Exercise 1C: Water Potential
advertisement

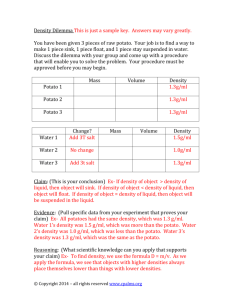
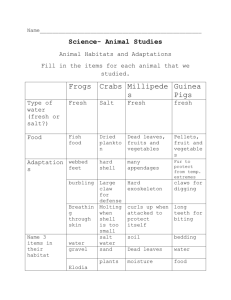
CLASS COPY DO NOT WRITE ON THIS COPY COPY #_______ PROBLEM: What will happen to cells when they are placed in a solution with a high concentration of salt? Introduction: Two shipwrecked sailors were stranded on a small raft with no fresh water to drink. The sailors knew they could last without food for up to a month, but if they didn't have water to drink they would be dead within a week. Hoping to postpone the inevitable, their thirst drove them to drink the salty seawater. They both died within two days. Why do you think drinking seawater killed the sailors faster than not drinking any water at all? Today we explore the cause of the sailors’ death. We'll prepare solutions of salt water to represent the sea, and we'll cut up slices of potato to represent the sailors. Potatoes and sailors are both made of cells, slightly different but similar! Objective: To determine the cause of the sailors’ death, we will determine the effects of salt water on slices of potato. Our assumption is that potato cells will behave like the cells in the sailors’ body. We will compare salt water to fresh water so we can see if there is an effect caused by the salt in the water. The fresh water acts as a control in this experiment. Hypothesis: Predict what will happen to a piece of fresh potato (cells) when it is placed into a solution with a high concentration of salt. Materials: potato cork bore saran wrap /foil 10% salt solution (stock) 4 - 5 oz. cups knife Electronic Balance rubber band Procedure: 1. A bottle of 10% salt solution (sodium chloride) is provided. You will need about 175ml of this salt solution for this lab. In a beaker from your cabinet, place 175 ml. of the stock 10% salt solution. Next, obtain and label four cups as follows: 10% salt solution 5% salt solution 1% salt solution distilled water In the 10% salt solution cup, place 100 ml of the salt solution and set this to the side for now. In the distilled water cup, place 100 ml. of distilled water and set to the side for now. 2. You must now make two of the sea water solutions by diluting the concentrated 10% salt solution to make 5% and 1% solutions. You need complete the calculations shown on page 2 in order to make 100 ml each of a 5% and 1% solution. Record the amounts you will need to make the solutions in Table 1 on page 2. BIOLOGY Page 1 CELL TRANSPORT A. Calculations for 5% salt water solution: Copy these in your notebook! Concentration of stock solution Desired final concentration 100 ml total volume needed amount of dilution = = 10 5 100 ? = ________ = amount of dilution (carry this to next row) = _____ml = amount of stock solution 100 ml total - amount of stock solution = ______ amount of distilled water to be added B. Calculations for 1% salt water solution: Copy these in your notebook! Concentration of stock solution Desired final concentration 100 ml total volume needed amount of dilution = = 10 1 100 ? = ________ = amount of dilution (carry this to next row) = _____ml = amount of stock solution 100 ml total - amount of stock solution = ______ amount of distilled water to be added TABLE 1: Concentration Calculations (Copy in your notebook) Percent Sea Water Vol. of 10% Salt Water (ml) Vol. of Fresh Water (ml) Total Vol. (ml) 10% Sea Water 100.0 5% Sea Water 100.0 1% Sea Water 100.0 Fresh Water 0.0 100.0 100.0 3. Using your calculations in the table, make 100 ml of each of the 5% and the 1% solutions and place each in the correctly labeled cup. Set these aside for later use. FILL IN THE TABLE ABOVE. 4. In order to determine if the salt water had any effect on the cells in the sailors’ body, we need to cut four equal sized slices of potato to represent the sailors. The best way to do this is to core a piece of potato. 5. Stand the potato on end and bore four vertical holes. Space them close together so that one or two other groups can use the rest of the potato. Weigh the cylinders as you remove from the cork borer and, if necessary, cut them so that they are all exactly the same weight (in grams). Keep weighing them until they are the same. WHY must we do this? _______________________________________ BIOLOGY Page 2 CELL TRANSPORT 6. The goal is to end up with four potato slices of exactly the same weight, so that we can compare the effects of the water and the salt. In order to draw any conclusions, the potato cylinders must be exactly the same weight when you begin. It is important to enter the INITIAL weight of EACH potato cylinder into the data table so you can compare it to the size of the potato cylinders tomorrow. Table 2 is located on the lower half of this page. 7. You will need a 5th piece of potato. Under the term “TURGIDITY” in Table 2, record your observations about the “crispness” of your potatoes. Crisp means that the potato if bent can snap or crack or is closed to being able to do this. Flaccid means that the potato, when held upright, will lean over to the side like a wilting plant. IB means you feel that your potatoes are “in between”. Analyze your potato and decide in which category it falls. Record this in Table 2. The answer should be the same for all four solution set-ups as the potatoes all start out the same before being placed in the liquids. 8. Lastly, place one cylinder of potato into each of the four cups. Cover the cups with saran wrap using a rubber band to hold the wrap securely in place. Why is it important to cover the cups overnight? ______________________________________________________________________ DAY TWO 1. Remove the potato cylinders one at a time, quickly weigh each one separately, and record this data in your table. 2. Measure the weight of the potato cylinders being careful to make a note of which cylinder belongs to which cup! Enter the change in weight into the data table. IMPORTANT: Use a plus (+) to indicate a cylinder has grown and a minus (–) to indicate a slice has shrunk overnight. (Example + 0.5 g means an increase in weight and overall size / –3.0 g means a decrease in weight and therefore overall size). TABLE 2: Potato Lab Data (Copy in your notebook) SAMPLE Initial Weight (g) (Day 1) Final Weight (g) (Day 2) TOTAL Change in Weight Turgidity Tonicity (Crisp/ IB/ Flaccid) Day 1 Day 2 Hypo- Iso- Hyper- 10 % Salt 5% Salt 1% Salt Distilled Water BIOLOGY Page 3 CELL TRANSPORT 3. You will be provided with graph paper to make a graph of your data from Table 2. Be sure you include the variables, units, and a title in your graph. (Hint: What are the variables we are testing in this laboratory? What data are we collecting?) Observations and Conclusions: 1. Write a formal conclusion for this lab using the format in your notebook. In the discussion, you MUST address the following questions: a. What happened overnight to the potato cylinders? Why did it happen? Include scientific terms in your explanation. b. Is diffusion or osmosis responsible for changes in weight of the potato cylinders? Define both these terms in your explanation. c. Which of the three solutions is isotonic to potato cells? How do you know this? d. Define the terms hypertonic, isotonic, and hypotonic in your explanation. e. What happened to the sailors’ cells after they drank the salt water? f. Why did the sailors die more quickly after drinking salt water than not drinking anything at all? What do you think killed the sailors? BIOLOGY Page 4 CELL TRANSPORT