Diapositiva 1
advertisement

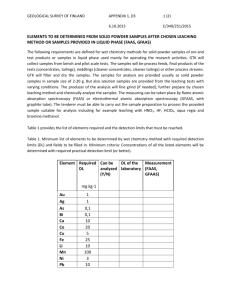
National Polytechnic School Extractive Metallurgy Department Quito - Ecuador VALORIZATION OF SOLID WASTE RICH IN NICKEL AND VANADIUM PRODUCED BY THE COMBUSTION OF FUEL OIL Ibujés Paulina, De la Torre Ernesto and Guevara Alicia The combustion of fuel oil in boilers Fuel oil Fly ash acumulation Slag incrustation The problem Fly ash Slag Fly ash methodology Acid leaching HNO3, HCl, H2SO4 25°C 24h 20 – 40 g/L Crystallization 75 °C solution Roasting 250 – 950 °C 0.5 – 2.0 h Characterization Atomic Absorption Spectrophotometer A X-ray diffraction (XRD) in a D8 advance equipment Acid leaching H2SO4 25°C 24h 20 – 200 g/L cake Stabilization Cake 33-60 % Cement 33-40 % Lime 10-33 % Fly ash characterization by Atomic Absorption Spectrophotometer Elements Concentration (%) Iron 5.42 Vanadium 5.31 Nickel 1.61 Calcium, sodium, potassium < 1.00 Fixed carbon 85.00 Moisture 1.24 Volatile matter 0.86 Mineralogical content by X-ray diffraction (XRD) equipment fly ash roasted at 550 - 950°C Mineral Formula Coulsonite FeV2O5 Sodium vanadium oxide Nickel vanadium oxide Calcium vanadium oxide Iron oxide Iron hydroxide oxide Grupo plagioclasa (albita, andesita, anortita) NaV6O15 / Na0,33V2O5 NiV2O6 Ca0,17V2O5 Fe2O3 Fe1,833(OH)0,5 O2,5 (Na,Ca)Al(Si,Al)Si2O8 Karelianite V2O3 Quartz SiO2 Magnesiun Aluminium iron oxide MgAl0,8Fe1,2 O4 Metal recovery in solution (%) Vanadium, nickel and iron recovery from stirred leaching (750rpm) with H2SO4 200 g/L and 25% solids on the fly ash burned at 350°C for 1.5 hours. 100 90 80 70 60 50 40 30 20 10 0 0 2 4 6 8 10 12 14 16 18 20 Leaching time (h) V Ni Fe 22 24 Crystallization of the strong solution The strong solution produced from stirred leaching (750rpm) with H2SO4 200 g/L and 25% solids on the fly ash burned at 350°C for 1.5 hours. Mineral Formula Approximate composition (%) Iron sulfate Fe2(SO4)3 60 Vanadium oxide V2O5 27 Morenosite NiSO4.7H2O 5 Jambornite (Ni,Fe,Ca)(OH)2(OH,S,H2O) 4 Stabilization of cake The cake produced from stirred leaching (750rpm) with H2SO4 200 g/L and 25% solids on the fly ash burned at 350°C for 1.5 hours Mineral Formula Approximate composition (%) Carbon C 90 Rhomboclase HFe(SO4)2.4H2O 10 The standard of public 38% Cake 31% Cement 31% Lime sewer system discharge (Ecuador) U.S. EPA 40 CFR 261.24 standards, EPA 2003 Slag Leaching HNO3, HCl, H2SO4, HNO3+HCl, NH3, NaOH, NaCl, NH4Cl, Na2CO3 Leaching H2SO4 + Br / H2O2 20-300 g/L 25-80°C Roasting 2h A scanning electron microscope (MEB-EDX) A X-ray diffraction (XRD) in a D8 advance equipment 20-100 g/L 450-550°C Roasting NaCl, Na2CO3 Characterization Atomic Absorption Spectrophotometer Leaching H2SO4 2h 450-550°C Leaching H2O 18-22% NaCl/Na2CO3 Crystallization Leaching H2SO4 Leaching Na2CO3 120-200 g/L 0.5-2.5 M Stabilization Slag characterization by Atomic Absorption Spectrophotometer Elements Concentration (%) Vanadium 28.72 Nickel 8.16 Sodium 1.62 Iron 1.60 Fixed carbon 46.57 Volatile matter 2.50 Moisture 1.01 Components distribution of the slag section by scanning electron microscope (MEB-EDX) Increase 200x V Increase 1500x Ni Si Fe O Mineralogical content of the slag by X-ray diffraction (XRD) in a D8 advance equipment Slag roasted at 550 and 650 °C Mineral Formula Calcium vanadium oxide Ca0,17V2O5 Sodium vanadium oxide Na0,33V2O5 / NaV6O15 Bannermanite Na0,76V6O15 Cristobalite SiO2 Grupo plagioclasa (albita, andesita, anortita) Nickel vanadium oxide (Na,Ca)Al(Si,Al)Si2O8 NiV2O6 Recovery of vanadium, nickel and iron in solution by mean slag leaching 100 90 80 70 60 50 40 30 20 10 0 V Ni Fe Metal recovery in solution (%) Vanadium, nickel and iron recovery in solution by agitated leaching (750 rpm) with H2SO4 200 g/L, 25% solids at 70°C. 100 90 80 70 60 50 40 30 20 10 0 0 4 V 8 12 16 Leaching time (h) Ni 20 Fe 24 Metal recovery in solution (%) Vanadium, nickel and iron recovery in solution from agitated leaching (750 rpm) of the solid residue (from H2SO4 leaching) with Na2CO3 2 M at 75°C. 100 90 80 70 60 50 40 30 20 10 0 0 4 8 12 16 20 Leaching time (h) V Ni Fe 24 Crystallization of the strong solutions Process Leaching 1 200 g/L H2SO4 70 °C 25 % solid Leaching 2 2 M Na2CO3 75 °C 25 % solid Approximate composition (%) Vanadium oxy sulphate V2O3(SO4)2 51 Nickel hidrate sulphate NiSO4.6H2O 27 Fe2(SO4)3 7 K2 NiSO4.6H2O 5 Na3H(CO3)2. H2O Na2CO3. H2O NaV6O16 Ca3(V2O7)(OH)2(H2O) 28 24 16 15 14 Mikasaite Nickel and potasium hidrate sulphate Trona Thermonatrita Barnesita Volborthita Calcium magnesium vanadium oxide CaMgV2O7 Stabilization of slag cake The cake produced from leaching sequence with H2SO4 and Na2CO3 was constituted by Formula Approximate composition (%) Vanadium nickel oxide Ni3(VO4)2 56 Calcium aluminum oxide CaAl2O4 15 Aluminum phospate AlPO4 8 Iron sulphur Fe7S8 6 Mineral 50% Cake 40% Cement 10% Lime The standard of public sewer system discharge (Ecuador) U.S. EPA 40 CFR 261.24 standards, EPA 2003 Conclusions Fly ash The fly ash has 5.42wt-% iron, 5.31wt-% vanadium and 1.61wt-% nickel, and amorphous material. (NaV6O15, NiV2O6, Na0.33V2O5, NiV2O6, Ca0,17V2O5, NaV6O15) Fly ash was roasted at 350°C for 1.5 hours and agitated leaching with H2SO4 200 g/L at 25°C, 25wt-% solids for 8 hours at 750 rpm in order to recover 91wt-% vanadium, 73wt-% nickel and 70wt-% iron in solution Conclusions Slag The slag has 28.72wt-% vanadium, 8.16wt-% nickel, 1.62wt-% iron. (Ca0,17V2O5, Na0,33V2O5, Na0,76V6O15, NaV6O15, NiV2O6, SiO2) Slag was leached with H2SO4 200 g/L at 70°C, 25wt-% solids for 4 hours stirring (750 rpm) followed by another leaching with Na2CO3 2 M, 75°C, 25wt-% solids and 8 hours in order to recover 75wt-% vanadium, 21wt-% nickel and 34wt-% iron The fly ash and slag processing is costly and complex. Their valorization for vanadium and nickel recovery is possible, but must be evaluated with large scale essays