PAIN
advertisement

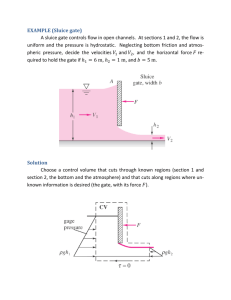
UNIVERSITY OF GUYANA BMP 3201 PAIN ASSIGNMENT 1 GROUP MEMBERS Kamini Reddi Jennifer Haynes Satrupa Singh ASSIGNMENT 1 PAIN - Acute, Chronic and Referred Pain -Mechanism Of Pain Reception And Transmission -Pain Modulation And ControlGate Control Theory & Endogenous Opioid System PAIN Pain can be defined as an unpleasant feeling that is a vital function of the nervous system because it warns the body of the potential or actual injury. Three types of Pain: (1) Acute (2) Chronic (3) Referred TYPES OF PAIN: ACUTE This type of pain can be defined as the duration of pain in less than 6 months which can be identified as an underlying pathology By limiting activity to prevent further damage and promote tissue healing and recovery, acute pain serves a protective function; however, it might also affect an individual’s quality of life and impair the ability to function well. ACUTE PAIN CONT’D Treatment resulting from musculoskeletal injury includes: resolving the underlying disorder, reduce inflammation and modify the transmission of pain from the Periphery to the CNS. CHRONIC PAIN Persists beyond the normal time for tissue healing. Chronic pain is usually as a result of activation of dysfunctional neurological or pathological responses that cause the person to continue to experience the sensation of pain even when no damaging or threatening stimulus is present. Acute pain is the start of chronic pain. Chronic pain maybe the result of changes in sympathetic nervous system and adrenal activity , reduced production of endogenous opioids or sensitization of primary afferents (peripheral sensitization) and spinal cord neurons. CLASSIFICATIONS OF CHRONIC PAIN 1) Nociceptive pain: caused by a stimulation of pain receptors by noxious chemicals, mechanical and thermal stimuli and ongoing tissue damage Some conditions under this class are: arthritis, ischemia, cancer chronic pancreatitis. CLASSIFICATIONS OF CHRONIC PAIN CONT’D 2) Neuropathic pain is the results of peripheral or central nervous system dysfunction without ongoing tissue damage. Some conditions under this classification include: diabetic neuropathy, postherpetic neuralgia and phantom limb pain. Mixed pain syndrome - are those with multiple or unclear pathophysiology. Example: recurrent headache and some vasculitis syndrome Psychological pain syndromes - are those in which the psychological process plays a major role. This type of pain may be seen in somata form disorder and conversion reactions. Chronic pain maybe the result of changes in sympathetic nervous system and adrenal activity, reduced production of endogenous opioids or sensitization of primary afferents (peripheral sensitization) and spinal cord neurons. Decreased levels of enkephalins and increases numbers and sensitivity of nociceptors have been observed in individuals with chronic pain. Those individuals have increased sensitivity to both noxious (hyeralgesia) and innocuous (allodynia) stimuli. This process is known as wind up or central sensitization. REFERRED PAIN This is the experience of pain in one area when the actual or tissue damage is in another area. The pain maybe referred from one joint to another, from a peripheral nerve to a distal area of innervation, or from an internal organ to an area of musculoskeletal tissue. E.g. Myocardial infarction or angina caused by cardiac ischemia that is felt in the upper chest, left shoulder, jaw and arm and pain originating from the central portion of the diaphragm that is frequently felt in the lateral tip of either shoulder. REFERRED PAIN CONT’D It is proposed that pain is referred in one of 3 ways: (1) - from a nerve to its area of innervation (2) - from one area to another derived from the same dermatome (3) - from one area to another derived from the same embryonic segment REFERRED PAIN CONT’D The peripheral neural pathways of these different areas conversed on the same or similar areas of the spinal cord and synapse with the same second order neurons to ascend the spinal cord and reach the central cortex. When pain that maybe of either viseral or musculoskeleton orgin converges on the same neuron in the spinal cord it is usually interpreted to be of musculoskeletal origin.This may be because musculoskeletal injury and pain are so much more common that the brain learns that activity arriving along that pathway is associated with pain stimulus in particular musculoskeleton. MEC HANISMS OF PAIN RECEPTION & TRANSMISSION NOCICEPTORS Specialised sensory receptors responsible for the detection of noxious (unpleasant) stimuli, transforming the stimuli into electrical signals, which are then conducted to the central nervous system. They are the free nerve endings of primary afferent Aδ and C fibres. Distributed throughout the body (skin, viscera, muscles, joints, meninges) they can be stimulated by mechanical, thermal or chemical stimuli. NOCICEPTORS CONT’D Inflammatory mediators (eg, bradykinin, serotonin, prostaglandins, cytokines, and H+) are released from damaged tissue and can stimulate nociceptors directly. The process of inflammatory mediators acting to reduce the activation threshold of nociceptors so that the stimulation required to cause activation is less can be known as Primary Sensitisation. PRIMARY AFFERENT FIBRES In addition to the Aδ and C fibres that carry noxious sensory information, there are primary afferent Aβ fibres that carry non-noxious stimuli. Each of these fibre types possesses different characteristics that allow the transmission of particular types of sensory information Aβ fibres are highly myelinated and of large diameter, therefore allowing rapid signal conduction. They have a low activation threshold and usually respond to light touch and transmit nonnoxious stimuli. Aδ fibres are lightly myelinated and smaller diameter, and hence conduct more slowly than Aβ fibres. They respond to mechanical and thermal stimuli. They carry rapid, sharp pain and are responsible for the initial reflex response to acute pain. C fibres are unmyelinated and are also the smallest type of primary afferent fibre. Hence they demonstrate the slowest conduction. C fibres are polymodal, responding to chemical, mechanical and thermal stimuli. C fibre activation leads to slow, burning pain PAIN MODULATION AND GATE – CONTROL THEORY PAIN MODULATION Pain is modulated by two primary types of drugs that work on the brain analgesics anesthetics. The term analgesic refers to a drug that relieves pain without loss of consciousness. The term anesthesia refers to a drug that depresses the CNS. It is characterized by the absence of all perception of sensory modalities, including loss of consciousness without loss of vital functions. PAIN MODULATION CONT’D In the CNS there is a circuit that can modulate incoming pain information. The gate control theory and the ascending/descending pain transmission system are two suggestions of such a circuit. GATE CONTROL THEORY The first pain modulatory mechanism called the "Gate Control" theory was proposed by Melzack and Wall in the mid-1960s. The concept of the gate control theory is that non-painful input closes the gates to painful input, which results in prevention of the pain sensation from traveling to the CNS (i.e., non-noxious input [stimulation] suppresses pain). GATE CONTROL THEORY CONT’D The theory suggests that collaterals of the large sensory fibers carrying cutaneous sensory input activate inhibitory interneurons, which inhibit (modulate) pain transmission information carried by the pain fibers. Non-noxious input suppresses pain, or sensory input “closes the gate” to noxious input. GATE CONTROL THEORY CONT’D The gate theory predicts that at the spinal cord level, non-noxious stimulation will produce presynaptic inhibition on dorsal root nociceptor fibers that synapse on nociceptors spinal neurons (T), and this presynaptic inhibition will block incoming noxious information from reaching the CNS (i.e., will close the gate to incoming noxious information). GATE CONTROL THEORY CONT’D The gate theory was the rationale for the idea behind the production and the use of transcutaneous electrical nerve stimulation (TENS) for pain relief. To be effective, the TENS unit produces two different current frequencies below the pain threshold that can be tolerated by the patient. This procedure has partial success in pain therapy. PICTURE SHOWING THE DIAGRAM OF GATE CONTROL THEORY OF PAIN STEPS OF PAIN MODULATION AND GATE – CONTROL THEORY Without any stimulation, both large and small nerve fibers are quiet and the inhibitory interneuron (I) blocks the signal in the projection neuron (P) that connects to the brain. The "gate is closed" and therefore NO PAIN. With non-painful stimulation, large nerve fibers are activated primarily. This activates the projection neuron (P), BUT it ALSO activates the inhibitory interneuron (I) which then BLOCKS the signal in the projection neuron (P) that connects to the brain. The "gate is closed" and therefore NO PAIN. With pain stimulation, small nerve fibers become active. They activate the projection neurons (P) and BLOCK the inhibitory interneuron (I). Because activity of the inhibitory interneuron is blocked, it CANNOT block the output of the projection neuron that connects with the brain. The "gate is open", therefore, PAIN!! THE ENDOGENOUS OPOID SYSTEM Pain perception is also modulated by endogenous opiate- like peptides; called opiopeptins, (previously known as endorphins). Opiopeptins control pain by binding to specific opiate receptors in the nervous system. An endogenous system of analgesia was first discovered by three independent group of researchers who were investigating the mechanism of morphine induced analgesia. In 1975, two peptides, met-enkephalin (methionineenkaphalin) and leu-enkaphalin (leucine-enkaphalin), which were isolated from the CNS of pig, were shown to produce physiological effects similar to those of morphine. These peptides also bind specifically to the opiate receptor, and their action and binding are blocked by naloxone, an opiate antagonist. Opiopeptins and opiate receptors have been found in many peripheral nerve endings and in neurons in several regions of the nervous system. Concentration of opiopeptins and opiate receptors have been identified in various area of the brain, including the periaqueductal grey matter (PAGM), and the raphe nucleus of the brain stem, which are structures that induce analgesia when electrical stimulated and various areas of the limbic system. Opiopeptins are also found in high concentrations in the superficial layer of the dorsal horn of the spinal cord (layers I&II) and the enteric nervous system as well as in nerve endings of C fibers. Opioids and opiopeptins always have an inhibitory action, causing presynaptic inhibition by suppressing the inward flux of calcium and cause postsynaptic inhibition by activating and outward potassium current. In addition opiopeptins indirectly inhibit pain transmission by inhibiting the release of gammaamniobutyric acid (GABA) in the PAGM and the raphe nucleus. ES of the area with high levels of opiopeptins, such as the PAGM, and the raphe nucleus, strongly inhibits the transmission of pain messages by some spinal dorsal horn neurons, thereby causing analgesia The endogenous opiate theory also provides an explaination of the paradoxical pain relieving effects of painful stimulation and acupuncture. The brain stimulates the pituitary gland and blocks the pain transmission by not allowing depolarization to occur, A-delta or C fibers; endogenous opiates; blocking noxious stimuli A noxious stimulus (through A-delta and C fibers) activates this descending pain control mechanism. KEY POINTS The pain experienced by patients is a result of the interaction between sensory and emotional experiences. Aδ fibres transmit rapid, sharp, localised pain. C fibres transmit slow, diffuse, dull pain. KEY POINTS CONT’D Pain transmission can be modulated at a number of levels, including the dorsal horn of the spinal cord and via descending inhibitory pathways. The spinothalamic and spinoreticular tracts are important ascending pain pathways Neuropathic pain can be spontaneous and is often described as burning, shooting, or stabbing CONCLUSION Pain is both a sensory and emotional experience, and patients past experiences, fears and anxieties can play an important role. Pain transmission is a result of complex peripheral and central processes. These processes can be modulated at different levels and pain perception is a result of the balance between facilitatory and inhibitory interactions. CONCLUSION CONT’D Current areas of interest in pain research include: investigating the effect of mood on pain processing in the brain and looking for novel drugs to block channels involved in pain transmission. REFERENCES An Introduction to Pain Pathways and Mechanisms. (n.d.). Retrieved February 26, 2015, from https://www.ucl.ac.uk: https://www.ucl.ac.uk/anaesthesia/StudentsandTrainees/PainPathw aysIntroduction H.Cameron, M. (2009). Physical Agents in Rehabilitation . Introduction to Pain Pathways and Mechanism. (n.d.). Retrieved February 27, 2015, from https://www.youtube.com: https://www.youtube.com/watch?v=i5V_q7XqQN8