Activity 1.3.3 Thermodynamics Answer Key
advertisement

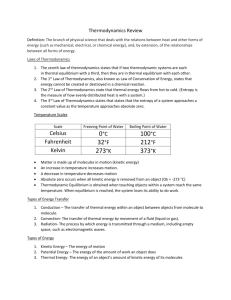
Activity 1.3.3 Thermodynamics Answer Key Introduction Think back to the last time someone complained about a door being left open. What did you notice about the temperature within the room as a result of the open door? In Activity 1.3.3 you will investigate the effects of work, thermal energy, and energy on a system, as in the case of the room with the door left open. Procedure Answer the following questions as your teacher discusses the Introduction to Thermodynamics presentation. 1. Define thermodynamics. The study of the effects of work, thermal energy, and energy on a system 2. List three examples of a thermodynamic system. a. Nuclear power b. Electronic heat sink c. Rocket launch 3. Define thermal energy. Kinetic energy in transit from one object to another due to temperature difference 4. Define temperature. The average kinetic energy of particles in an object 5. Fill in the table below with the correct scale and unit. Scale Celsius Fahrenheit Kelvin Freezing point of water 0°C 32°F 273K Boiling point of water 100°C 212°F 373K © 2012 Project Lead The Way, Inc. Principles of Engineering Activity 1.3.3 Thermodynamics Answer Key – Page 1 6. Define absolute zero. All kinetic energy is removed - 0 K 7. Define thermal equilibrium. Touching objects within a system reach the same temperature 8. Define the Zeroth Law of Thermodynamics. If two systems are separately found to be in thermal equilibrium with a third system, the first two systems are in thermal equilibrium with each other. 9. Define the First Law of Thermodynamics. Thermal energy can change form and location, but it cannot be created or destroyed. 10. List two ways thermal energy can be increased in a system. a. Adding thermal energy b. Performing work on the system 11. Define the Second Law of Thermodynamics. Thermal energy flows from hot to cold. 12. Define entropy. The measure of how evenly distributed heat is within a system 13. Define convection. The transfer of thermal energy by movement of fluid (liquid or gas) 14. List two examples of convection. a. Weather b. Boiler systems 15. Define conduction. The transfer of thermal energy within an object or between objects from molecule to molecule 16. List two examples of conduction. a. Metal spoon b. Heat through a wall © 2012 Project Lead The Way, Inc. Principles of Engineering Activity 1.3.3 Thermodynamics Answer Key – Page 2 Conduction Equations: 17. Define the following variables. Q = Energy transferred m = Mass of material absorbing or releasing energy c = Specific heat capacity of a material (J/kg °C) P = Rate of energy transfer Δ t = Change in time k = Thermal conductivity A = Area of thermal conductivity L = Thickness of material Δ T = Change in Temperature © 2012 Project Lead The Way, Inc. Principles of Engineering Activity 1.3.3 Thermodynamics Answer Key – Page 3 (18–22) A 1 kg piece of aluminum metal at 90 °C is placed in 4 liters (= 4 kg) of water at 25 °C. Determine the final temperature (Tf). 18. List all known values. mAl = 1.00 kg mwater = 4.00 kg TAl = 90.0 °C Twater = 25.0 °C Cp-Al = 900. J/kg °C Cp-water = 4184 J/kg °C 19. List all unknown values. Tfinal 20. Select equations. 21. Apply known values. 22. Solve. 81,000J - 900. (J/ oC)(Tf ) = 16,720(J/ oC) (Tf ) - 418,000J (81,000 + 418,000)J = (16,720 + 900.)(J/ oC) (Tf ) Tf = (81,000 + 418,000)J 499,000 J o = = 28.3 C o o (16,720 + 900.)(J/ C) 17,620(J/ C) © 2012 Project Lead The Way, Inc. Principles of Engineering Activity 1.3.3 Thermodynamics Answer Key – Page 4 (23–27) The top of a 3/16 inch thick acrylic testing box is covered with an unknown ½ inch insulation material (black). The dimensions of box are 10 inch x 10 inch on each side. The sides and are wrapped with a control insulting material (blue) so that the almost all heat loss is through the 10 inch x 10 inch unknown insulating material on the top of the box (assume no heat is lost from the bottom or sides of the box). Determine the thermal conductivity for the insulating material if a 25 W bulb is used to heat the box. You may assume the only heat loss from the box occurs through the 10 x 10 test area. The bulb maintains the inside temperature at 10 ºC higher than the outside temperature. 23. List all known values. A = 10.0 in. x 10.0 in. = 0.254 m x 0.254 m = 0.064516 m2 ΔT = 10.0 °C P = 25.0 W L = 0.50 in. = 0.0127 m 24. List all unknown values. k = Thermal conductivity 25. Select equations. 26. Apply known values. k= 25.0W×0.0127m 0.064516m 2 ×10.0°C 27. Solve. © 2012 Project Lead The Way, Inc. Principles of Engineering Activity 1.3.3 Thermodynamics Answer Key – Page 5 k = 0.49 J s× m ×°C © 2012 Project Lead The Way, Inc. Principles of Engineering Activity 1.3.3 Thermodynamics Answer Key – Page 6 28. Define U-value. Measure of a material’s ability to conduct heat 29. Define R-value. Measure of a material’s ability to resist heat movement (30–31) Use the provided R-value chart and illustration below to calculate the Rvalue of the wall cavity and the R-value at the stud location. 30. Wall cavity R-value 5/8 drywall = 0.56 Fiberglass batt = 11.00 (3.5 in. x 3.142) Extruded polystyrene = 4.00 Wood bevel lap siding = 0.80 0.56 + 11.00 + 4.00 + 0.80 = R16.36 31. R-value at stud location 5/8 drywall = 0.56 2 x4 stud = 4.38 Extruded polystyrene = 4.00 Wood bevel lap siding = 0.80 0.56 + 4.38 + 4.00 + 0.80 = R9.74 © 2012 Project Lead The Way, Inc. Principles of Engineering Activity 1.3.3 Thermodynamics Answer Key – Page 7 32. Define radiation. The process by which energy is transmitted through a medium, including empty space, as electromagnetic waves 33. List two examples of radiation. a. Microwave oven b. Heat transfer from the human body 34. Define Stefan’s Law. All objects lose and gain thermal energy by electromagnetic radiation. (35–39) A student travels on a school bus in the middle of winter from home to school. The school bus temperature is 58 °F. The student’s skin temperature is 91.4 °F. Determine the net energy transfer from the student’s body during the 20 min ride to school due to electromagnetic radiation. Note: Skin emissivity is 0.90, and the surface area of the student is 1.50 m2. 35. List all known values. A = 1.50 m2 e = 0.90 ϭ = 5.6696 x 10-8 w/ m2 k 4 t = 20.00 min = 1200. s Bus temperature = 58.0 °F = 287.60 K Skin temperature = 91.4 °F = 306.15 K 36. List all unknown values. P = Rate of energy transfer Q = Energy transfer 37. Select equations. Q = P • Δt Pnet = σAe(T 4 - T 4 ) © 2012 Project Lead The Way, Inc. Principles of Engineering Activity 1.3.3 Thermodynamics Answer Key – Page 8 38. Apply known values to equations. Pnet = σAe(T 4 - T 4 ) Solve for (T4 –T4) 287.604 K = 6,841,566,093.34 K 306.154 K = 8,784,904,710.59 K 8,784,904,710.59 K - 6,841,566,093.34 K = 1,943,338,617.25 K Pnet = (5.6696 x 10-8 w/ m2 k 4) x (1.50 m2) x (0.90) x (1,943,338,617.25 K) = 148.74 W Q = 148.74 W x 1200. s 39. Solve. Q = 178,000 J 40. Define geothermal energy. Energy generated from the thermal energy stored beneath the Earth’s surface © 2012 Project Lead The Way, Inc. Principles of Engineering Activity 1.3.3 Thermodynamics Answer Key – Page 9 R-Value Chart Construction Material ½ in. Drywall 5/8 in. Drywall Particle Board – ½ in. Particle Board – ¾ in. Fiberboard ½ in. Extruded Polystyrene 1 in. Extruded Polystyrene 1 ½ in. Foil-faced Polyisocyanurate 1 in. 2x4 2x6 Hardwood Masonry Systems Brick 4 in. common Brick 4 in. face Concrete Block – Normal wt. 12 in. empty core Concrete Block – Light wt. 12 in. empty core Cement Mortar Sand and Gravel Stucco Roofing Asphalt Roll Asphalt Shingle Slate Wood R-Value 0.45 0.56 0.63 0.94 1.32 4.00 6.00 7.20 4.38 6.88 0.90 R-Value 0.80 0.44 1.23 2.60–2.30 0.20 0.60 0.20 R-Value 0.15 0.44 0.05 0.94 Siding Wood Shingles Wood Drop Wood Bevel Lapped Aluminum/Steel – Hollow Aluminum/Steel – with 3/8 in. Backer Insulation Fiberglass Batt Blankets – Rock Wool Loose Fill – Cellulose Loose Fill – Fiberglass 0.7 lb/cu.ft Loose Fill – Rock Wool Loose Fill – Vermiculite Extruded Polystyrene R-Value 0.87 0.79 0.80 0.61 1.82 R-Value per in. 3.142 3.0–3.8 2.8–3.7 2.2–4.0 3.1 2.2 4.00 © 2012 Project Lead The Way, Inc. Principles of Engineering Activity 1.3.3 Thermodynamics Answer Key – Page 10