Supplementary Data
advertisement

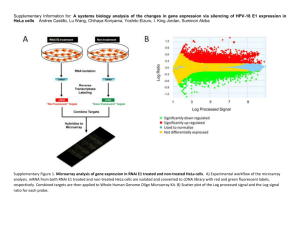
Supplemental Method Generation of PGC1mice and animal care PGC1mice were generated as described previously1. Both WT and PGC1mice were kept in a temperature-controlled facility (20-22C) with 12-h light/dark cycles. Food and water were available ad libitum unless noted. All animal protocols and procedures used in this study were approved by the UK Home Office and the University of Cambridge (Schedule 1, UK Animal Procedures Act 1986). Histological sample preparation and analysis Cardiac samples from 12-week-old female mice were prepared for morphological analysis according to published protocols1. Interstitial fibrosis was demonstrated by picrosirius staining as described previously2. Left ventricular samples were frozen in optimum cutting temperature (OCT) and lipid accumulation was determined with Oil Red O staining3. Similar to the mild phenotype of our previously published PGC1-/-1, there were no differences between PGC1-/- and WT mice with respect to heart weight to body weight ratios, and heart structure, hypertrophy, morphology, interstitial fibrosis and lipid accumulation (Supplementary Fig. 1A, B, C respectively). RNA preparation and real-time quantitative RT-PCR. The cardiac samples from 12-week-old male mice were extracted for RNA and purified using the RNA clean-up protocol from the RNeasy Mini Kit (Qiagen Ltd, Crawley, UK). RNA was quantified spectroscopically at 260 nm using a GeneQuant Nucleotide calculator (Amersham Biosciences, Little Chalfont, UK) and checked for integrity on a 1% TBE gel using ethidium bromide staining. Realtime quantitative PCR was performed using a TaqMan 7900 (Abi) according to standard protocols. Oligonucleotide primers and TaqMan probes were designed using Primer Express, version 2.0 (Applied Biosytems). Primer and probe 1 sequences, together with gene abbreviations, can be found in Supplementary Table 1. RNA amplification and hybridisation The study was carried out with Affymetrix GeneChip® Mouse Genome 430 2.0 array (Affymetrix, Santa Clara, CA, USA). Extracted RNA was assessed for quality using spectrophotometry and Agilent Bioanalyser 2100. For each sample, 4µg of total RNA was amplified and labelled using the Affymetrix One-Cycle Target Labelling kit (Affymetrix, Santa Clara, CA). The amplified targets were hybridised to the array according to manufacturer’s protocol (Affymetrix, USA) and scanned using a GeneChip Scanner 3000 7G. CEL files were extracted from the image files automatically by Affymetrix GeneChip Operating Software (GCOS). Microarray data analysis Microarray gene expression analysis was performed using GeneSpring GX 11.0.2 software (Agilent Technologies Inc., USA). Robust Multichip Average (RMA) with Quantile normalisation ware used for background correction and normalisation to pre-process the CEL files 4. An unpaired t-test was performed with multiple comparisons (Benjamini-Hotchberg correction). Genes differentially expressed were identified by using a fold-change cut-off of 1.5. A list of differentially expressed genes was created solely based on the fold change as it was observed that the gene expression changes between the two groups was very moderate after applying a t-test with multiple hypothesis testing to the data. Initial analysis was done using Ingenuity Pathway Analysis (IPA) to define the functional networks of differentially expressed genes. Genes that changed in expression level by 1.5 fold (Supplementary Table 2) in PGC1β-/- compared to WT were used to determine significant gene networks and canonical pathways in IPA version 8.6 (Ingenuity® Systems, www.ingenuity.com). A dataset containing gene identifiers (HUGO gene symbol) and corresponding fold change was uploaded and each gene identifier was mapped to its corresponding gene object 2 to create the list of focus molecules. The core analysis was performed using both direct and indirect relationships with 35 molecules per network option. IPA computes a score for each network; higher scores indicate the increased probabilities of the focus genes in a network being found together other than because of random chance. Supplementary Table 3 lists the canonical pathways showing the ratios and the p-values. Ratios were calculated by comparing the number of focus genes found in that pathway to the total number of molecules present and it reflects the amount of association and the negative logarithm of the p-value gives the confidence of association. Among several networks identified, cell death and proliferation, cell signalling and lipid metabolism were the most significantly altered (Supplementary Table 4).Since the differential gene expression analysis showed individual genes to be weakly and non-significantly differentiated, functional Analysis based on gene expression data was done using Gene Set Enrichment Analysis (GSEA, Broad Institute, Cambridge, MA). The expression data set containing Affymetrix Probe Set ID, sample name and an expression value for each feature in each sample was uploaded onto GSEA web application. The gene set file c2.v2.symbols.gmt, containing the C2 curated genes sets of the Molecular Signatures Database (MSigDB) was used to perform the gene ranking. Genes represented by more than one probe were collapsed using the gene symbols before analysis. The expression dataset had 45101 features and after collapsing the features into gene symbols, there were 21891 genes. The analysis was restricted to a minimum number of 5 genes and a maximum number of 500. The weighted enrichment statistic was calculated using the Signal2Noise metric for ranking genes. GSEA is a computational method that determines whether a defined set of genes shows statistically significant concordant differences between two biological states5. The enrichment score (ES), was calculated showing the degree to which a gene set is over represented. A nominal p-value≤ 0.01 and qvalue with false discovery rate (FDR) ≤0.25 was used as parameter to determine the differentially regulated gene sets. 3 Lipid profiling Approximately 20 mg of cardiac tissue was weighed and 20 µl of an internal standard mixture6, and 50 µl of NaCl (0.9 %) were added to the sample. Lipids were extracted from the samples with 200 µl of chloroform: Methanol (2:1) solvent and the tissue was homogenised with a glass rod. After vortexing for 2 min and incubating for 1 h at room temperature, the lower layer (approximately 100 µl) was separated by centrifugation at 10,000 rpm for 3 min at room temperature. Twenty microliters of labelled standard mixture (three stable isotope-labelled reference compounds) was added to the lipid extract and 1.0 μl injection was used for LC/MS analysis. Sample order for analysis was established by randomization. Lipid extracts were analyzed on a Q-ToF Premier mass spectrometer (Waters) combined with an Acquity Ultra Performance Liquid chromatography (UPLC/MS). Mass spectrometry was carried out on Q-Tof Premier (Waters, Inc.) run in ESI+ mode. The data was collected over the mass range of m/z 300-1200 with a scan duration of 0.2 sec. The source temperature was set at 120 °C and nitrogen was used as desolvation gas (800 L/h) at 250 °C. The voltages of the sampling cone and capillary were 39 V and 3.2 kV, respectively. Reserpine (50 µg/L) was used as the lock spray reference compound (5 µl/min; 10 sec scan frequency). The obtained data was converted into netCDF file format using Dbridge software from MassLynx (Waters, Inc.). The converted data was processed using MZmine software version 0.60 7. Lipids were identified based on their retention time (RT) and mass to charge ratio (MZ) using our in-house built lipid database as previously described 6. All the identified lipids were quantified by normalizing with corresponding internal standards. In vivo electrocardiography (ECG) and pharmacological studies Mice were anesthetised by intraperitoneal injection of ketamine and xylazine (150 and 5 mg/kg respectively). After 20 mins of anaesthesia, mice were placed on a bed heated by a water bath at 37C and breathing and movement of mice was monitored continuously. ECGs were measured from two 29-gauge needle electrodes inserted in the left and right foreleg and signals were pre-amplified 4 using an NL100AK headstage amplified with an NL104A amplifier and then bandpass filtered between 50 Hz and 500 Hz using an NL125/6 filter (Neurolog, Hertfordshire, UK). Filtered signals were digitized by a model 1401+ interface and processed and stored using Spike 2 software (Cambridge Electronic Design, Cambridge, UK). ECG records were compared both before and 10 min following intraperitoneal isoproterenol (2 mg/kg) injection. . Arrhythmogenic assessment in Langendorff- perfused hearts Monophasic action potential (MAP) recording was performed as described previously8. In brief, an epicardial MAP electrode (Harvard Apparatus, Edenbridge, UK) was placed against the basal left ventricular epicardium and electrical signals were amplified, band-pass filtered (0.5 Hz to 1 kHz: Gould 2400S, Gould-Nicolet Technologies, UK), digitized at a sampling frequency of 5 kHz (1401, Cambridge Electronic Design, CED, Cambridge, UK) and recorded with Spike 2 software (CED, Cambridge, UK). During external pacing, hearts were excited with 2 ms square pulses of twice the threshold voltage (Grass S48 stimulator, Grass-Telefactor, Slough, UK) using bipolar platinum electrodes placed on the right ventricular epicardium. Arrhythmogenicity was assessed with using a programmed electrical stimulation (PES) procedure. This consisted of eight regularly paced stimuli (S1, 8 Hz) followed by an extrasystolic stimulus (S2). With each successive cycle, the interval between the 8 th S1 and S2 stimuli was reduced by 1 ms until the S2 stimulus either failed to generate an AP or induced ventricular tachycardia (VT). Ventricular myocyte isolation and Ca2+ imaging Isolation of ventricular myocytes and loading of the Ca 2+ indicator Fluo-4 (Invitrogen, Paisley, Scotland) were performed as described previously 8. 50 l of Fluo-4 loaded cells were added to a custom-made recording chamber containing external Hepes-buffered medium (in mM: 145 NaCl, 5 KCl, 1 CaCl2, 1 MgCl2, 10 glucose,10 Hepes, pH adjusted to 7.4 with NaOH). The loaded Fluo-4 was excited at a 488 nm wavelength and emission collected at a 510 nm wavelength. 5 To achieve higher temporal resolution, fluorescence signals were acquired in line-scan mode using a Leica SP5 confocal microscope (Leica, Jena, Germany). The scan line was aligned along the long axis of the ventricular myocytes examined and signal was processed with SP5 software. The amplitudes of the Ca2+ transients were expressed as their peak value (F) normalised to baseline fluorescence (F0). To minimise the photo-damage of cells due to overexposure to laser, Ca2+ signals were acquired for only 10 seconds every minute before and after drug application. All the experiments were conducted at room temperature (25oC). Cells were continuously field-stimulated using constant voltage pulses (30 V, 2 ms duration) at a frequency of 1 Hz to generate action potentials. Isoproterenol (10 nM) was applied through a gravity-driven perfusion system (3 ml/min) for at least 5 mins. Whole cell patch-clamp experiments The external saline contained (mM) 145 NaCl, 5 KCl, 1 CaCl 2, 1 MgCl2, 10 glucose, 10 Hepes, pH adjusted to 7.4 with NaOH. 50 l of myocytes were resuspended in external saline in a recording chamber (PDMI-2, Harvard Apparatus, Edenbridge, Kent) and visualized with an inverted microscope (Nikon Eclipse, TE2000-U, Nikon UK, Kingston upon Thames, Surrey, UK). Experiments were carried out at 37oC. Two different types of pipette solutions were used. For Ca2+ current measurements, the pipette solution contained (mM) 57.26 CsCl, 52.74 Cs-Aspartate, 5 Na2-ATP, 5.37 MgCl2, 10 Hepes, 5 Cs4-BAPTA, pH adjusted to 7.2 with CsOH. For the action potential measurements the pipette solution contained (mM) 115 K-Aspartate, 5 KCl, 10 NaCl, 10 Hepes, 5 Mg-ATP, 5 BAPTA with pH adjusted to 7.2 with KOH. Action potentials were induced in current-clamped ventricular myocytes by applying 2 ms long depolarizing, 500 pA to 1000 pA, currents. 2-4 M pipettes were pulled using a vertical puller (PC-10, Narishige, Japan) from filamented borosilicate glass (GC150F-10, Harvard Apparatus, Edenbridge, UK). Electrophysiological recordings were made in voltage-clamp mode of a Multiclamp 700B (Molecular Devices, CA, USA). Data was digitized at 5 KHz using a Digidata 1440A interface and acquired at 10 kHz 6 using Clampex 10.2 software (Molecular Devices, CA, USA). Recordings were made 5 min after achieving the whole cell configuration. Activation of voltage-gated L-type Ca2+ current was assessed using 5 mV or 10 mV incremental (200 ms long) depolarization pulses from -40 mV to 50 mV. Double pulse protocol consisting of series of 5 mV incremental preceding prepulses (200 ms) from -40 mV to 50 mV followed by constant test pulse from 40 to 0 mV (200 ms) was used to assess steady-state inactivation. Currentvoltage relationships were obtained by plotting peak current elicited at different test pulses against respective voltage steps. To obtain conductance-voltage relationships, currents were normalized to maximum currents and plotted against voltage of test pulse (activation) and pre-pulses (inactivation). To measure voltage-dependent outward K+ currents, 500 ms long depolarization pulses were applied from a holding potential of -40 mV in successive 10 mV increments up to 80 mV. Transient K + current was measured as the peak K+ current at the beginning of pulse and sustained K+ current was measured towards the end of depolarization pulses (at 490 ms). Holding at -40 mV inactivated voltage-gated Na+ current and 10 M nifedipine or 5 mM Co2+ was added to block voltage-gated L-type Ca2+ current. Inward rectifying K+ currents were elicited by applying hyperpolarizing pulses of 500 ms from a holding potential of -40 mV to -120 mV in 10 mV increments. Unless otherwise stated, statistical data from the physiological studies used paired t-tests for continuous and Fisher’s exact tests for categorical data concerned with incidences of arrhythmia. 7 Supplementary Figures and Legends Supplementary Fig 1. (A) Heart weight (HW) to body weight (BW) ratio of WT and PGC1-/- mice (PGC1-/- HW 193±22 mg, WT HW 215±15 mg, p>0.05, n=5; BW PGC1-/- 33±2 g versus WT BW 38±2, p>0.05, n=9). (B) picrosirius staining for interstitial fibrosis in WT and PGC1-/- ventricles. (C) Lipid accumulation stained by Oil Red O staining in left ventricles. Legends to supplementary Tables. Supplementary Table 1. Primers used for qRT-PCR Supplementary Table 2. List of differentially expressed genes with 1.5 fold increases or decreases in expression in PGC1-/- compared with WT. Supplementary Table 3. Selected genetic networks with score(>=15) in PGC1/- compared to wt (ingenuity pathway analysis) Supplementary Table 4. Selected canonical pathways with ratio and p-value (ingenuity pathway analysis) Supplementary Table 5. Gene set enrichment analysis(GSEA) of the gene expression data (PGC1-/- vs WT) 8 Supplemental Reference List 1. Lelliott,CJ, Medina-Gomez, G, Petrovic, N, Kis, A, Feldmann, HM, Bjursell, M, Parker, N, Curtis, K, Campbell, M, Hu, P, Zhang, D, Litwin, SE, Zaha, VG, Fountain, KT, Boudina, S, Jimenez-Linan, M, Blount, M, Lopez, M, Meirhaeghe, A, Bohlooly, Y, Storlien, L, Stromstedt, M, Snaith, M, Oresic, M, Abel, ED, Cannon, B, and Vidal-Puig, A. Ablation of PGC-1beta results in defective mitochondrial activity, thermogenesis, hepatic function, and cardiac performance. PLoS. Biol. 4:e369. 2. Kis,A, Murdoch, C, Zhang, M, Siva, A, Rodriguez-Cuenca, S, Carobbio, S, Lukasik, A, Blount, M, O'Rahilly, S, Gray, SL, Shah, AM, and Vidal-Puig, A. Defective peroxisomal proliferators activated receptor gamma activity due to dominant-negative mutation synergizes with hypertension to accelerate cardiac fibrosis in mice. Eur J Heart Fail 11:533-541. 3. Gray,SL, Nora, ED, Grosse, J, Manieri, M, Stoeger, T, Medina-Gomez, G, Burling, K, Wattler, S, Russ, A, Yeo, GSH, Chatterjee, VK, OÇÖRahilly, S, Voshol, PJ, Cinti, S, and Vidal-Puig, A. Leptin Deficiency Unmasks the Deleterious Effects of Impaired Peroxisome ProliferatorÇôActivated Receptor +¦ Function (P465L PPAR+¦) in Mice. Diabetes 55:2669-2677. 4. Irizarry,RA, Bolstad, BM, Collin, F, Cope, LM, Hobbs, B, and Speed, TP. Summaries of Affymetrix GeneChip probe level data. Nucleic Acids Res 31:e15. 5. Subramanian,A, Tamayo, P, Mootha, VK, Mukherjee, S, Ebert, BL, Gillette, MA, Paulovich, A, Pomeroy, SL, Golub, TR, Lander, ES, and Mesirov, JP. Gene set enrichment analysis: a knowledge-based approach for interpreting genome-wide expression profiles. Proc Natl. Acad. Sci U. S. A 102:1554515550. 6. Laaksonen,R, Katajamaa, M, Paiva, H, Sysi-Aho, M, Saarinen, L, Junni, P, Lutjohann, D, Smet, J, Van Coster, R, Seppanen-Laakso, T, Lehtimaki, T, Soini, J, and Oresic, M. A systems biology strategy reveals biological pathways and plasma biomarker candidates for potentially toxic statininduced changes in muscle. PLoS ONE 1:e97. 7. Katajamaa,M, Miettinen, J, and Oresic, M. MZmine: toolbox for processing and visualization of mass spectrometry based molecular profile data. Bioinformatics. 22:634-636. 8. Gurung,IS, Kalin, A, Grace, AA, and Huang, CL. Activation of purinergic receptors by ATP induces ventricular tachycardia by membrane depolarization and modifications of Ca2+ homeostasis. J Mol. Cell Cardiol 47:622-633. 9