Answers
advertisement

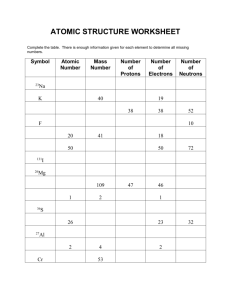
Mr. Sheehan’s Pd Name: Answers Chem Extra Chp 4 Practice 1. Summarize the FOUR parts of Dalton’s Atomic Theory. Hints: Made of..., same...., combine...., chemical reactions. a. All elements are made of atoms b. Atoms of the same element are identical c. Atoms combine in whole-number ratios to form compounds d. In chemical reactions, atoms are combined, separated, or rearranged – but never changed into atoms of another element 2. The atomic number shows the # of _protons____, which equals the number of _electrons. 3. Written as an equation, what does the mass number show? Protons + Neutrons 4. Fill in the rest of the table using the information provided. Do not use a periodic table. Atomic symbol Atomic number Protons Neutrons Electrons Mass Number B Na Ga Y Cu Tc Yb Ac Tl Fm Sg 5 11 31 39 29 43 70 89 81 100 106 5 11 31 39 29 43 70 89 81 100 106 6 13 37 50 35 57 102 136 125 159 159 5 11 31 39 29 43 70 89 81 100 106 11 24 68 89 64 100 172 225 206 259 265 Mr. Sheehan’s Pd Name: 5. Fill in the table given the element. Round the atomic mass to the nearest whole number. Atomic symbol Atomic Mass Neutrons Protons Electrons number Number 68 68 68 167 99 50 50 50 119 69 13 13 13 27 14 6. Find the average atomic mass of the problems below: (Mass * percent abundance) + (Mass * percent abundanc) + (… a. The relative abundance and atomic masses of copper isotopes are 69.2% for mass = 62.93 amu, and 30.8% for mass = 64.93 amu. Calculate the average atomic mass of copper. (0.692 x 62.93) + (0.308 x 64.92) = 63.6 amu b. Calculate the atomic mass of bromine. The two isotopes of bromine have atomic masses and relative abundance of 78.92 amu (50.69%) and 80.92 amu (49.31%) (0.5069 x 78.92) + (0.4931 x 80.92) = 79.90 amu c. Neon has two isotopes. Neon with 19.992 amu is 90.00% abundant and the mass of 21.99 amu is 10.00% abundant. Calculate the average atomic mass of neon. (0.9000 x 19.992) + (0.1000 x 21.99) = 20.19 amu d. What is the atomic mass of silicon if 92.21% of its atoms have mass 27.977 amu, 4.70% have mass 28.976, and 3.09% have mass 29.974 amu? (0.9221 x 27.977) + (0.0470 x 28.976) + (0.0309 x 29.974) = 28.1 amu