Chapter 2: Data Analysis
advertisement

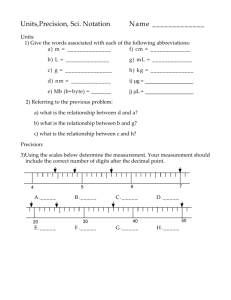
Dr. Chin Chu Measurements Temperature demonstration What have been learned here? Human senses are not reliable indicator of physical properties. We need instruments to give us unbiased determination of physical properties. A system must be established to properly quantify the measurements. Scales and units. Measurements Definition – comparison between measured quantity and accepted, defined standards (SI). Quantity: Property that can be measured and described by a pure number and a unit that refers to the standard. Measurement Requirements Know what to measure. Have a definite agreed upon standard. Know how to compare the standard to the measured quantity. Tools such as ruler, graduated cylinder, thermometer, balances and etc… Measurements – Units SI Units – (the metric system) Universally accepted Scaling with 10 Base Units: Time (second, s) Length (meter, m) Mass (kilogram, kg) Temperature (Kelvin, K) Amount of a substance (mole, mol) Electric current (ampere, A) Luminous intensity (candela, cd) Measurements - Temperature Temperature Scales: Celsius (°C, centigrade) Water freezing: 0 °C Water boiling: 100 °C Kelvin (K, SI base unit of temperature) Same spacing as in Celsius scale. Conversion: Celsius + 273 = Kelvin Fahrenheit (°F) Not the same spacing as the other two. Conversion: Fahrenheit = (5/9)(Celsius -32) Measurements – Units Derived Units: Volume volume length height depth Units: (length)3, such as cm3,m3, dm3 (liter) Density Defined as mass per unit volume the substance occupies. mass density volume Problem Solving Process THE PROBLEM 1. Read the problem carefully. 2. Be sure to understand what it is asking you. SOLVE THE UNKNOWN 1. Determine whether you need a sketch to solve the problem. 2. If the solution is mathematical, write the equation and isolate the unknowns. 3. Substitute the known quantities into the equation. 4. Solve the equation. 5. Continue the solution process until you solve the problem.. ANALYZE THE PROBLEM 1. Read the problem again. 2. Identify what you are given and list the known data. 3. Identify and list the unknowns. 4. Gather information you need from graphs, tables or figures. 5. Plan the steps you will follow to find the answer. EVALUATE THE ANSWER 1. Re-read the problem. Is the answer reasonable. 2. Check your math. Are the units and the significant figures correct? (Sect. 2.2 and 2.3) Problem Solving Process – Example THE PROBLEM: A metal cube is 2 cm on each edge and has a mass of 20 g. Is the cube made of pure aluminum? Density of pure aluminum is 2.7 g/cm3. THE APPROACH: Determining the nature of the metal, density is the parameter to compare. WHAT’S KNOWN: Density of pure Al WHAT’S UNKNOWN: THE MATH: Density = Mass/Volume WHAT ARE NEEDED: Mass and Volume Density of the metal cube THE MATH: Volume=(Edge)3 The edge of the cube is known. HOW TO CALCULATE THE VOLUME OF A CUBE? WHAT’S KNOWN: Mass WHAT’S UNKNOWN: Volume Problem Solving Process – Example THE SOLUTION: 1. Construct the logic flow chart. 2. Write backwards from the flow chart, the last step first then the 2nd last and so on. Actual solution of the problem: 1) Volume of the cube = (edge of the cube)3 = (2 cm) 3 = 8 cm3 2) Density of the cube = (mass of the cube)/(volume of the cube) = 20 g/8 cm3 = 2.5 g/cm3 3) Comparison between the densities: density of the metal cube (2.5 g/cm3) is less than the density of the pure Al (2.7 g/cm3) 4) Conclusion: the metal cube is not made of pure Al. Problem Solving Process – Challenge What determines whether the object will float or sink in water? Density of the object relative to water (1 g/cm3). Sink if density of the object is higher than water. Float if density of the object is smaller than water. For a solid piece of aluminum (Al),the density is 2.7 g/cm3. Given a piece of Al that weights 27.0 grams. •Will it float or sink in water? Why? •If your answer is sink, what would you do to make it float? Dr. Chin Chu Significant Figures Measurements are always done against a standard. When we measure something, we can (and do) always estimate between the smallest marks. 4.5 mm mm 1 2 3 4 5 Significant Figures The better marks the better we can estimate. Scientist always understand that the last number measured is actually an estimate, where the level of uncertainty is defined. 4.55 mm object mm 1 2 3 4 5 Significant Digits and Measurement Measurement Done with tools. The value depends on the smallest subdivision on the measuring tool. Significant Digits (Figures): consist of all the definitely known digits plus one final digit that is estimated in between the divisions. Significant Figures Only measurements have significant figures. Counted numbers and defined constants are exact and have infinite number of significant figures. A dozen is exactly 12 1000 mL = 1 L Being able to locate, and count significant figures is an important skill. Significant Figures - Examples Measured Value Uncertainty Ruler Division Known digits Estimated digit 1.07 cm +/-0.01 cm 0.1 cm 1, 0 7 3.576 cm +/-0.001 cm 0.01 cm 3,5,7 6 22.7 cm +/- 0.1 cm 1 cm 2, 2 7 Significant Figures: Examples What is the smallest mark on the ruler that measures 142.15 cm? ____________________ 142 cm? ____________________ 140 cm? ____________________ Does the zero count? We need rules!!! Rules of Significant Figures If there is a decimal point present start counting from the left to right until encountering the first nonzero digit. All digits thereafter are significant. If the decimal point is absent start counting from the right to left until encountering the first nonzero digit. All digits are significant. Rules of Significant Figures - Examples Pacific Ocean Example 1 Atlantic Ocean 78638 0.00078638 decimal point Example 2 78638 78638000 No decimal point Significant Figures - Exercise In the following measurements , identify the number of significant figures, uncertainty level and estimated digit: 120 cm 0.00347 kg 0.23400 L 11.24 s 1100. km 4.560 x 10-3 m 0.09720 g/mL Significant Figures - Answers In the following measurements , identify the number of significant figures, uncertainty level and estimated digit: 120 cm [2 sig. fig.; ±10cm; 2] 0.00347 kg [3 sig. fig.; ±0.00001kg; 7] 0.23400 L [5 sig. fig.; ±0.00001L; the last “0”] 11.24 s [4 sig. fig.; ±0.01s; 4] 1100. km [4 sig. fig.; ±1km; the last “0”] 4.560 x 10-3 m [4 sig. fig.; ±0.001x10-3m; 0] 0.09720 g/mL [4 sig. fig.; ±0.00001g/mL; 0] Rounding Rules Rounding is always from right to left. Look at the number next to the one you’re rounding. 0 - 4 : leave it 5 - 9 : round up With one exception: when the number next to the one you’re rounding is 5 and not followed by nonzero digits (a.k.a. followed by all zeros) – round up if the number (rounding to) is odd; don’t do anything if it is even. Rounding Rules Further explanation for the special rule regarding the last digit to be exactly 5: Rounding Rules - Examples Example 1 2.532 <5 leave it 2.53 Last significant digit Example 2 2.536 >5 2.54 round up Last significant digit Example 3 2.5351 >5 2.54 round up Last significant digit Example 4 2.5350 the 2.54 exception odd round up Last significant digit Rounding – Exercise Round the following measurements to the specified number of significant figures: 4.256 cm to 2 sig. fig. 123500 g to 3 sig. fig. 0.00374 L to 2 sig. fig. 2.3451 s to 3 sig. fig. 5.675 miles to 3 sig. fig. 0.34625 mm to 4 sig. fig. Rounding – Answers Round the following measurements to the specified number of significant figures: 4.256 cm to 2 sig. fig. [4.3 cm] 123500 g to 3 sig. fig. [124000 g] 0.00374 L to 2 sig. fig. [0.0037 L] 2.3451 s to 3 sig. fig. [2.35 s] 5.675 miles to 3 sig. fig. [5.68 miles] 0.34625 mm to 4 sig. fig. [0.3462 mm] Mathematical Operations Involving Significant Figures Addition and Subtraction The answer must have the same number of digits to the right of the decimal point as the value with the fewest digits to the right of the decimal point. Why? The result from the addition or subtraction would have the same precision as the least precise measurement. Mathematical Operations Involving Significant Figures Addition and Subtraction Example: 28.0 cm 23.538 cm 25.68 cm 28.0 cm 23.538 cm 25.68 cm 77.218 cm 77.2 cm 1. Arrange the values so that decimal points line up. 2. Do the sum or subtraction. 3. Identify the value with fewest places after decimal point. 4. Round the answer to the same number of places. Mathematical Operations Involving Significant Figures Multiplication and Division The answer must have the same number of significant figures as the measurement with the fewest significant figures. Mathematical Operations Involving Significant Figures Multiplication and Division Example: 28.0 cm 23.538 cm 25.68 cm 28.0 cm 23.538 cm 25.68 cm 16924.76352 cm3 16900 cm3 3 1. Carry out the operation. 2. Identify the value with fewest significant figures. 3. Round the answer to the same significant figures. Math Operation – Exercise Complete the following math calculations with proper significant figures: 12.45 m + 34 m = _____ m 1100 g + 123 g + 823.6 g = _______ g 23.45 L - 5.572 L = ______ L 24.1 mm x 2.7 mm = _______ mm2 0.965 m x 2.63 m x 0.5472 m = _______ m3 45.76 kg ÷ 25.67L = _________ kg/L Math Operation – Answers Complete the following math calculations with proper significant figures: 12.45 m + 34 m = [46] m 1200 g + 123 g + 823.6 g = [2100] g 23.45 L - 5.572 L = [17.88] L 24.1 mm x 2.7 mm = [65] mm2 0.965 m x 2.63 m x 0.5472 m = [1.39] m3 45.76 kg ÷ 25.67L = [1.783] kg/L Dr. Chin Chu How Reliable are Measurements? Multiple measurements are taken to ensure data integrity. Assessments have to be made regarding how close the data are to the actual value (accuracy) and how close those multiple measurements are relative to each other (precision). Let’s use a golf analogy Accurate? No Precise? Yes Accurate? Yes Precise? Yes Precise? No Accurate? Maybe? Accurate? Yes Precise? We cannot say! Accuracy vs. Precision - Exercise Three students measure the room to be 10.2 m, 10.3 m and 10.4 m across. Were they precise? Were they accurate? Accuracy vs. Precision - Answers Three students measure the room to be 10.2 m, 10.3 m and 10.4 m across. Were they precise? [Yes] Were they accurate? [Not sure since the actual width of the room was not provided.] Dr. Chin Chu Scientific Notations Mass of a proton is 0.00000000000000000000000000167262 kg Mass of an electron is 0.000000000000000000000000000000910939 kg Which one has more mass? Hard to handle those numbers, right? A better way has to be somewhere! Scientific Notations THE MATH OF 10’s 1 = 1 = 100 10 = 10 = 101 100 = 10x10 = 102 1,000 = 10x10x10 = 103 10,000 = 10x10x10x10x10 = 104 0.1 = 1/10 = 1/101 = 10-1 0.01 = 1/100 = 102 = 10-2 0.001 = 1/1000 = 1/103 = 10-3 0.0001 = 1/10000 = 1/104 = 10-4 Did you see the pattern? Scientific Notation The exponent of 10 indicate the number of digits away from the decimal point: Positive value: to the left of the decimal point. Negative value : to the right of the decimal point. It provides us a way to shorten very large (or small) numbers into manageable parts. Scientific Notation: multiple of two factors Factor 1: a number between 1 and 10. Factor 2: 10 raised to a power, or exponent Scientific Notation Example 1: 12670000 12670000 = 1267000 x 10 = 1267 00 x 10 x 10 = 12670 x 10 x 10 x 10 = 1267 x 10 x 10 x 10 x 10 = 126.7 x 10 x 10 x 10 x 10 x 10 = 12.67 x 10 x 10 x 10 x 10 x 10 x 10 = 1.267 x 10 x 10 x 10 x 10 x 10 x 10 x 10 = 1.267 x 107 Alternatively, 12670000 12670000. 7 6 5 4 3 2 1.267 x 107 1 Scientific Notation Example 2: 0.0000001267 0.0000001267 = 0.000001267 x (1/10) = 0.00001267 x (1/10) x (1/10) = 0.0001267 x (1/10) x (1/10) x (1/10) = 0.001267 x (1/10) x (1/10) x (1/10) x (1/10) = 0.01267 x (1/10) x (1/10) x (1/10) x (1/10) x (1/10) = 12.67 x (1/10) x (1/10) x (1/10) x (1/10) x (1/10) x (1/10) = 1.267 x (1/10) x (1/10) x (1/10) x (1/10) x (1/10) x (1/10) x (1/10) = 1.267 x 10-7 Alternatively, 0.0000001267 1 2 3 4 5 6 7 1.267 x 10-7 Scientific Notation – Prefixes Used with SI Units Prefix Symbol Factor Scientifi c Notation Example giga G 1,000,000,000 109 gigameter (Gm) mega M 1,000,000 106 megagram (Mg) kilo k 1,000 103 kilometer (km) deci d 1/10 10-1 deciliter (dL) centi c 1/100 10-2 centimeter (cm) milli m 1/1,000 10-3 milligram (mg) micro m 1/1,000,000 10-6 microgram (mg) nano n 1/1,000,000,000 10-9 nanometer (nm) pico p 1/1,000,000,000,000 10-12 picometer (pm) Scientific Notation – Operations Addition and Subtraction How does one add 34562 and 76541290? Add them in columns! 34562 +) 76541290 76575852 3.4562 x104 7.6541290x107 Obviously wrong answer! 11.1103290x10? Notice that the first digit 3 of the 1st number is right on top of the forth digit 4 of the 2nd number. How about move digits to line up? 3.4562 x 104 +) 7654.1290 x 104 7657.5852 x 104 76575852 The right answer! When adding or subtracting numbers written in scientific notation, one must be sure that the exponents are the same before doing the arithmetic! Scientific Notation – Operations Multiplication and Division Example 1 10 x 100 = 1000 101 102 1+2=3 103 Example 2 100 102 1 = 0.01 100 10-2 0 - 2 = -2 Add exponents for multiplication and subtract for division. Scientific Notation – Operations Multiplication and Division a x b x c x d= (a x c) x (b x d) axb = a x a cxd c c • Two-steps: • Multiply (or divide) the first factors. • Multiply (or divide) the 2nd exponent factors. Scientific Notation – Operations Examples – see the in-class worksheets. Dr. Chin Chu Percent Error Definitions: Accepted Value: a value that is considered true. Sometimes also referred to as “expected” value. Experimental Value: the value measured in the experiment Error: difference between an experimental value and the accepted value. Could be either positive or negative. – Percent Error: ratio of an error to an accepted value. Only positive values. error Percent Error = accepted value 100% = exp. - accepted accepted value 100% Representing Data – Graphing What is a graph? A visual display of data. “A picture is worth a thousand words.” Often used to make it easier to understand large quantity of data and relationships between parts of the data. Types of graphs/charts: Circle (a.k.a. Pie Charts) Bar Line Representing Data – Graphing Common Features of a Graph/Chart: Typical graph/chart is graphical and contains very little text. A title: most important, and generally appears above the main graph to provide a succinct description of what the data in the graph refers to. Axes: display of dimensions in the data. Scales Axis label: briefly describe the dimension represented. If numerical scales, units are usually included in the axis labels. Grid lines: major and minor. Legend: for displaying data with multiple variables. Representing Data – Graphing Histogram of Class Scores on the 2nd Quiz Graphing – Pie Chart What is this chart about? What is the percentage of seats occupied by the Conservatives? What is the percentage of seats occupied by the Liberals? Graphing – Line Chart Speed vs. Time What is this chart about? What happens with respect to speed as time elapses? Graphing – Line Chart Growth of a Tree Independent variable: 12 Height (m) 10 8 A (x1, y1) 6 4 B (x2, y2) 2 0 0 1 2 3 4 5 6 7 time (year) Dependent variable: height of the tree. Best fit line is straight. Linear relationship between the two. Direct related. Year y2 y1 y slope x2 x1 x • Positive: dependent variable increases with the independent variable • Negative: decreases . Graphing – Line Graph Steps to Use to Make a Line Graph: 1. 2. 3. 4. 5. 6. Identify the independent and dependent variables. Determine the range of data that needs to be plotted for each axis. Choose intervals for the axis to spread out the data. Number and label each axis. Plot the data points. Draw a best fit line for the data. The line may be straight or curved, and not all points may fall on the line. Give the graph a title. Representing Data – Graphing What do you see?