelii. Such infections follow tooth extractions or other operative procedures in the mouth, and the disease progresses by slow and direct extension of the infection through the tissues. “Lumpy jaw” • • • • • • • • • • www.articleroad.com 2. Thoracic actinomycosis is an infection of the lungs and thoracic cage. Until extension through the thoracic skin results in multiple draining sinuses, the diagnosis is often not suspected. Infection usually is confined to the hilar region and the base of the lungs. Direct extension through the tissue to the lung surface and pleura may result in an inflammatory process, with pleural thickening, empyema, and osteomyelitis of the ribs. Hematologic spread from a primary lung infection can result in the formation of foci in tissues, such as the liver, kidneys, and brain. 3. Abdominal actinomycosis originates in the region of the cecum and may stimulate acute or subacute appendicitis. Infection by A. israelii should be expected when an apendectomy wound fails to heal and irregular tender masses appear in the abdomen. - typically produces a characteristic white, heaped, rough lobate colony resembling a "molar tooth" - a large number of cases of Actinomycosis is associated with the use of intrauterine devices (IUD) - pus from lesions often contain "sulfur granules" - slow grower; hold all specimens for 7 days; may grow micro-aerophilic but better anaerobically - thio broth has "bread crumb" appearance or tight aggregates - produces major acetic, succinic and lactic acid in GLC - most Actinomyces are nitrate positive and all are indole negative - Penicillin G is the drug of choice; Metronidazole is relatively inactive Actinomyces continued: • • • • • • • • • 3. Abdominal actinomycosis originates in the region of the cecum and may stimulate acute or subacute appendicitis. Infection by A. israelii should be expected when an apendectomy wound fails to heal and irregular tender masses appear in the abdomen. - typically produces a characteristic white, heaped, rough lobate colony resembling a "molar tooth" - a large number of cases of Actinomycosis is associated with the use of intrauterine devices (IUD) - pus from lesions often contain "sulfur granules" - slow grower; hold all specimens for 7 days; may grow micro-aerophilic but better anaerobically - thio broth has "bread crumb" appearance or tight aggregates - produces major acetic, succinic and lactic acid in GLC - most Actinomyces are nitrate positive and all are indole negative - Penicillin G is the drug of choice; Metronidazole is relatively inactive “molar tooth colonies” www.microblog.me.uk Abdominal actinomycotic abscess www.carene.org www.carene.org Non-Spore Forming, Gram Positive Rods • • • • • • • • BIFIDOBACTERIUM - normal flora in the GI tract and oral cavity - rarely isolated from clinical material - most diagnostic feature is their various shapes, with pronounced clubbing at both ends (“dog bones”) or bifurcated ends - B. dentium, species of significance - like Lactobacillus, they are acidophilic and grow best on low pH agar - produce major amounts of acetic and lactic acid • • • • • • • • • • • • • EUBACTERIUM - normal flora in the GI tract and oral cavity, dental caries or plaque - E. lentum now classified as Eggerthella lenta - E. aerofaciens now classified as Collinsella aerofaciens - not particularly pathogenic - gram stain appearance varies; mostly diphtheroid like - colonies are non-descriptive; raised to convex and transparent to translucent - ID by default; any anaerobic gram positive rod that does not fit any of the other genera is classified as Eubacterium - catalase negative; indole negative, nitrate positive; most are non-motile - can be presumptively identified on the basis of arginine stimulation in broth culture and nitrate reduction - produce acetic and butyric acids Non-Spore Forming, Gram Positive Rods PROPIONIBACTERIUM • - referred to as the "anaerobic diphtheroid" • - colonies are small and white to gray-white with older colonies often yellow • - aerotolerant but grow best anaerobically • - P. acnes, most often recovered • - presumptive ID can be made by catalase positive, spot indole positive and diphtheroid like GPR see on • gram stain • - normal flora of the skin; sometimes GI tract, upper respiratory tract and uro-genital tract • - often contaminants of blood cultures and sterile body fluids due to improper decontamination of the • skin prior to obtaining specimen • - P. acnes has been implicated in infections related to heart valves and prosthetic devices which may • lead to osteomyelitis, bacteremia, endocarditis and meningitis • - P. acnes produces a lipase that frees long chain fatty acids in the skin that may be irritating enough to • contribute to the formation of acne pustules • - produces major amounts of propionic and acetic acids • - very sensitive to Penicillin and like most aerotolerant anaerobic gram positive bacilli, resistant to • metronidazole www.microbewiki.kenyon.edu Non-Spore Forming, Gram Positive Rods • LACTOBACILLUS ACIDOPHILUS • - also known as "Döderlein’s Bacillus" • - prominent as normal flora in the vagina and colon and found in small numbers in the mouth • - straight and uniform rods with rounded ends and may form chains • - microaerophilic but grow best anaerobically; we see this organism aerobically as small pinpoint colonies (especially from urine cultures) • - grow best on a low pH medium • - produces major amounts of propionic and acetic acids • - very sensitive to Penicillin; resistant to Metronidazole Anaerobic Cocci • • • • • • • • VEILONELLA - anaerobic gram negative diplococci - although several genera of anaerobic gram-negative cocci are found in the indigenous microflora, only Veillonella (especially species parvula) are implicated as pathogens - small, convex translucent to transparent colonies - nitrate positive, catalase positive, red fluorescence under long wave UV light - strict anaerobic cocci that occurs in pairs, short chains and irregular clumps - normal mouth flora, normal upper respiratory, GI and vaginal flora - causes disease usually associated with fractured jaws, gums or lung infections PEPTOCOCCUS - except for Peptococcus niger all pathogenic species in genus Peptococcus have been transferred to the genus Peptostreptococcus. Peptococcus niger – gram positive cocci occurring in pairs, tetrads and irregular masses - colony morphology reveals tiny, black pigmented, convex, shiny, smooth, circular, entire colonies Anaerobic Cocci continued: • • • • • • • • • • • • PEPTOSTREPTOCOCCUS - normal flora in bowel, female genital tract, oral cavity and skin - most prevalent in respiratory tract and related infections - all species are catalase negative and produce acetic acid from PYG broth on GLC - all species are susceptible to Metronidazole (Flagyl), if not susceptible then probable anaerobic Streptococcus or Staphylococcus - may act synergistically with Bacteroides or Staph aureus to produce tissue necrosis Most commonly encountered species in infections are: Ps. magnus: now called Finegoldia magna (resembles Staphylococci on gram stain) Ps. asaccharolyticus – one of two species that is indole positive Ps. indolicus – one of two species that is indole positive Ps. anaerobius – glucose positive and only species that produces isocaproic acid Ps. micros: now called Micromonas micros (GS reveals tiny, little cocci in short chains) Anaerobic Cocci STREPTOCOCCUS SPECIES • the Streptococcus species are facultative anaerobes • Streptococcus intermedius is found occasionally in our lab • Usually treat with Penicillin G, Cephalothin or Metronidazole • *** Definitive ID of the anaerobic cocci depends on gas chromatographic analysis of end products or enzymatic substrate productivity. Anaerobic Gram Negative Rods Family Bacteroideaceae • - obligate anaerobic, non-spore forming bacilli • - indigenous flora of humans primarily on mucosal surfaces • - constitute one third of the total anaerobic isolates from clinical specimens • - infection with anaerobic gram negative rod is characterized by abscess formation and tissue destruction • - most common infections include pleuropulmonary, intra-abdominal and female genital tract • - produce many enzymes that aid in the pathogenicity (collagenase, heparinase, DNase ) • - the Bacteroides Group grows well in 20% bile and are usually resistant to penicillin (thus beta-lactamase positive) Anaerobic Gram Negative Rods: Bacteroides species BACTEROIDES: • normal flora of the oropharynx, GI tract and genitourinary tract • many species are catalase positive, produce acetic and succinic acids from PYG broth on GLC • Bacteroides fragilis group forms the largest part of the gut flora • the pigmented Bacteroides reside in the oropharynx, upper respiratory tract, genitourinary and GI tract • Clindamycin, Metronidazole and Chloramphenicol are active against the Bacteroides groups Bacteroides fragilis group: • Bacteroides fragilis: is the most commonly encountered pathogen of all the anaerobic bacteria; pale staining, pleomorphic rods with rounded ends; resembles a “safety pin” • • • • • Bacteroides thetaiotaomicron: is the second most commonly encountered species of this group; - catalase positive and indole positive - usually causing infections below the diaphragm - non-pigmented and growth enhanced by bile - resistant to penicillin (and kanamycin). • - other members include: B. distasonis, B. caccae, B. uniformis, B. vulgatus, and B. ovatus Bacteroides Pigmented Saccharolytic Species: • placed in the genus Prevotella • have been implicated in head and neck infections, aspiration pneumoniae and lung abscesses; some from urogenital tract • typically produce protoporphyrin, a dark pigment that causes colonies to turn color (brown to black) with age (best seen on KVLB agar); also fluoresces brick-red on long wave UV light • ferment glucose and bile sensitive • members include: most common P. melaninogenica,(black pigment), P.intermedia & P. corporis,(dark brown to black, dry), P. bivia, P. loescheii and P. denticola • resistant to Penicillin Bacteroides Pigmented Asaccharolytic Species: • - placed in the genus Porphyromonas • - Porphyromonas is primarily isolated from dental structures and may play a major role in gingivitis and periodontitis • - mostly isolated from the oral cavity (infected root canals) • - forms tan to buff colonies that fluoresce brick-red under UV light (long wave) • - does not ferment glucose • - fails to grow on KV agar • - inhibited by bile, produces indole • - sensitive to penicillin and rifampin not kanamycin • *NOTE: The pigmented species are often fastidious and slow growing, taking 2 days to 3 weeks to form a pigment. They are the second most common group of anaerobic bacteria encountered in human infections. Other Bacteroides species: • normal flora in the oral cavity, GI and genitourinary tract • • Bilophila wadsworthia • • • • • • Bacteroides ureolyticus • • • • • • • Bacteroides gracilis grows in bile, catalase positive, nitrate positive, urea positive, H2S positive causes pitting of the agar; colonies are usually thin, flat, spreading, irregular translucent that extend out from a slightly raised central area urease positive; actually an anaerobic Eikenella corrodens most frequently isolated from superficial soft tissue or bone infections only species that is susceptible to penicillin moved to a new genus Campylobacter and renamed Campylobacter gracilis due to its microaerophilic nature (not a true anaerobe after all) urea negative isolated from more serious deep-seated infections, including head and neck, pulmonary and sites within the abdomen and pelvis more resistant to antibiotics and more virulent than B. ureolyticus. Fusobacterium species FUSOBACTERIUM • - long, slender, spindle-shaped rods with tapered ends (fusiform) • - normal flora in the upper respiratory tract, GI tract and genitourinary tract • - most commonly encountered species in human infections are F. nucleatum and F. necrophorum • - produces large amounts of butyric acid , fluoresce chartreuse • - resistant to Vancomycin and susceptible to Kanamycin and Colistin • • • • • • • • • Fusobacterium nucleatum: - important pathogen in head, neck and lower respiratory tract; can cause serious infections/abscesses in the brain and lung, intra-abdominal, pleuropulmonary and blood 3 colony types: a) white, bread-crumb like b) speckled www.microbewiki.kenyon.edu c) smooth - gram stain has “pick up sticks” appearance with long, thin tapered rods - colonies may be convex, glistening , alpha-hemolytic colony with flecked internal structure and grows in balls in broth culture - Fusobacterium nucleatum when combined with Borrelia vincentii (spirochete) causes “Vincent's Angina” or “Trench mouth” an acute, necrotizing, ulcerative gingivitis (See Unit VII) Fusobacterium species continued: Fusobacterium necrophorum: • - more virulent • - known to invade the bloodstream and cause a widespread metastatic infection that often includes lungs, liver and bones • - produces an umbonate colony on media but grows in balls in broth culture • - indole and lipase positive Other Fusobacterium species: • - F. mortiferum and F. varium have a distinct fried-egg appearance with raised, opaque centers and a flat, translucent margin • - F. mortiferum is Esculin hydrolysis positive Synergistic Gangrene Causative agents of Synergistic gangrene: • 1. Peptostreptococcus with a gram negative rod • 2. Peptostreptococcus and Staphylococcus aureus • 3. Eikenella with Alpha strep - often found in cultures around the face and mouth as both are normal respiratory flora Identification Methods IDENTIFICATION OF ANAEROBES • Several methodologies are available for use: • 1. PRAS biochemicals • 2. Miniaturized biochemical systems (API 20A) • 3. Rapid, preformed enzyme detection panels (RapID ANA) • 4. Gas Liquid Chromatography (GLC) - for end products of glucose fermentation • 5. High Resolution GLC - for cellular fatty acid analysis • • • Probably the 2 most common methods are: 1. Preformed Enzymes 2. Gas Liquid Chromatography • • Preformed Enzymes: - Several rapid ID systems are available that do not require growth of the organism as much as relying on chromogenic and conventional substrate breakdown by preformed enzymes. A pure culture of the organism grown on an individual non- selective agar plate medium is harvested and suspended into inoculating fluid to a specific turbidity. Panels are inoculated, incubated (aerobically 350 C) for 4 hours. Factors that can affect the ID results are the culture plate medium, inoculum density, purity, correct filling of wells, incubation time and atmosphere. ID Methods continued: Gas Liquid Chromatography: • • • - Some cells can not oxidize carbohydrates and other substrates to carbon dioxide and water without oxygen. Instead they produce partially oxidized organic acids and gases that are characteristic for certain groups of anaerobic bacteria. With Gas Liquid Chromatography (GLC) it has become practical to identify these anaerobic bacteria on the basis of their metabolic pathways. - Anaerobic metabolism produces volatile and non-volatile fatty acids and alcohols. Nutrient broths such as a) Pre-reduced Peptone-Yeast extract-Glucose (PYG) and b) chopped meat with carbohydrates support the growth of anaerobic bacteria. The end products from the metabolism of the peptone and carbohydrates are volatile fatty acids that help to identify the bacteria. After incubation, the broth (peptone yeast or chopped meat) is centrifuged and the supernatant is used on the chromatograph to analyze the metabolic pathways. By-products are extracted (with ethyl-ether) and injected into a column (either glass or tubular metal) where helium gases pass through at a controlled rate. The by-products are eluted off the column and produce an electrical displacement on a thermal conductivity detector, thus displacing the needle of the graph to make a peak. These peaks are measured, compared to a known standard, which in turn identifies the anaerobic bacteria in question. GLC is a valuable procedure for the analysis of metabolic products of anaerobic bacteria. It is also a practical, inexpensive procedure that provides both speed and accuracy. It is a valuable diagnostic and research tool for large institutions or reference laboratories. ** To ensure accurate identification one should use a combination of either PRAS biochemicals and GLC or HR-GLC. References Most of the lecture notes produced in this power point presentation were acquired from: • Required Textbook: Mahon, Connie R., Lehman, Donald C., Manuselis, George,(2015). Textbook of diagnostic microbiology (5th edition). Saint Louis, Missouri: Saunders-Elsevier • Journals/Publications: Notes have also been ascertained using the following: a) Laboratory Medicine b) Advance for Medical Laboratory Professionals • Laboratory Procedures: The procedures used in the following notes with and without pictures were obtained with permission from Carolinas Medical Center-Main, Microbiology Laboratory Procedure Manuals, to be used for educational purposes only by the faculty of Carolinas College of Health Sciences, School of Clinical Laboratory Sciences, Medical Laboratory Science Program. • Hand-outs and Lecture Notes from various members and lecturers of the South Eastern Association for Clinical Microbiologists (SEACM) Annual Meeting Conference Any Questions??? Write them down or highlight them in your notes and bring them with you to Test Review.