AP Chem Agenda and Assignments – Week Starting Sept 23, 2013
advertisement
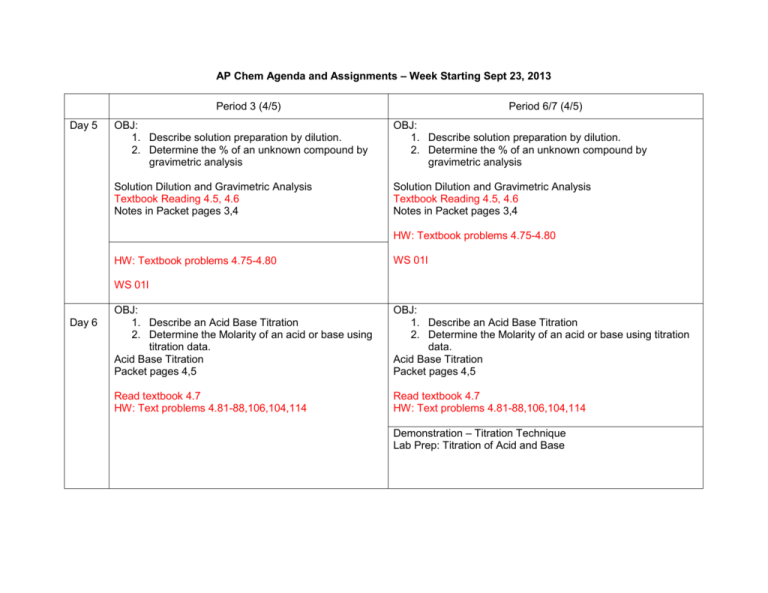
AP Chem Agenda and Assignments – Week Starting Sept 23, 2013 Period 3 (4/5) Day 5 Period 6/7 (4/5) OBJ: 1. Describe solution preparation by dilution. 2. Determine the % of an unknown compound by gravimetric analysis OBJ: 1. Describe solution preparation by dilution. 2. Determine the % of an unknown compound by gravimetric analysis Solution Dilution and Gravimetric Analysis Textbook Reading 4.5, 4.6 Notes in Packet pages 3,4 Solution Dilution and Gravimetric Analysis Textbook Reading 4.5, 4.6 Notes in Packet pages 3,4 HW: Textbook problems 4.75-4.80 HW: Textbook problems 4.75-4.80 WS 01l WS 01l Day 6 OBJ: 1. Describe an Acid Base Titration 2. Determine the Molarity of an acid or base using titration data. Acid Base Titration Packet pages 4,5 OBJ: 1. Describe an Acid Base Titration 2. Determine the Molarity of an acid or base using titration data. Acid Base Titration Packet pages 4,5 Read textbook 4.7 HW: Text problems 4.81-88,106,104,114 Read textbook 4.7 HW: Text problems 4.81-88,106,104,114 Demonstration – Titration Technique Lab Prep: Titration of Acid and Base Day 1 OBJ: 1. Describe general properties of aqueous solutions. 2. Write Ionic equations for precipitation reactions Note packet pages Packet pages 1-3 Textbook Reading Section 4.1-4.2 Chapter 4 Problems: 17, 20, 22, 24 OBJ: 1. Describe general properties of aqueous solutions. 2. Write Ionic equations for precipitation reactions Note packet pages Packet pages 1-3 Textbook Reading Section 4.1-4.2 Chapter 4 Problems: 17, 20, 22, 24 Demonstration – Titration Technique Day 2 Day 5 OBJ: 1. Describe properties of Acid and Base 2. Describe Aqueous Reactions of Acid and Base OBJ: 1. Make a quantitative solution using correct analytical technique. 2. Determine the % acid in an unknown acid salt. 4.3 Acid Base Reactions Packet pages 3-6 Lab: Quantitative Analysis of an Acid Salt Read section 4.3 Chapter 4 Problems: 26, 27, 29, 30, 31, 33 Lab Continued OBJ: 1. Make a quantitative solution using correct analytical technique. 2. Determine the % acid in an unknown acid salt. 3. Lab: Quantitative Analysis of an Acid Salt OBJ: 1. Describe properties of Acid and Base 2. Describe Aqueous Reactions of Acid and Base 4.3 Acid Base Reactions Packet pages 3-6 Read section 4.3 Chapter 4 Problems: 26, 27, 29, 30, 31, 33 Lab: Continued Day 6