Inorganic Chemistry
advertisement

Inorganic Chemistry
Professor Pickett
Inorganic Reaction Mechanisms
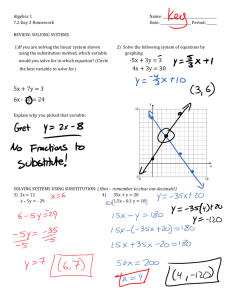
Introduction
Substitution Reactions of Octahedral Complexes
Insertion Reactions
Case Study of an Inorganic Reaction Mechanism
Substitution Reactions of Tetrahedral Complexes
Electron Transfer Reactions
Introduction
Substitution Reactions of Square Planar Complexes
EVERYONE SAY THIS OUT ALOUD
TOGETHER
THE EFFECT OF A LIGAND,
UPON THE RATE OF LIGAND REPLACEMENT,
OF THE GROUP TRANS TO ITSELF
Introduction
THE TRANS EFFECT
The Kinetic Model
The pathway from reactants to products which requires the smallest change
in potential energy is the reaction pathway or coordinate
The arrangement of atoms at the point of maximum energy is called the
transition state or activated complex
The energy of the transition state compared with that of the reactant(s) is
the activation energy ∆𝑮T ( T = double dagger)
In the majority of reactions a series of events occur, each even can be
considered as an elementary step
The elementary step with the highest activation energy is the rate
determining step
Understanding the details of the rate determining step provides knowledge of
the intimate mechanism
Introduction
The Kinetic Model
TS 1
TS 2
∆𝐺 𝑇
Intermediate
Reactants
The intermediate is stable to some
extent and can be detected using
spectroscopy
Products
Reaction Coordinate
Introduction
Free
Energy
Substitution Reactions
General Classification of Reaction Pathways
Introduction
Three Processes
Associative
A
Detectable Intermediate
Interchange
I
Transition State
Dissociative
D
Detectable Intermediate
No intermediates are detected but it can determine the character of substitution
As Y attacks M, X has substantially dissociated
from the coordination sphere
As Y attacks M, X remains bonded to M
Introduction
Two more advanced mechanisms
Introduction
Substitution Reactions of Square Planar Complexes
Substitution Reactions of Octahedral Complexes
Insertion Reactions
Case Study of an Inorganic Reaction Mechanism
Substitution Reactions of Tetrahedral Complexes
Electron Transfer Reactions
Substitution Reactions of Square
Planar Complexes
Inorganic Reaction Mechanisms
THE TRANS EFFECT
THE EFFECT OF A LIGAND,
UPON THE RATE OF LIGAND REPLACEMENT,
OF THE GROUP TRANS TO ITSELF
Substitution Reactions of Square
Planar Complexes
EVERYONE SAY THIS OUT ALOUD
TOGETHER
Rate:
𝑑 𝑀𝐿3 𝑋
= 𝑀𝐿3 𝑋 {𝑘𝑠𝑜𝑙𝑣𝑒𝑛𝑡 + 𝑘2 𝑌 }
𝑑𝑡
Rate Law:
𝑘𝑜𝑏𝑠 = 𝑘𝑠𝑜𝑙𝑣𝑒𝑛𝑡 + 𝑘2 𝑌
𝑘𝑠𝑜𝑙𝑣𝑒𝑛𝑡 = 𝑘1 [𝑠𝑜𝑙𝑣𝑒𝑛𝑡]
Substitution Reactions of Square
Planar Complexes
Substitution Reactions
Square Planar Complexes (IA)
STEREOCHEMISTRY IS CONSERVED IN BOTH PATHWAYS
Substitution Reactions of Square
Planar Complexes
Due to the associative mechanism
kobs
Hexane
Methanol
[Y] = NHMe2
• The methanol can attack
the metal centre due to the
lone pair on oxygen
• The concentration of
methanol does not effect
the observed rate
• Hexane can not coordinate
Substitution Reactions of Square
Planar Complexes
Role of solvent
Shape?
Trigonal Bipyramidal
R = Me
Favours Clintermediates. As is
more electropositive
than N or P.
Substitution Reactions of Square
Planar Complexes
Some evidence supporting associative
mechanism
Stable Five-Coordinate Pt(II)
Test to whether the solvent has a role in the substitution
In water, trap the intermediate using a base. Hydroxyl ligand is formed
Substitution Reactions of Square
Planar Complexes
Evidence for solvent role
Substitution Reactions of Square
Planar Complexes
Activation Magnitudes for substitution of
square planar complexes Pt/Pd(II)
Nature of the:
Entering group Y
Leaving group X
Metal M
Spectator ligands L
Steric effect
The Trans Effect
The Entering Group Y, Nucleophile
Rates are strongly influenced by the nucleophile and increases along the
series:
PR3 > I- > N3- > NO2- > Br- > Py > NH3 > Cl- > H2O > HO-
This order is similar to nucleophilicity in SN2 reactions
Substitution Reactions of Square
Planar Complexes
Factors which can influence the rate of
substitution of square planar complexes
Can define a kinetic scale for relative rate of attack by ligand Y on a Pt(II)
complex relative to the rate of attack by the solvent (methanol)
The nucleophilic reactivity consant nPt is defined for an attacking ligand Y as:
𝑛𝑃𝑡 = log
𝑘𝑌
𝑘𝑠𝑜𝑙𝑣𝑒𝑛𝑡
The Leaving Group X
In all substitution mechanisms, including predominately associative, the
stronger the metal bond with the leaving group the slower the substitution
reaction
For association reactions the effect of the leaving group depends on the
extent of breaking in the transition state
In general, good entering groups (nucleophiles) are poor leaving groups:
NO3 > H2O > Cl > Br > I > SCN > NO2 > CN
Substitution Reactions of Square
Planar Complexes
A Nucleophilicity Scale
The greater the tendency for the metal to form a five coordinate complex,
the greater the association of the transition state and hence the faster
substitution reaction
Bimolecular rate enhancement ( Ni > Pd > Pt )
The Spectator Ligands L
Steric Hindrance
Bulky spectator ligands make it
difficult for entering and leaving
groups to move around the system
Substitution is increased when
smaller, less bulky, spectator
ligands are present
Substitution Reactions of Square
Planar Complexes
The Metal M
THE EFFECT OF A LIGAND, UPON THE RATE OF LIGAND REPLACEMENT, OF THE
GROUP TRANS TO ITSELF
Not to be confused with trans influence which is a ground state effect
phenomenon concerned primarily with bond length changes
Termed a LABILIISING effect, emphasising kinetic nature
Different trans ligands T exert different labilising influence on X
The kinetic effect has a practical consequence in controlling the
stereochemistry if substitution products at square planar complexes
Chloride is a stronger trans-labilsing ligand than amine
Substitution Reactions of Square
Planar Complexes
The Spectator Ligands L
The Trans Effect
F- , H2O , -OH
NH3 , py
ClBrI- , SCN- , NO2- , SC(NH2)2 , PhSO32PR3 , AsR3 , SR2 , CH3H- , NO , CO , CN- , C2H4
Stronger Trans-Labile Ligand
Substitution Reactions of Square
Planar Complexes
Weaker Trans-Labile Ligand
Raises ground
state energy,
lowers the
activation
energy and
makes the
transition state
more accessible
T2 has a greater effect trans
effect than T1 because it
destabilises the ground
state
H- & Cl- donates negative
charge to metal centre
which weakens the bond
trans to itself.
Stabilisation of the Transition State
T2 has a greater trans effect than T1
because it stabilises the transition
state
CO can withdraw electron density on
metal centre due to backbonding, so
lowers the transition state energy
Substitution Reactions of Square
Planar Complexes
The Spectator Ligands L
The Trans Effect
Destabilisation of Ground State
Purely thermodynamic phenomenon
Ligands can influence the ground state properties of the groups to which they
are trans
Such properties include
Metal-Ligand bond lengths
Vibrational frequency or force constants
NMR coupling constants
The trans influence series is based on structural data:
R- | H
=PR3
Strong Effect
Large Effect
CO | C=C | Cl- | NH3
The Cis Effect
Much weaker than the Trans Effect
Little / No Effect
Substitution Reactions of Square
Planar Complexes
Trans Influence
Define the trans effect.
10%
Definition
‘The effect of a ligand upon the rate of ligand replacement of the group
trans to itself, primarily refers to substitution at group VIII M(II) square
planar complexes ’ A simple diagram would aid definition.
Substitution Reactions of Square
Planar Complexes
Test Question
Hydride and carbon monoxide are both strong trans effect ligands, discuss how
their trans effect operates.
10%
Hydride lowers the activation energy for subsitution at eg Pt(II) by
destabilising the ground state, it places more electron density onto the
metal centre, weakening the bond trans to it moreso than does eg Cl-.
Carbon monoxide lowers the activation energy by stabilising the TS. It
relieves the increased electron density brought in by the lone
pair/negative charge on the incoming ligand by accepting this into pi* anti
bonding orbitals.
Substitution Reactions of Square
Planar Complexes
Test Question
Briefly discuss how the trans effect can provide a means of kinetically
controlling the nature of the isomer formation in Pt(II) substitution chemistry.
[10%]
The selective formation of cis or trans isomers of [PtCl2(NH3)2] can controlled
by either starting with [Pt (NH3)4]2+ and reacting with Cl- which gives
exclusively the trans-product or by starting with [Pt Cl4]2- whch gives the cisproduct. This is a consequence of Cl- being a greater trans effect than has
NH3. Scheme as in notes would aid clarity.
Substitution Reactions of Square
Planar Complexes
Test Question
Introduction
Substitution Reactions of Square Planar Complexes
Substitution Reactions of Octahedral Complexes
Insertion Reactions
Case Study of an Inorganic Reaction Mechanism
Substitution Reactions of Tetrahedral Complexes
Electron Transfer Reactions
Substitution Reactions of
Octahedral Complexes
Inorganic Reaction Mechanisms
THE TRANS EFFECT
THE EFFECT OF A LIGAND,
UPON THE RATE OF LIGAND REPLACEMENT,
OF THE GROUP TRANS TO ITSELF
Substitution Reactions of
Octahedral Complexes
EVERYONE SAY THIS OUT ALOUD
TOGETHER
Rates similar to water exchange rates, suggests dissociation of water is important
Rates insensitive to nucleophilicity of Y
A or IA pathways unlikely
Rates sensitive to charge on anion
Ion-pair formation
Rates strongly dependent on nature of M
Normally D or ID mechanism
Substitution Reactions of
Octahedral Complexes
Substitution Reactions
Octahedral Substitution (ID)
The D Mechanism
1.
Complex gains enough energy to break the M-X bond completely
2.
Intermediate is attacked by incoming Y
The ID Mechanism
As the M-X bond starts to break, M begins to form a new bond with Y
The entering group Y must be present in the region around the complex when the M-X bond begins to lengthen
Thus before substitution occurs Y must enter the outer sphere
Ligands swap over in rate determining step before X leaves
Substitution Reactions of
Octahedral Complexes
Two elementary steps:
Nature of the:
Entering group Y
Metal M
Leaving group X
Spectator ligands L
Electronic Effects
Jahn-Teller Effect
The Entering Group Y
Expect that rate of a dissociative substitution would be insensitive to the
entering group Y
By varying the nature of the entering group the effect is much smaller
The Metal M
The relatively small 3d transition metals do not sterically easily accommodate
7 coordination, D or ID mechanisms dominate their substitution chemistry
Substitution Reactions of
Octahedral Complexes
Factors which can influence the rate of
substitution of octahedral complexes
The Leaving Group X
Reactions proceeding via I or ID mechanisms are expected to be sensitive to
the nature of the leaving group X since the M-X bond is to be cleaved
Experimental results for the aquation reaction below shows influence of X on
rate can span 5 orders of magnitude
The strength of the M-X bond is crucial in the magnitude of the activation
energy in the rate determining step
X
NO3- I-
Cl- F-
N3- NCS-
K
10-5 10-6 10-6 10-8 10-9 10-10
The Spectator Ligands L
In D or ID mechanism the coordination number is decreased in the transition
state
If the metal centre is surrounded by bulky ligands then steric strain is
released on attaining the transition state
Enhancement is then given on the substitution rate
Substitution Reactions of
Octahedral Complexes
Electronic Effects
The five coordinate transition
state in the dissociative
mechanism can be stabilised if
the remaining ligands can
donate more electron density to
the metal
This is possible if there is a πdonor group CIS to the leaving
group
This does not happen if it is
trans.
Jahn-Teller Effect
The most labile transition metal
ions are Cu(II) and Cr(II) and this
is due to Jahn-Teller distortion
Octahedral d9 and d4 high spin
complexes are unstable because
of the two long metal ligand
bonds
Substitution Reactions of
Octahedral Complexes
Base Hydrolysis
Replacement of ligand by HO-
Often faster than acid hydrolysis
Sensitive to nature of entering group
Only observed when deprotonatable group present
First step is removal of proton from acidic ligand
Dissociative Conjugate Base Mechanism (DCB)
Garrick Mechanism
Addition of charge
makes it easier to
remove the chloride
Substitution Reactions of
Octahedral Complexes
Briefly discuss the following. [15%]
(i) The influence of the leaving group in substitution reactions of
(a)
square planar complexes
(b)
first transition series octahedral complexes
A metal ligand bond begins breaking in the TBP TS therefore the stronger this
bond the slower is the substitution reaction. For example NO3- is weakly
bound, it is a labile ligand and its complexes Pt(II) are rapidly substituted
whereas SCN- is tightly bound and its corresponding substitution reactions are
very slow.
Again bond breaking takes place in the TS the stronger the M-X bond the
slower is the substitution reaction. For example, [Co(NH)5X]2+ aquates 5
orders of magnitude faster for X = NO3- than for SCN-
Substitution Reactions of
Octahedral Complexes
Test Question
Briefly discuss how the steric properties of spectator ligands can influence the
rate of substitution of
(a) Octahedral
and (b) Square planar complexes [10%]
As TS becomes less crowded by departure of leaving group then steric
strain relieved in the GS is relieved. This leads to a lower activation
energy and faster substitution kinetics. An example would aid discussion.
The converse to above, higher steric strain in TS on going from 4 to 5
coordinate can raise activation energy decreasing substitution rate.
Substitution Reactions of
Octahedral Complexes
Test Question
Discuss the mechanism of substitution of [Co(NH3)5Cl] + by hydroxide. What
happens if another ligand such as SCN - is present when hydroxide is added to the
aqueous solution of the complex?
Garrick Mechanism, Dissociative, conjugate base mechanism D-CB. A key point
is that substitution shows first order dependence on complex and first-order
on [OH]-concentrations, ie overall 2nd order kinetics. This is explained by
hydroxide removing a proton from an ‘acidic’ NH3 ligand. This decreases the
net charge on the complex making the electrostatic separation of Cl- from an
1+ charged complex easier (faster) than from the 2+ charged parent complex.
Following Cl- dissociation hydroxide attacks the five coordinated
intermediate. In the presence of SCN- competive quenching of this
intermediate can occur giving both OH- and SCN- substitution products after
the re-protonation step. A scheme as below would aid discussion.
Substitution Reactions of
Octahedral Complexes
Test Question
2+
NH3
NH3
OH-
H2 N
Co
H 3N
- H2 O
Cl
NH3
NH3
NH3
Co
H3 N
Cl
NH3
-Cl 1+
H2 N
NH3
NH3
2+
H2 N
Co
H3 N
Co
OH
H 3N
NH3
H2 O
2+
H3 N
NH3
NH3
Co
H3 N
OH
NH3
NH3
NH3
NH3
Substitution Reactions of
Octahedral Complexes
H3 N
1+
Substitution one dinitrogen ligand of trans-[Mo(N2)2(dppe)2)] by MeCN to give
trans-[Mo(N2)MeCN)(dppe)2)] shows a first order dependence on the
concentration of the complex and a zero order dependence on concentration of
the incoming ligand under pseudo-first order conditions. Use the steady state
approximation to derive an expression for the rate equation consistent with
these observations and a mechanism involving loss of dinitrogen as the rds.
Briefly explain why the rate of alkylation of dinitrogen by MeBr in the same
complex to give a diazenide shows identical kinetics.25%
Substitution Reactions of
Octahedral Complexes
Test Question
Substitution Reactions of
Octahedral Complexes
Applying the steady-state approximation
leads to an expression containing the concentration of I
k1 [A] = k-1 [I] [N2] + k2 [I][L]
k2 [L] >> {k-1 [N2]
This reduces to :
dP / dt = k1[A] = - dA/dt
Thus
k1 = kobserved
Rearranging gives
[I] = k1 [A] / {k-1 [N2] + k2 [L]}
In terms of I the rate of formation of the product P is
dP /dt = k2 [I][L]
Substitute for [I] gives
dP /dt = k2 [L][k1[A] / {k-1 [N2] + k2 [L]}
= [k1 [A] / {k-1 [N2]/ k2 [L] + 1]}
The rate of reaction of the
complex with MeBr shows
the same kinetics because
the rate determining step is
the same, that is the loss of
a dinitrogen ligand to give a
five coordinate intermediate,
the subsequent reaction of
which with MeBr is fast.
Substitution Reactions of
Octahedral Complexes
dI /dt = 0
Since:
Introduction
Substitution Reactions of Square Planar Complexes
Substitution Reactions of Octahedral Complexes
Insertion Reactions
Case Study of an Inorganic Reaction Mechanism
Substitution Reactions of Tetrahedral Complexes
Electron Transfer Reactions
Insertion Reactions
Inorganic Reaction Mechanisms
THE TRANS EFFECT
THE EFFECT OF A LIGAND,
UPON THE RATE OF LIGAND REPLACEMENT,
OF THE GROUP TRANS TO ITSELF
Insertion Reactions
EVERYONE SAY THIS OUT ALOUD
TOGETHER
Insertion Reaction (Migration)
The entering group Y does not insert between the M-X bond but is bound CIS
in the product
Generally dissociative, D, in character with the formation of the trigonal
bipyramid intermediate as the rate determining step
Insertion Reactions
How do they take place
The R group migrates towards the CO
Proceed with the retention of stereochemistry and accelerated by Lewis
acids
Insertion Reactions
Introduction
Substitution Reactions of Square Planar Complexes
Substitution Reactions of Octahedral Complexes
Insertion Reactions
Case Study of an Inorganic Reaction Mechanism
Substitution Reactions of Tetrahedral Complexes
Electron Transfer Reactions
Case Study of an Inorganic
Reaction Mechanism
Inorganic Reaction Mechanisms
THE TRANS EFFECT
THE EFFECT OF A LIGAND,
UPON THE RATE OF LIGAND REPLACEMENT,
OF THE GROUP TRANS TO ITSELF
Case Study of an Inorganic
Reaction Mechanism
EVERYONE SAY THIS OUT ALOUD
TOGETHER
A
Rate:
−
𝑑𝐴
= 𝑘𝑜𝑏𝑠 [𝐴]
𝑑𝑡
• Independent of [MeCN]
• kobs independent of a wide variety of L such as –isocyanide, -CO, -amine
Case Study of an Inorganic
Reaction Mechanism
Case Study of an Inorganic Reaction
Mechanism
Apply Steady State Approximation
Rate of formation of I is equal to its loss
𝑑𝐼
𝑑𝑡
Therefore:
𝑘1 𝐴 = 𝑘−1 𝐼 𝑁2 + 𝑘2 𝐼 [𝐿]
Rearranging gives:
𝑘1 𝐴
𝐼 =
𝑘−1 𝑁2 + 𝑘2 [𝐿]
Rate of formation of P:
𝑑𝑃
= 𝑘2 𝐼 [𝐿]
𝑑𝑡
Substitute in [I]:
𝑑𝑃
𝑘2 𝑘1 [𝐴][𝐿]
=
𝑑𝑡 𝑘−1 𝑁2 + 𝑘2 [𝐿]
=0
𝑑𝐴
= 𝑘𝑜𝑏𝑠 [𝐴]
𝑑𝑡
Since k2[L] >> k1[N2]:
𝑑𝑃
𝑘2 𝑘1 [𝐴][𝐿]
=
𝑑𝑡 𝑘−1 𝑁2 + 𝑘2 [𝐿]
=
𝑘2 𝑘1 [𝐴][𝐿]
𝑘2 [𝐿]
= 𝑘1 [𝐴]
Thus k1 = kobs
Case Study of an Inorganic
Reaction Mechanism
−
Alkylation of dinitrogen ligand
Case Study of an Inorganic
Reaction Mechanism
All proceed via the same rate of substitution
Alkylation of dinitrogen ligand – Mechanism
Attacks Mo(I) radical to form diazenide
Evidence : EPR
Case Study of an Inorganic
Reaction Mechanism
MeBr coordinates
and homolytically
cleaves with
formation of
methyl radical
Introduction
Substitution Reactions of Square Planar Complexes
Substitution Reactions of Octahedral Complexes
Insertion Reactions
Case Study of an Inorganic Reaction Mechanism
Substitution Reactions of Tetrahedral Complexes
Electron Transfer Reactions
Substitution Reactions of
Tetrahedral Complexes
Inorganic Reaction Mechanisms
THE TRANS EFFECT
THE EFFECT OF A LIGAND,
UPON THE RATE OF LIGAND REPLACEMENT,
OF THE GROUP TRANS TO ITSELF
Substitution Reactions of
Tetrahedral Complexes
EVERYONE SAY THIS OUT ALOUD
TOGETHER
Tetrahedral cyclopentadienyl complexes
Might expect dissociative mechanism
Experimentally 2nd order kinetics rate = k[Rhenium Complex]1[PMe3]1[CO]0
Ring Slippage
Now to apply the
SSA again
Substitution Reactions of
Tetrahedral Complexes
Therefore:
𝑘−1 𝐼 + 𝑘2 𝐼 𝑃𝑀𝑒3 = 𝑘1 [𝐴]
Rearrange to get [I]:
𝑘1 𝐴
𝐼 =
𝑘−1 + 𝑘2 [𝑃𝑀𝑒3 ]
Rate of product P formation:
𝑑𝑃
= 𝑘2 𝐼 𝑃𝑀𝑒3
𝑑𝑡
Substitute in [I]:
𝑑𝑃
𝑘2 𝑘1 𝐴 𝑃𝑀𝑒3
=
𝑑𝑡 𝑘−1 + 𝑘2 [𝑃𝑀𝑒3 ]
𝑑𝐼
𝑑𝑡
=0
Since k-1 >>> k2[PMe3]
because k1 is an
intramolecular process…
𝑑𝑃
𝑑𝑡
=
𝑘2 𝑘1 𝐴 𝑃𝑀𝑒3
𝑘−1 +𝑘2 [𝑃𝑀𝑒3 ]
𝑘2 𝑘1 𝐴 𝑃𝑀𝑒3
=
𝑘−1
= 𝑘2 𝐾𝑒 𝐴 [𝑃𝑀𝑒3 ]
Ke = Equilibrium constant for
ring slippage
Ke = k1/k-1
Substitution Reactions of
Tetrahedral Complexes
Rate of loss of I is equal to its formation
What if dissociative pathway?
[PMe3] >> [CO]
=
𝑘2 𝑘1 𝐴 𝑃𝑀𝑒3
𝑘−1 [𝐶𝑂] + 𝑘2 [𝑃𝑀𝑒3 ]
=
𝑘2 𝑘1 𝐴 𝑃𝑀𝑒3
𝑘2 [𝑃𝑀𝑒3 ]
= 𝑘1 [𝐴]
Experimental results say that there is not zero
order in [PMe3] therefore this cant be true
Substitution Reactions of
Tetrahedral Complexes
𝑑𝑃
𝑘2 𝑘1 𝐴 𝑃𝑀𝑒3
=
𝑑𝑡 𝑘−1 [𝐶𝑂] + 𝑘2 [𝑃𝑀𝑒3 ]
Substitution Reactions of
Tetrahedral Complexes
Tetrahedral Substitution
Introduction
Substitution Reactions of Square Planar Complexes
Substitution Reactions of Octahedral Complexes
Insertion Reactions
Case Study of an Inorganic Reaction Mechanism
Substitution Reactions of Tetrahedral Complexes
Electron Transfer Reactions
Electron Transfer Reactions
Inorganic Reaction Mechanisms
THE TRANS EFFECT
THE EFFECT OF A LIGAND,
UPON THE RATE OF LIGAND REPLACEMENT,
OF THE GROUP TRANS TO ITSELF
Electron Transfer Reactions
EVERYONE SAY THIS OUT ALOUD
TOGETHER
May occur by either or both of two mechanisms
Outer Sphere Electron Transfer
Inner Sphere Electron Transfer
Outer Sphere Electron Transfer
In principle all outer sphere mechanisms involve electron transfer from
reductant to oxidant with the coordination shells or spheres staying intact
One reactant becomes involved in the outer or second coordination sphere of
the other reactant and an electron flows from the reductant to oxidant
Such a mechanism is established when rapid electron transfer occurs between
two substitution inert complexes
Electron Transfer Reactions
Electron Transfer Reactions
Reductant and oxidant share a ligand in which their inner or primary
coordination sphere
The electron is transferred across the bridging group
Undergoes aquation to hexaquo
Electron Transfer Reactions
Inner Sphere Electron Transfer
1. Formation of precursor complex in a shared solvent cage
A + B {A---B}
KAB
2. Activation of precursor, electron transfer, and relaxation of successor complex
{A---B} {A+---B-}
kET
3. Products separate from solvent cage
{A+---B-} A+ + BImportant Factors
Solvent reorganisation
Electronic structure
Metal-Ligand reorganisation is small
fast
Electron Transfer Reactions
Outer Sphere Electron Transfer
Elementary Steps
Undergoes Outer Sphere Electron Transfer Mechanism
Requires the transfer of an electron from one eg orbital to the other
Note:
Same ligand complex but always in constant exchange
Aromatic ligands give fast transfer
[Co(Phen)3]2+ + [Co(Phen)3]3+ [Co(Phen)3]3+ + [Co(Phen)3]2+
Electron Transfer Reactions
Simplest Reaction
Electron Self Exchange | Low Spin Ligand
Extremely slow due to change in multiplicity of the system
[Co(NH3)6]2+ + [Co(NH3)6]3+ [Co(NH3)6]3+ + [Co(NH3)6]2+
Electron Transfer Reactions
Electron Self Exchange | High Spin
Ligand
Electron Transfer Reactions
Electron Transfer
Bond Length changes
Oxidant and reductant must reorganise coordination spheres before electron transfer
so energies of the transition states are equal
If electron transfer took place at their equilibrium bond positions, then we would have
a compressed Fe2+ ion and a stretched Fe3+ ion. These are in vibrationally excited
states and would release energy which contradicts thermodynamics.
The reagents must match their energies before electron transfer can take place
Electron Transfer Reactions
Frank-Condon Barrier to Electron
Transfer
Cross reactions and the Marcus Equation
k1,1
[MLn]y+ +
[MLn]y+
K1,2
[MLn]y+ + [MLn]x+
k2,2
[MLn]x+
f12 = z21,2 / z1,1 . z2,2
k12 = (k11k22K12f12)1/2
Equilibrium constant, can consider as
driving force.
WITH APPROXIMATION f = 1
k12 = (k1k2K12)1/2
Can be calculated from
redox potentials of reactants
Rate constant
Electron-self exchange
rate constants
Electron Transfer Reactions
[MLn]x+ +
Past Exam Question
Square planar Pt(II) complexes usually undergo substitution by an associative, A,
or intimate associative, Ia, mechanism. Discuss this observation. [15%]
Past Exam Questions
Past Exam Question
Square planar Pt(II) complexes usually undergo substitution by an associative, A,
or intimate associative, Ia, mechanism. Discuss this observation. [15%]
Past Exam Questions
Past Exam Question
What is the principal stereochemical feature of ligand substitution at Pt(II) and
how does this arise? [15%]
Past Exam Questions
Past Exam Question
How does a bulky ligand, placed in the cis-position relative to the substituted
ligand, influence the rate of substitution of a Pt(II) complex? [15%]
Past Exam Questions
Past Exam Question
What is the trans-effect and how does it operate? [25%]
Past Exam Questions
Past Exam Question
What is the trans-effect and how does it operate? [25%]
Past Exam Questions
Past Exam Question
Past Exam Questions
Past Exam Question
Past Exam Questions
Past Exam Question
Past Exam Questions
Past Exam Question
Past Exam Questions