Preanalytical Variability and the Stability of Human Plasma Proteins
advertisement
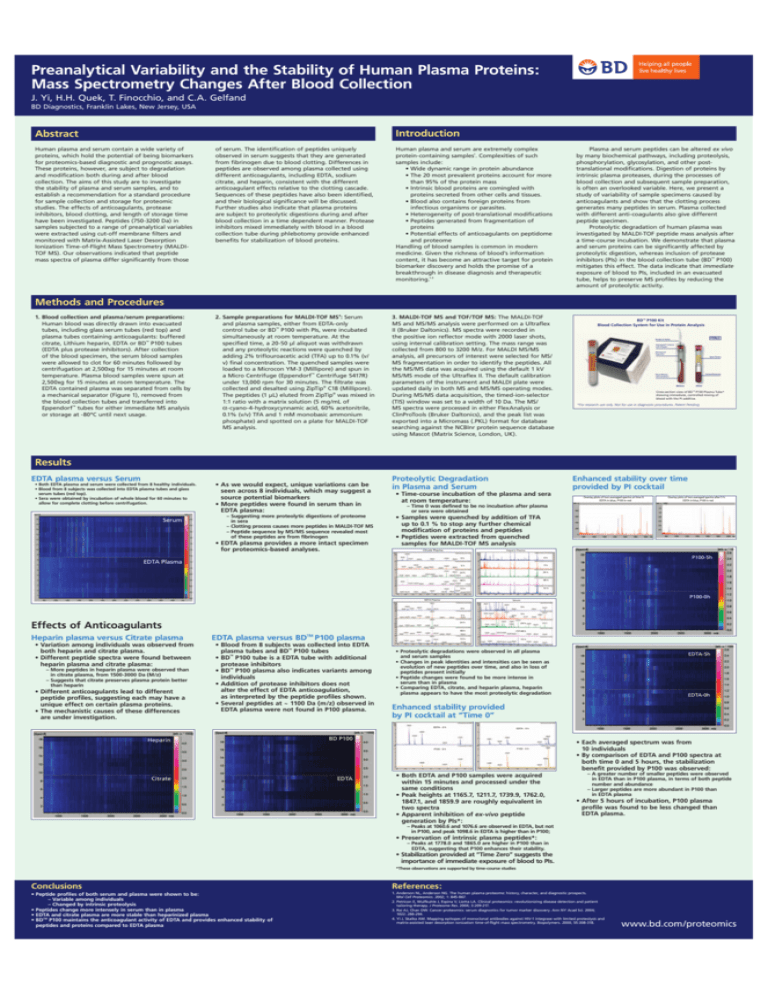
Preanalytical Variability and the Stability of Human Plasma Proteins: Mass Spectrometry Changes After Blood Collection J. Yi, H.H. Quek, T. Finocchio, and C.A. Gelfand BD Diagnostics, Franklin Lakes, New Jersey, USA Introduction Abstract Human plasma and serum contain a wide variety of proteins, which hold the potential of being biomarkers for proteomics-based diagnostic and prognostic assays. These proteins, however, are subject to degradation and modification both during and after blood collection. The aims of this study are to investigate the stability of plasma and serum samples, and to establish a recommendation for a standard procedure for sample collection and storage for proteomic studies. The effects of anticoagulants, protease inhibitors, blood clotting, and length of storage time have been investigated. Peptides (750-3200 Da) in samples subjected to a range of preanalytical variables were extracted using cut-off membrane filters and monitored with Matrix-Assisted Laser Desorption Ionization Time-of-Flight Mass Spectrometry (MALDITOF MS). Our observations indicated that peptide mass spectra of plasma differ significantly from those of serum. The identification of peptides uniquely observed in serum suggests that they are generated from fibrinogen due to blood clotting. Differences in peptides are observed among plasma collected using different anticoagulants, including EDTA, sodium citrate, and heparin, consistent with the different anticoagulant effects relative to the clotting cascade. Sequences of these peptides have also been identified, and their biological significance will be discussed. Further studies also indicate that plasma proteins are subject to proteolytic digestions during and after blood collection in a time dependent manner. Protease inhibitors mixed immediately with blood in a blood collection tube during phlebotomy provide enhanced benefits for stabilization of blood proteins. Human plasma and serum are extremely complex protein-containing samples1. Complexities of such samples include: • Wide dynamic range in protein abundance • The 20 most prevalent proteins account for more than 95% of the protein mass • Intrinsic blood proteins are comingled with proteins secreted from other cells and tissues. • Blood also contains foreign proteins from infectious organisms or parasites. • Heterogeneity of post-translational modifications • Peptides generated from fragmentation of proteins • Potential effects of anticoagulants on peptidome and proteome Handling of blood samples is common in modern medicine. Given the richness of blood’s information content, it has become an attractive target for protein biomarker discovery and holds the promise of a breakthrough in disease diagnosis and therapeutic monitoring.1-3 2. Sample preparations for MALDI-TOF MS4 : Serum and plasma samples, either from EDTA-only control tube or BD™ P100 with PIs, were incubated simultaneously at room temperature. At the specified time, a 20-50 µl aliquot was withdrawn and any proteolytic reactions were quenched by adding 2% triflouroacetic acid (TFA) up to 0.1% (v/ v) final concentration. The quenched samples were loaded to a Microcon YM-3 (Millipore) and spun in a Micro Centrifuge (Eppendorf™ Centrifuge 5417R) under 13,000 rpm for 30 minutes. The filtrate was collected and desalted using ZipTip® C18 (Millipore). The peptides (1 µL) eluted from ZipTip® was mixed in 1:1 ratio with a matrix solution (5 mg/mL of α-cyano-4-hydroxycynnamic acid, 60% acetonitrile, 0.1% (v/v) TFA and 1 mM monobasic ammonium phosphate) and spotted on a plate for MALDI-TOF MS analysis. 3. MALDI-TOF MS and TOF/TOF MS: The MALDI-TOF MS and MS/MS analysis were performed on a Ultraflex II (Bruker Daltonics). MS spectra were recorded in the positive ion reflector mode with 2000 laser shots, using internal calibration setting. The mass range was collected from 800 to 3200 M/z. For MALDI MS/MS analysis, all precursors of interest were selected for MS/ MS fragmentation in order to identify the peptides. All the MS/MS data was acquired using the default 1 kV MS/MS mode of the Ultraflex II. The default calibration parameters of the instrument and MALDI plate were updated daily in both MS and MS/MS operating modes. During MS/MS data acquisition, the timed-ion-selector (TIS) window was set to a width of 10 Da. The MS/ MS spectra were processed in either FlexAnalysis or ClinProTools (Bruker Daltonics), and the peak list was exported into a Micromass (.PKL) format for database searching against the NCBInr protein sequence database using Mascot (Matrix Science, London, UK). Plasma and serum peptides can be altered ex vivo by many biochemical pathways, including proteolysis, phosphorylation, glycosylation, and other posttranslational modifications. Digestion of proteins by intrinsic plasma proteases, during the processes of blood collection and subsequent sample preparation, is often an overlooked variable. Here, we present a study of variability of sample specimens caused by anticoagulants and show that the clotting process generates many peptides in serum. Plasma collected with different anti-coagulants also give different peptide specimen. Proteolytic degradation of human plasma was investigated by MALDI-TOF peptide mass analysis after a time-course incubation. We demonstrate that plasma and serum proteins can be significantly affected by proteolytic digestion, whereas inclusion of protease inhibitors (PIs) in the blood collection tube (BD™ P100) mitigates this effect. The data indicate that immediate exposure of blood to PIs, included in an evacuated tube, helps to preserve MS profiles by reducing the amount of proteolytic activity. Methods and Procedures 1. Blood collection and plasma/serum preparations: Human blood was directly drawn into evacuated tubes, including glass serum tubes (red top) and plasma tubes containing anticoagulants: buffered citrate, Lithium heparin, EDTA or BD™ P100 tubes (EDTA plus protease inhibitors). After collection of the blood specimen, the serum blood samples were allowed to clot for 60 minutes followed by centrifugation at 2,500xg for 15 minutes at room temperature. Plasma blood samples were spun at 2,500xg for 15 minutes at room temperature. The EDTA contained plasma was separated from cells by a mechanical separator (Figure 1), removed from the blood collection tubes and transferred into Eppendorf™ tubes for either immediate MS analysis or storage at -80ºC until next usage. "$©0+IT "LOOD#OLLECTION3YSTEMFOR5SEIN0ROTEIN!NALYSIS .EEDLE(OLDER (EMOGARD©#LOSURE WITH3TOPPER -ECHANICAL 3EPARATOR 3PUN0LASMA &ORMED%LEMENTS "LOOD-IXING WITH)NHIBITORS "EFORE !FTER #ROSSSECTIONVIEWOF"$©00LASMA4UBE SHOWINGIMMEDIATECONTROLLEDMIXINGOF BLOODWITHTHE0)ADDITIVE &ORRESEARCHUSEONLY.OTFORUSEINDIAGNOSTICPROCEDURES0ATENT0ENDING Results EDTA plasma versus Serum • Both EDTA plasma and serum were collected from 8 healthy individuals. • Blood from 8 subjects was collected into EDTA plasma tubes and glass serum tubes (red top). • Sera were obtained by incubation of whole blood for 60 minutes to allow for complete clotting before centrifugation. 3ERUM • As we would expect, unique variations can be seen across 8 individuals, which may suggest a source potential biomarkers • More peptides were found in serum than in EDTA plasma: – Suggesting more proteolytic digestions of proteome in sera – Clotting process causes more peptides in MALDI-TOF MS – Peptide sequence by MS/MS sequence revealed most of these peptides are from fibrinogen • EDTA plasma provides a more intact specimen for proteomics-based analyses. Proteolytic Degradation in Plasma and Serum Enhanced stability over time provided by PI cocktail • Time-course incubation of the plasma and sera at room temperature: #ITRATE0LASMA (EPARIN0LASMA H H H H H H H H H H %$4!0LASMA H Effects of Anticoagulants – More peptides in heparin plasma were observed than in citrate plasma, from 1500-3000 Da (M/z) – Suggests that citrate preserves plasma protein better than heparin • Different anticoagulants lead to different peptide profiles, suggesting each may have a unique effect on certain plasma proteins. • The mechanistic causes of these differences are under investigation. EDTA plasma versus BDTM P100 plasma • Blood from 8 subjects was collected into EDTA plasma tubes and BD™ P100 tubes • BD™ P100 tube is a EDTA tube with additional protease inhibitors • BD™ P100 plasma also indicates variants among individuals • Addition of protease inhibitors does not alter the effect of EDTA anticoagulation, as interpreted by the peptide profiles shown. • Several peptides at ~ 1100 Da (m/z) observed in EDTA plasma were not found in P100 plasma. H H H H H H • Proteolytic degradations were observed in all plasma and serum samples • Changes in peak identities and intensities can be seen as evolution of new peptides over time, and also in loss of peptides present initially • Peptide changes were found to be more intense in serum than in plasma • Comparing EDTA, citrate, and heparin plasma, heparin plasma appears to have the most proteolytic degradation %$4!H %$4!H "$0 %$4! %$4!H Enhanced stability provided by PI cocktail at “Time 0” 0H #ITRATE 0H H H %$4!H (EPARIN 0H 3ERUM H • Variation among individuals was observed from both heparin and citrate plasma. • Different peptide spectra were found between heparin plasma and citrate plasma: /VERLAYPLOTSOFTWOAVERAGEDSPECTRAAFTERH %$4!INBLUE0INRED • Samples were quenched by addition of TFA up to 0.1 % to stop any further chemical modification of proteins and peptides • Peptides were extracted from quenched samples for MALDI-TOF MS analysis %$4!0LASMA Heparin plasma versus Citrate plasma /VERLAYPLOTSOFTWOAVERAGEDSPECTRAATTIME %$4!INBLUE0INRED – Time 0 was defined to be no incubation after plasma or sera were obtained 0H • Both EDTA and P100 samples were acquired within 15 minutes and processed under the same conditions • Peak heights at 1165.7, 1211.7, 1739.9, 1762.0, 1847.1, and 1859.9 are roughly equivalent in two spectra • Apparent inhibition of ex-vivo peptide generation by PIs*: • Each averaged spectrum was from 10 individuals • By comparison of EDTA and P100 spectra at both time 0 and 5 hours, the stabilization benefit provided by P100 was observed: – A greater number of smaller peptides were observed in EDTA than in P100 plasma, in terms of both peptide number and abundance – Larger peptides are more abundant in P100 than in EDTA plasma • After 5 hours of incubation, P100 plasma profile was found to be less changed than EDTA plasma. – Peaks at 1060.6 and 1076.6 are observed in EDTA, but not in P100, and peak 1098.6 in EDTA is higher than in P100; • Preservation of intrinsic plasma peptides*: – Peaks at 1778.0 and 1865.0 are higher in P100 than in EDTA, suggesting that P100 enhances their stability. • Stabilization provided at “Time Zero” suggests the importance of immediate exposure of blood to PIs. *These observations are supported by time-course studies Conclusions References: • Peptide profiles of both serum and plasma were shown to be: – Variable among individuals – Changed by intrinsic proteolysis • Peptides change more intensely in serum than in plasma • EDTA and citrate plasma are more stable than heparinized plasma • BDTM P100 maintains the anticoagulant activity of EDTA and provides enhanced stability of peptides and proteins compared to EDTA plasma 1. Anderson NL, Anderson NG. The human plasma proteome: history, character, and diagnostic prospects. Mol Cell Proteomics. 2002; 1: 845-867. 2. Petricon E, Wulfkuhle J, Espina V, Liotta LA. Clinical proteomics: revolutionizing disease detection and patient tailoring therapy. J Proteome Res. 2004; 3:209-217. 3. Rai AJ, Chan DW. Cancer proteomics: serum diagnostics for tumor marker discovery. Ann NY Acad Sci. 2004; 1022: 286-294. 4. Yi J, Skalka AM. Mapping epitopes of monoclonal antibodies against HIV-1 integrase with limited proteolysis and matrix-assisted laser desorption ionization time-of-flight mass spectrometry. Biopolymers. 2000; 55:308-318. www.bd.com/proteomics