Document

Gerald Karp
Cell and Molecular Biology
Fifth Edition
CHAPTER 5 Part 1
Aerobic Respiration and the Mitochondrion
Copyright © 2008 by John Wiley & Sons, Inc.
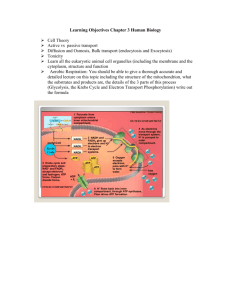
5.1 Mitochondrial structure and function
1. Living fibroblast
2. TEM
3. Sperm midpiece
1-4μm, 0.2- 1.0μm
1. Mitochondrial membranes
The outer membrane is thought to be homologous to an outer membrane present as part of the cell wall of certain bacterial cells.
The inner membrane is highly impermeable; all molecules and ions require special membrane transporters to gain entrance to the matrix.
2. The mitochondrial matrix
Possess ribosomes, circular DNA to manufacture their own RNAs and proteins
5.2 Oxidative metabolism in the mitochondrion
An overview of carbohydrate metabolism in eukaryotic cells
Pyruvate + HS-CoA + NAD + → acetyl
CoA + CO
2
+ NADH + H +
In matrix
1. The Tricarboxylic Acid (TCA) cycle
Acetyl CoA + 2 H
2
GDP + P i
→ 2 CO
2
O + FAD + 3 NAD + +
+ FADH
2
+ 3 NADH
+ 3H + + GTP +HS-CoA
The glycerol phosphate shuttle
Electrons are transferred from NADH to dihydroxyacetone phosphate (DHAP) to form glycerol 3-phosphate, which shuttles them into the mitochondrion.
These electrons then reduce FAD at the inner membrane, forming FADH the electron-transport chain.
2 which can transfer the electrons to a carrier of
An overview of carbohydrate metabolism in eukaryotic cells
2. The importance of reduced coenzymes in the formation of
ATP (Chemiosmosis)
1. High-energy electrons are passed from FADH2 or NADH to the first of a series of electron carriers in the electron transport chain.
2. The controlled movement of protons back across the membrane through an
ATP-synthesizing enzyme provides the energy required to form ATP from ADP.
A summary of the process of oxidative phosphorylation
5.3 The role of mitochondria in the formation of ATP
1. Electron transport
2. Oxidation-reduction potentials
3. Types of electron carriers
1. Oxidation – reduction potential
Oxidizing agents can be ranked in a series according to their affinity for electrons: the greater the affinity, the stronger the oxidizing agent.
Reducing agents can also be ranked according to their affinity for electrons:
The lower the affinity, the stronger the reducing agent
Reducing agents are ranked according to electron-transfer potential, such as
NADH is strong reducing agent, whereas those with low electrontransfer potential such as H
2 weak reducing agents.
O, are
Oxidizing and reducing agents occur as couples such as NAD+ and NADH.
Strong reducing agents are coupled to weak oxidizing agents and vice versa.
For example, in NAD + - NADH, NAD + is a weak oxidizing agent, in O
2
– H
2
O, O
2 is a strong oxidizing agent
The affinity of substances for electrons can be measured by instruments that detect voltage — oxidation-reduction (redox) potential.
2. Electron transport
1. Five of the nine reactions in matrix in Fig.
5.7 are catalyzed by dehydrogenases that transfer pairs of electrons from substrates to coenzymes, NADH and FADH
2
→ electrontransport chain
2. NADH and FADH mitochondria.
2 dehydrogenase are located in the inner membrane of
3. Types of electron carriers
Flavoproteins
Cytochromes
Ubiquinone
Iron-sulfur proteins
Electron-transport complexes
1. Complexes I, II, III, IV ----Fixed in place
2. I, III, VI in which the transfer of electrons is accompanied by a major release of free energy.
2. Ubiquinone (lipid-soluble), cytochrome c
(soluble protein in the intermembrane space)-
---move within or along the membrane
I, III, VI are described as proton pump which drive the production of ATP .
5.5 The mechinery for ATP formation
1. The structure of ATP synthase
2. The basis of ATP formation according to the binding change mechanism
3. Other roles for the proton-motive force in addition to ATP synthesis
RECALL THAT:
1. Enzymes do not affect the equilibrium constant of the reaction they catalyze
2. Enzymes are capable of catalyzing both the forward and reverse reactions
Mammalian liver has Ca 15000 copies of ATP synthase.
Homology are found in the bacterial plasma membrane, inner membrane of mitochondria, and thylakoid membrane
F
1
: head (90A) in matrix
F0: basal region embedded in the inner membrane
The structure of
ATP synthase
2. The basis of ATP formation according to the binding change mechanism
1979 Paul Boyer (UCLA): published a hypothesis “ binding change mechanism ”
Using the proton gradient to drive the catalytic machinery:
1. What is the path taken by protons as they move through the F
0 complex?
2. How does this movement lead to the synthesis of ATP?
3. The role of the F
0 synthase portion of ATP
All of the following presumptions were confirmed by data collection between
1995-2001
1. The c subunit of the F
0 base were assembled into a ring that resides within the lipid bilayer.
2. The c ring is physically bound to the γsubunit of the stalk.
3. The “downhill” movement of protons through the membrane drives the rotation of the ring of c subunit.
4. The rotation of the c ring of F
0 provides the twisting force that drives the rotation of the attached γsubunit, leading to the synthesis and release of ATP.
“ Seeing is beliving ”
Masasuke Yoshida et al. at the Tokyo
Institute Technology in Japan
They devised an system to watch the enzyme catalyze the reverse reaction from the normally operating cell.
Only two biological structures are known that contain rotating parts
1. ATP synthase
2. Bacterial flagella
3. Both are described as rotary
“ nanomachines ”
4. Invent nanoscale devices
5. Someday, human may be using ATP instead of electricity to power some of their most delicate instruments.
Rotation of the c ring drives rotation of the attached γsubunit
H + movements drive the rotation of the c ring
4. From the middle of the “a” subunit into the matrix
1. Each “a” subunit has two halfchannels that are physically separate
2. From intermembrane space into “a” subunit
3. Binding of the H+ to the carboxyl group of aspartic acid generates a major conformational change in the c subunit to rotate 30 o in a Counterclockwise direction.
1. one H+ would remove the ring 30 o
2. In this case, the association/dissociation of 4 protons in the manner described would move the ring 120 o .
3. This would drive a corresponding rotation of the attached γsubunit 120 o and lead to release newly synthesized ATP.
4. The translocation of 12 protons would lead to the full 360 o rotation of the c ring and γunit and synthesis of 3 molecules of ATP.
Other roles for the proton-motive force in addition to ATP synthesis