5 unbiased medical doctors were involved in the
advertisement
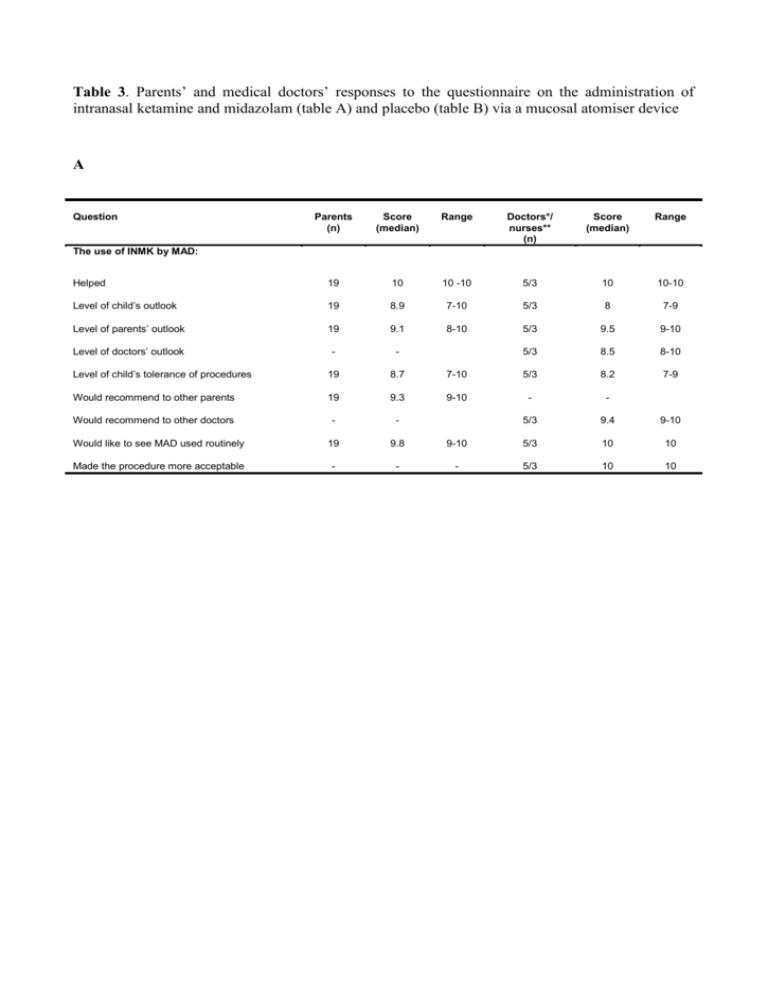
Table 3. Parents’ and medical doctors’ responses to the questionnaire on the administration of intranasal ketamine and midazolam (table A) and placebo (table B) via a mucosal atomiser device A Question Parents (n) Score (median) Range Doctors*/ nurses** (n) Score (median) Range Helped 19 10 10 -10 5/3 10 10-10 Level of child’s outlook 19 8.9 7-10 5/3 8 7-9 Level of parents’ outlook 19 9.1 8-10 5/3 9.5 9-10 Level of doctors’ outlook - - 5/3 8.5 8-10 Level of child’s tolerance of procedures 19 8.7 7-10 5/3 8.2 7-9 Would recommend to other parents 19 9.3 9-10 - - Would recommend to other doctors - - 5/3 9.4 9-10 Would like to see MAD used routinely 19 9.8 9-10 5/3 10 10 Made the procedure more acceptable - - - 5/3 10 10 The use of INMK by MAD: B Question Parents (n) Score (median) Range Doctors*/ nurses** (n) Score (median) Range Helped 19 5 3-7 5/3 3 2-4 Level of child’s outlook 19 5.8 5-7 5/3 3 2-4 Level of parents’ outlook 19 4.9 3-7 5/3 5 3-7 Level of doctors’ outlook - - 5/3 4 3-5 Level of child’s tolerance of procedures 19 8.5 7-10 5/3 8 7-9 Would recommend to other parents 19 4 3-6 - - Would recommend to other doctors - - 5/3 3 1-5 Would like to see MAD used routinely 19 4 3-6 5/3 3 1-5 Made the procedure more acceptable - - - 5/3 3 1-5 The use of intranasal sedation (placebo) by MAD: *5 unbiased medical doctors were involved in the painful or diagnostic procedures carried out in the study. *3 unbiased nurses were involved in the painful or diagnostic procedures carried out in the study. INKM, intranasal midazolam and ketamine MAD, mucosal atomiser device.






