CHARLES LAW PRACTICE PROBLEMS
advertisement

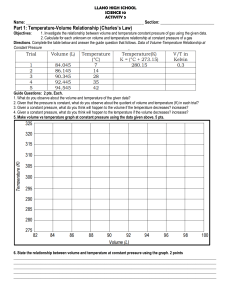
CHARLES LAW PRACTICE PROBLEMS NAME____________________ 1. State Charles Law: 2. What variables must be held constant for Charles Law to be true? 3. This gas law works only if temperature is recorded in ______________________ units. 4. What equation is used to convert from Celsius to these desired units? 5. Convert the following temperature readings into the desired units: 25.0˚C = 100˚C = -273˚C = 6. A gas at 20.0˚C is increased to 40.0˚C, will its volume: A) go up? B) double? C) if the original volume was 1.00 l, what would the new volume be? D) what would the new volume be if the original volume was 3.50 l ? 7. 8. A sample of O2 under 2.00 atm occupies 500 ml at 25.0˚C. A) What volume will the sample occupy at 0.0˚C ? B) What temperature will be needed to produce a volume of 100 ml ? If a gas has a pressure of 450 mm Hg and a volume of .250 l at a temperature of 22.0˚C, what would the new volume be at 450 mm Hg and 85.0˚C ?