Spatial Analysis
advertisement

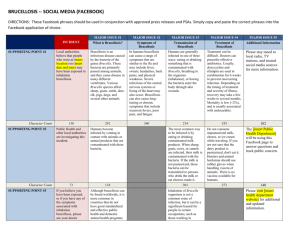
Mapping the Dynamics of Livestock Brucellosis in Georgia Using Serological Data and Spatial Statistics E. Mamisashvili1, T. Onashvili 1 L. Kerdzevadze1, K. Goginashvili 1, T.Tigilauri1, M. Donduashvili 1 , M. Nikolaishvili 1 , I. Beradze 1, M. Zakareishvili, M. Kokhreidze, M. Gelashvili1, Ian Kracalik2, S. Elizabeth Rácz3, and Jason K. Blackburn2 1Laboratory of the Ministry of Agriculture, Tbilisi, Georgia; 2 Spatial Epidemiology and Ecology Research Lab, Department of Geography and Emerging Pathogens Institute, University of Florida, Gainesville, FL, 32611, U.S.A.;3 H.W. Manter Laboratory, University of Nebraska-Lincoln, Lincoln, Nebraska, 65855 U.S.A Introduction Results Continued Spatial Analysis continued In Georgia, brucellosis is the most common of all bacterial zoonoses affecting animal populations and resulting in significant economic losses. Multiple species of Brucella contribute to chronic health complications in both animal and human populations in the country. Monitoring the occurrence of brucellosis is not only crucial for mitigating agricultural losses brought on by the disease, but also for controlling impacts on human populations as well. Areas of the former Soviet Union have been burdened by some the highest incidences of human brucellosis in world (Pappas et al. 2006). Therefore, implementing surveillance strategies on livestock populations could potentially have a two fold impact; reduced economic losses and improved rates of disease. Recent research in Armenia has shown that implementing spatial analyses in epidemiological investigations of veterinary health provided vital insight into the spatial distribution of livestock brucellosis and also aided in indentifying factors that may contribute to the presence of the disease (Porphyre et al. 2010). Similar research in Mexico was also able to elucidate risk factors for brucellosis among livestock by utilizing statistical methodologies (Mikolon et al. 1998). In addition to generating density surfaces for SP’s and SN’s a surface displaying the smoothed odds of Brucella. Spp. was created using the density of SN’s as the denominator and the density of SP’s as the numerator. This identified areas where the odds of Brucella. Spp. were the highest (Stevenson et al. 2005). Locating areas on the landscape where cases are higher than expected may be beneficial for initiating interventions and public health management strategies. In order to determine whether or not there was clustering or a higher than expected number of cases (SP ‘s) in relation to controls (SN’s) at the village level, a Bernoulli model was employed within the SaTScan v.9.0 statistical software package (Kulldorff 1997). SaTScan uses a series of overlapping circles up to a predetermined size to statistically identify areas, which given a certain likelihood contain a higher number of cases in side a circle than outside of the circle. The Bernoulli model was run using a maximum cluster size of 50% of the population at risk with no geographic overlap allowed . Results from the Bernoulli model run in SaTScan revealed the presence of statistically significant clusters of SP’s (Figure 5). The primary cluster for the model was located in the southwestern part of the country and encompassed an area approximately 80Km in diameter. There were also five secondary clusters identified across the landscape. A secondary cluster encompassing approximately a 7 km area was located in the west while the additional secondary cluster all were comprised of single locations. In an effort to build capacity and increase public health infrastructure geographic information systems (GIS) and global positioning systems were used in conjunction with sampling efforts in order to detail a baseline prevalence of brucellosis across selected regions of Georgia. Recent advances in GIS and GPS have greatly increased the ability of users to incorporate advanced analyses and data management. As part of a collaborative effort to initialize brucellosis surveillance during the spring and fall seasons of 2008 – 2010, the Laboratory of the Ministry of Agriculture (LMA) sampled livestock in three regions (provinces) of Georgia including Kakheti, Kvemo Kartli and Imereti and consisting of 16 administrative rayons. The primary goals of this study were the following: 1) to conduct surveillance for livestock brucellosis across specific rayons of Georgia designated as high risk zones 2) to enhance the capacity and knowledge of veterinary and public health management in order to adopt appropriate strategies for continued vigilance 3) to incorporate GIS and GPS technology into surveillance strategies in order to analyze spatial patterns in the distribution of brucellosis through the use of advanced spatial analytical techniques and 4) to apply new diagnostic methods. 212 254 Figure 2. Distribution of seropositives and seronegatives by rayon across Georgia. Map in red displays the total number of seropositives by rayon while the map in blue displays the total number of seronegatives by rayon. The bottom map illustrates the ratio of seropositives to seronegatives as a percent. 109 3224 1985 Sero Positive Sero Negative Kakheti 212 3224 Kvemo Kartli 254 1985 1648 Imereti 109 1648 1 0 Tbilisi 1 0 Figure 1. Total number of seropositive and seronegative samples obtained from collection efforts in major regions of Georgia. The bar chart area in purple show the number of seronegatives collected in each region, while are in pink shows the number of seropositive collected in each area. Figure 5. Map shows spatial clustering using a Bernoulli case /control model in SaTScan. The circle in red depicts the primary cluster with a radius of 39.46 Km. Secondary clusters are shown with the black circle with a radius of 3.47 Km and red stars, which depict clusters at s ingle location. Green dots represent the location of seropositives while yellow dots represent seronegatives. Results The proprtion of SP’s to SN’s was greatest in the Tskatubo, Tetri Tskaro, and Gardabani rayons (figure 2). Crude and EBS smoothed prevalence estimates show higher proportions of SP’s in the southwest in the Marneuli and Gurajanni rayons (Figure 3). Higher proportions were also indicated in the East Central area of the country in the Zestaponi and Terjola rayons. The odds of Brucella spp. were lower in centralized areas of the sampling while the odds increased along the outer fringes (Figure 4). Kernel density surfaces show a higher density of SN’ across most of the sampling sites. Discussion Identifying areas with an increased occurrence of disease may be useful for public health professionals. The mapping and analyses performed using the brucellosis seroprevalence study undertaken by scientists at LMA represents the first stage in elucidating factors that could potentially promote the presence of brucellosis. Spatial analysis of the data revealed areas with increased crude and smoothed prevalence estimates. These estimates may be actual rayons with an increased presence of the disease or they could be an artifact of the sampling effort. The sampling effort may also affect the Kernel density estimation, which showed a higher odds of Brucella spp. , although the density of SN’s was greater in most locations. The proportion of SP’s to SN’s shown in Figure 2 may reveal why the odds of Brucella spp. were higher in some Materials and Methods From July 2008 through January 2011 a team from LMA collected animal specimens seasonally (Spring, Fall) in three regions designated: Kakheti, Kvemo Kartli and Imereti (Figure 1). In total the field sampling efforts yielded 4430 blood/ serum samples and 1597 milk samples from cattle; 2522 bloods/serum samples, and 233 milk samples from sheep; and 811blood/serum samples and210 milk samples from goats. Testing of samples was performed using serology and molecular diagnostics (RoseBengal, Bacteriological, RT-PCR, and AMOS PCR). In total 552 serological tests were positive on Rose-Bengal. An additional 20 bacterial isolates were recovered and identified as Brucella melitensis n=8 or Brucella abortus n=12 obtained from AMOS PCR across the 16 rayons. locals. The cluster results from the SaTScan were successful in locating areas of concern or key locations that public health Spatial Analysis of disease to human populations. professional may want to target for additional research. Future studies may incorporate risk factors that are associated with the presence of brucellosis in livestock in order to better assess spatial differences in the level of possible exposures. Furthermore, case-control studies could potentially be developed that revisit locations of SP’s and SN’s to examine potential exposures. Overall these analysis were useful in establishing baseline rates of brucellosis and identifying potential areas to target in control efforts. Understanding the distribution of livestock patterns is the first step in helping to prevent the spread Latitude and longitude pairs identifying the location of seropositive (SP) and seronegative (SN) cases by were recorded to the nearest village. The total number of positives and negatives were aggregated to the rayon level and the ratio the of positives to negative per were rayon were calculated as a percent (Figure 2). Crude prevalence estimates per 100,000 were taken and calculated as the total number of from SP’s in a rayon divided by the estimated livestock population including small ruminants sheep and goats and cattle. Smoothed prevalence estimates were also calculated using the Empirical Bayesian smoother (EBS) in the GeoDa software package (Anselin et al. 2006). The EBS algorithm adjusts for large population variances that may lead to unstable prevalence estimates. To describe the spatial distribution of sampling SP’s and SN’s a Kernel density estimation was used to visualize a smoothed density surface . The optimal bandwidth for the Kernel density estimation was chosen following Fotheringham et al. 2000: References Figure 3. Crude prevalence estimates per 100,000 are shown in map A and Empirical Bayes smoothed rates are shown in map B. Estimates were calculated as the total number of seropositives divided by the estimated livestock population of goats, sheep, and cattle. Figure 4. Kernel density surfaces for seropositives are shown in map A and for seronegatives in map B. The odds of Brucella spp. are displayed in map C. Anselin, Luc, I. Syabri, and Y. Kho. 2006. GeoDa: An introduction to spatial data analysis. Geographical Analysis. 38:5–22. Beyer, H. L. 2004. Hawth's Analysis Tools for ArcGIS. Available at http://www.spatialecology.com/htools. Jacquez, Geoffrey M. 1996. A k nearest neighbor test for space-time interaction. Statistics in Medicine. 15:1935-1949. Kulldorff, Martin. 1997. A spatial scan statistic. Communications in statistics. 26(6):1481-1496. Mikolon, A.B., I.A. Gardner, J. H. De Anda, and S.K. Hietala. 1998. Risk factors for brucellosis seropositivity of goat herds in the Mexicali Valley of Baja Californai, Mexico. Preventive Veterinary Medicine. 37:185-195. Pappas, Georgios, Photini Papadimiriou, Nikolas Akritidis, Leonidas Christou, and Epameinondas Tsianos. 2006. The new global map of human brucellosis. The Lancet. 6: 91-99. Porphyre, T., R. Jackson, C. Sauter-Louis, D. Ward, G. Baghyan, and E. Stepanyan. 2010. Mapping brucellosis risk in commnities in the Republic of Armenia. Geospatial Health. 5(1):1-3-118. Stevenson, M.A., H. Benard, P. Bolger, and R.S. Morris. Spatial epidemiology of the Asian honey bee mite (Varro destructor) in the North islan of New Zealand. Preventive Veterinary Medicine. 71(3-4):241-252. Acknowledgements This Cooperative Biological Research project was funded by the United States Defense Threat Reduction Agency (DTRA) as part of the Biological Threat Reduction Program in Georgia. UF funding is administered through the Joint University Partnership under the University of New Mexico.