RADTOACTIVITY MEASUREMENT OF HALF.LIFE
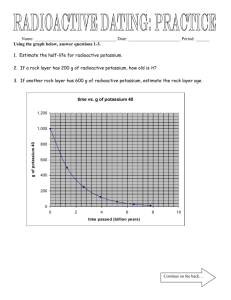
advertisement

Experlment# i3
# 13
Experiment
RADTOACTIVITY MEASUREMENTOF HALF.LIFE
Purpose: To measure the half-lives of Ag-108 and Ag-110. To study the relative
absorption of radiation in maffer.
Apparatus: activatedsilver foil, Geiger-Mueller counter, digital clock timer.
References:
lntroduction:
One way of looking at a nucleus is to think of it as a box in which there are many
particles, each with some enerry. These particles continually exchangeenergy and, in
some cases,an individual particle may acquire sufficient energy to penetratethe walls of
the box. In the caseof stablenuclei, the particles cannotget out of the box and the nuclei
"live" forever. ln the case of unstable nuclei, a particle does eventually get out of the
box. Some unstable nuclei have high, thick walls -- these are the longJived nuclei.
Others have low, thin walls -- these are the short-lived nuclei, those that have a high
probability of emitting particles and changing into differen! more stablenuclei.
For all radioactive isotopes one finds that the "decay " curue (i.e., the plot of the
number of disintegrationsper second as a function of time) appearsas shown in the graph
below (RadioactiveDecay of Silver 108and 110).When there are very many radioactive
nuclei in a sample, then the number of disintegrations per second can be described
extremely well by a probability curve. Even though each decay is a random even! the
totality of eventsis describedby a well defined equation. In the caseof nuclear decay,
the equationdescribingthe number of nuclei remainingattime t is an exponentialdecay
curve:
N(t) : No e'ttr
(l)
where t is the elapsedtime, N(t) is the number of nuclei that have not decayedafter the
time t, No is the number of radioactive nuclei present at time t : 0, and r is the mean
lifetime of the nucleus. Frequently, it is more convenient to express the exponential
decay in terrns of the "half-life" of the nucleus, the time required for half of the nuclei to
disintegrate. The relationship befween the half-life , T, , and the mean life, is the
following:
(2)
T,n : (ln 2) t -- 0.693 t
so we can write the decayequationin termsof T, :
'0'6e3t/r%
N(t) - No 0
(3)
Note that the clock can be started at any instant. Hence, in a time Ty,,half the starting
number of nuclei will have decayedregardlessof when the clock was started.
-54-
Experiment
# 13
The magnitudeof the decayrate, R(t) - dNidt (numberof disintegrationsper second),
is simply relatedto equation(3):
(4)
R(t)=& e'o'6e3ttrA
_
where R.
0.693N, / T,t,.
Graph A (p.58) shows a plot of data on linear scale gaph paper. One can exfiact
information about exponential behavior as linear information by using semi-logarithrnic
scale gaph paper. Graph B (p.58) is a plot of the samedata as is used for Graph A;
however, it is now plotted on semi-log paper. Why and to what advantage? Taking the
ln of both sidesof eq. 4 gives
ln R(t) : ln R{ - 0.693t lT%
(s)
which is an equationof a snaight line of the form y = mx + b. This makes analysis of
the data to determinethe half-life much simpler, especially when there are present two or
more radioactiveisotopes. Data analysiswill be dissussedin detail later.
The Geiger-MuellerCounter:
One of the frst devices used to detect radioactivity was a Geiger-Mueller tube,
which is a gas filled cylinder with a very thin metal wire down its center. A voltage
difference is maintained between the center wire and the cylinder. If radiation interacts
with the gas in the cylinder, ionization of the gas atoms takes place. Because of the
voltage difference, the electronsmove toward the center wire and the positive ions rnove
toward the outer cylinder. This is an electrical dischargeproducing a pulse of current
which can be counted. Each pulse indicates that a nucleus has decayed and the decay
rate, R(t), is measuredby the number of pulsesper second.
Warning: Do not exceed the Voltage setting for the GM tube as listed for each
instrument. Note the very thin beryllium window on the front of the tube. This window is
fragile - keep all objectsaway from it.
Thermal Neutron Activation:
Someradioactive isotopesoccur in nature. Somestableisotopes,when bornbarded
with nuclear particles of appropriateenergy, may be convertedto different isotopes which
are unstable (radioactive). In this exercise, stable silver isotopes are bombarded by
neutronsand made into radioactiveisotopesof silver.
ln the room adjacentto the laboratory is located a large tub of water, the hydrogen
of which absorbs neufrons. This tub of water seryes as a protective shield against
neutrons. ln the center of the tub of water is a small amount of polonium, which is
radioactive and emits alpha particles (helium nuclei). The alpha particles strike a
berylliumtargetandproduce
neufrons
by thefollowingnuclearreaction:
+
Beqyllium(9) o(4)
-55-
Experiment
i: -.3
Experunent# l3
When the silver foil is insertedinto the tub, some of the silver nuclei will capture a
neutronthus creatinga new isotopewhichis radioactive.There are two stableisotopesof
silverthat can captureneutronsandbecomeradioactive:
(i) Ag-107 + neutron
Tn (Ag-108) _ 2.41min.
(ii) Ag-109 + neutron
T, (Ag-110) : 24.4 sec.
Procedure- PartI.
Familiarizeyourself with the apparatus.Check to see that the voltagefor the
Geiger-Mueller tube is off. Practicesynchronizingthe starting and stoppingof the
counterwith the clock timer, using the counterin the test mode.In the test mode,the
cowttergenerates60 counts/sec
internally. Let the clock timer run continuouslyas you
start and stop the counteqalternatelycountingfor 10 secondsand waiting for 10 seconds
as describedbelow (your instructorwill demonstrate).
After each l0-second countinginterval, record the information and clear the
counterduring the next lO-secondinterval. Do not stopthe clock timer !! The clock is
recording the total elapsedtime. You must be preparedto restart the corurterat the
beginningof the next l0-secondinterval,and repeatthis process,without stoppingthe
clock, until the rneasurement
hasbeencompleted.Practicethis techniqueuntil you can
reliably get closeto 600countsfor eachl0-secondintervalof countingin the testmode.
Tum on the voltagefor the Geiger-Muellertube,beingvery carefulto setit at
the correctvoltagelistedon your apparatus.Ask your instructorto check. You arenow
readyto begin the backgroundmeasruemen!as describedbelow. During the background
countingthere shouldbe no radioactivesourcesin the vicinity of your equipment.The
major sourceof the backgroundcountsis cosmicradiation and decayof radioactive
isotopesfound in the constnrctionmaterialof the building.
The silver foils will be activatedby bombardingwith neufronsfor about five
minutes.This will be doneby the instructor. Be preparedto starttaking dataas soonas
an activatedfoil is givento you. Remember,
in one minute the silver-Il0 isotopewill
have been decayingfor almost three half-lives, leaving about an eighth of the active
material with which you started.The instuctor will review proceduresfor handling
activatedfoils andtaking data.
1. Measurebaskgroundradiation.A sampledatasheetwhich will helpyou in the
experimentis included. Turn on the clock and simultaneouslystart the counter.Do not
turn off the clock for the entire period during which you are making a measurernent.
After 10 secondsstopthe counter. In the next 10 secondinterval,recordthe numberof
countsandresetthe counter.Repeattheprocedureuntil you haveobtained10 datapoints
for the background.Stopthe clock and rc-zeroi! resetyour counterandbe preparedto
beginmeasuringthe astivatedsampleassoonasit is givento you.
-56-
Experiirren':
Experiment# l3
2. Measure the activity from the activated silver foil using the same procedure as
in (l). Record the datafor at least 8 minutes.
3. If your data do not look sensibleor if you miss too rnany counting intervals then
you shouldrepeatthe measurementin (2) using a freshly activatedsilver foil.
4. After subtractingthe averagebackgroundcount for l0 secondsfrom each of the
datapointsof (2), plot your dataon semi-loggaph paperas illustrated in Graph B.
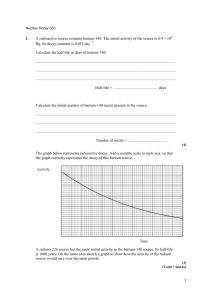
Data Analysis and Interpretation of Data:
There are two silver isotopes which have been activated, each having a different
half-life. Referring to Graph B, determine the longer half-life by drawing the best
straight line through the data points in the time interval of data taken from about three to
eight minutes (In the report discusswhy). This is illustratedas line (l) on the graph. On
line (l), choosevaluesof N(t,) and N(t ): N(t,)/2. For example,referringto the sa:nple
databelow, we might choosepoints "a" and "b" with N(t,) :320 and N(t") : 160. The
time interval (tr- t,) is the half-life of the longer-lived isotope(Ag-108); in this exampleit
ts (220 - 78) sec : 142 sec: 2.37 minutes.Repeat this determination for 2 other pairs
of points on the curve and take the average of the 3 determinations. Estirnatethe
experimentaluncertainty in your measuredvalue for the half-life.
Nexf subtractthe value of line (l) from eachof your datapoints. Plot the results
of the subtraction on the graph and draw the best straight line through these points, i.e.,
'
line (2). Determine the half-life of the second isotope from line (2) i" the samemanner as
you did for line (1). The secondisotopeis the shorter-livedone (Ag-110).
Compare your experimental results with the accepted values given for the two
isotopesof silver. Calculatethe percentdeviations.
The example below illustrates the procedure for data analysis. The graphs (A and B)
are for the data given in the following table. Backgroundcountshave been subtracted.
Time (sec) N - Bksd.
0
20
40
60
80
100
120
140
160
Stralght Line
476
392
323
282
347
315
286
214
194
180
200
177
176
220
161
146
260
280
300
320
340
360
380
400
129
77
37
22
12
5
1
259
235
248
219
196
240
Time (sec) N - Bkgd.
Difference
it to Tlonc CIshort)
1200
464
736
841
421
419
614
382
231
424
119
112
98
87
82
71
70
440
62
53
0
460
160
1
480
145
0
500
-57-
StraightLine
Difference
Fitto Tlonc Clshort)
132
2
134
120
109
99
89
E1
-1
3
-1
-3
0
74
-3
67
61
3
1
55
-3
51
50
0
47
42
45
1
41
0
)
Experiment
# i:
Experiment# L3
1240
1000
$
z
(l)
.E
800
I
600
()
(l)
.n
o
F
t-
o
o-
400
Q
J
o
200
()
0
500
100
time(sec)
Elapsed
Decayof Silver 108and 110
GraphA. LinearPlot of Radioactive
1E4
(U
e
o
.g
o
o
oI
1E3
o
\
o
CL
o
c
:'
o
()
\
1E2
\
\
\
1E1
0
100
300
200
Elapsedtime(sec)
500
GraphB. Semi-togarithmic
Plot of Radioactive
Decayof Silver 108and SilverI 10
-58-
Experirr,ei:t; t -:;
Experiment# l3
RadioactiveDecav of Silver
Count
lnterval
Sec,0-l 0
20-30
Elapsed
Time
Sec.
Backgnd
Counts
Trial 1
Counts
Tljal2
Counts
0
1 0 0 - l10
120-130
140-150
160-t7A
180-190
200-210
220-230
240-2sA
260-270
280-290
300-310
320-330
340-3s0
20
40
60
80
100
120
140
160
180
204
220
240
260
280
300
320
344
360-370
360
380-390
400-410
420-430
44A-4s0
460-474
480-490
490-510
380
400
420
440
460
480
500
40-s0
60-70
80-90
Experiment# 13
DataSheet
on
is optional,at the discretionof your instructor.It is described
Part II of this experiment
the following page.
-59-
Expenment# l3
Part II.
lntroduction:
When any kind of radiation(o, F, y) passesthroughmatter someof the
radiation is absorbedor scattered.
We usethe term"affenuation"to refer to the reduction
in intensityof the transmiuedradiation.The attenuationdependson severalfactors:
i) the type of radiation
ii) the kind of materialthroughwhich the radiationpasses
iii) the thicknessof the absorbingmaterial
ln this part of the experimentyou will be studyingthe absorbingproperties
of severaldifferent materialsfor two t1pes of radiation, P (energeticelectrons)and y
(energeticphotons correspondingto high-frequencyelectromagneticradiation). The
radiationis producedby standardradioactivesourceswhich are labeledaccordingto the
tlpe of radiationproduced.They will be distributedby the instnrctorwhenyou areready
to use them. Note that the sourcesire very weakand are entirely safeto handle(but be
sureto returnthemwhenyou arefinished!)
Procedure: Absorptionof p-rays(yellowdisc)andy-rays(blackdisc).
lnsert a disc two slots below the GM tube.
Measurethe number of countsin one minute..
Insert one of the shields , i.e., paper in the slot next to the GM tube and again
measurethe number of counts for one minute.
Repeatwith the Al and separatelythe lead shields.
Using the other disc, repeatall of the above.
Record your data (number of countsin one minute) below.
BetaAbsorptionData:
No Shield
; Paper-;
Al-;
Lead
Gamrna Absorption Data: No Shield_
; Paper-;
Al-;
Lead
In your repor! discuss the effectivenessof each shield as protection from each of the
radiations.
-60-