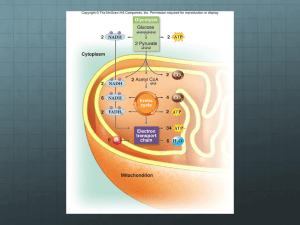
Insights into pH-induced Metabolic Switch by Flux Balance Analysis
advertisement

Insights into pH-induced Metabolic Switch by Flux Balance Analysis Supplementary Material Marija Ivarsson, Heeju Noh, Massimo Morbidelli and Miroslav Soos* Institute for Chemical and Bioengineering, Department of Chemistry and Applied Biosciences, ETH Zurich, Zurich, Switzerland * Corresponding author. E-mail: miroslav.soos@chem.ethz.ch; HCI F137, Vladimir-Prelog-Weg 1, CH-8093 Zurich; Tel.: +41 44 6334659, Fax.: +41 44 6321082 1 Supplementary Table 1. List of target genes investigated by gene expression analysis Gene Symbol 1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 20 21 22 23 24 25 26 27 28 29 30 31 32 Hk1 Hk2 Pfkl Pgam2 Pkm2 Pepck1 Pepck2 Ldha Pcx Pdha1 Pdha2 Dlat Pdk1 Pdk2 Pdk3 Cs Idh3a Ogdh Sdha Fh1 Mdh1 Mdh2 Acly Me1 Me2 Me3 Gpt2 Got1 Got2 Glud1 Gls G6pdh Name hexokinase 1 hexokinase 2 phosphofructokinase, liver phosphoglycerate mutase 2 pyruvate kinase muscle phosphoenolpyruvate carboxykinase 1, cytosolic phosphoenolpyruvate carboxykinase 2, mitochondrial lactate dehydrogenase A pyruvate carboxylase pyruvate dehydrogenase E1 alpha 1 pyruvate dehydrogenase E1 alpha 2 dihydrolipoamide S-acetyltransferase pyruvate dehydrogenase kinase, isoenzyme 1 pyruvate dehydrogenase kinase, isoenzyme 2 pyruvate dehydrogenase kinase, isoenzyme 3 citrate synthase isocitrate dehydrogenase 3 alpha oxoglutarate dehydrogenase succinate dehydrogenase complex, subunit A fumarate hydratase 1 malate dehydrogenase 1, NAD malate dehydrogenase 2, NAD, mitochondrial ATP citrate lyase malic enzyme 1, cytosolic malic enzyme 2, mitochondrial NADP dependent malic enzyme 3, mitochondrial NAD dependent glutamic pyruvic transaminase 2 glutamate oxaloacetate transaminase 1 glutamate oxaloacetate transaminase 2 glutamate dehydrogenase 1 (=GDH) glutaminase glucose-6-phosphate dehydrogenase 2 Supplementary Table 2: Reactions in the metabolic network used for flux balance analysis. Glycolysis 1 2 3 4 5 GLC6P <-> F6P F6P + ATP -> ADP + 2 GAP GAP + NAD + ADP <-> [3PG] + NADH + ATP 3PG -> PEP PEP + ADP + H -> PYR + ATP 6 7 8 9 Pentose Phosphate Pathway GLC6P + 2 NADP + H2O -> RBL5P + 2 NADPH + CO2 RBL5P <-> R5P RBL5P <-> X5P 2 X5P + R5P <-> 2 F6P + G3P Pyruvate Metabolism 10 11 12 13 14 PYR + H -> PYR_m + H_m PYR_m + CoA_m + NAD_m -> CO2 + AcCoA_m + NADH_m + H_m PYR_m + HCO3_m + ATP_m -> OAA_m + ADP_m + Pi MAL_m + NAD_m <-> PYR_m + NADH_m + H_m + CO2 MAL + NADP <-> PYR + NADPH + H + CO2 15 16 17 18 19 20 TCA Cycle OAA_m + AcCoA_m + H2O <-> CIT_m + CoA_m CIT_m + NAD_m + H2O <-> AKG_m + NADH_m + CO2 + H_m AKG_m + NAD_m + CoA_m -> SUCCoA_m + NADH_m + H_m + CO2 SUCCoA_m + ADP_m + Pi_m + FAD_m <-> FUM_m + ATP_m + CoA_m + FADH2_m FUM_m + H2O <-> MAL_m MAL_m + NAD_m <-> OAA_m + NADH_m + H_m 21 22 23 24 25 Malate Aspartate Shuttle OAA_m + GLU_m <-> AKG_m + ASP_m AKG + ASP <-> OAA + GLU ASP_m + GLU -> ASP + GLU_m OAA + NADH + H <-> MAL + NAD MAL + AKG_m <-> MAL_m + AKG Glutaminolysis 26 GLN + H2O -> GLU + NH4 27 GLU_m + H2O + NAD_m <-> AKG_m + NADH_m + NH4 28 GLU <-> GLU_m Acetyl CoA Formation 29 CIT + CoA + ATP + H2O -> AcCoA + OAA + ADP + Pi 3 Amino Acid Synthesis 30 31 32 33 34 GLU + PYR <-> AKG + ALA 3PG + NAD + GLU + H2O -> NADH + H + AKG + Pi + SER GLU + ATP + H + NADPH + H + NADH -> ADP + NADP + Pi + H2O + NAD + PRO SER + THF <-> GLY + MTHF + H2O GLN + ASP + ATP + H2O <-> GLU + ASN + AMP + 2 Pi + H Amino Acid Degradation 35 SER <-> PYR + NH4 36 THR + NAD + CoA -> GLY + AcCoA + NADH + H 37 ARG_m + H2O + AKG_m + NAD_m <-> 2 GLU_m + UREA_m + NADH_m + H_m 38 PRO_m + 2 H2O + NAD_m -> GLU_m + NADH_m + H_m 39 HIS_m + 3 H2O -> GLU_m + Formamide_m + NH4 40 LYS_m + NADPH + 2 AKG_m + 4 NAD_m + 2 H2O + 2 CoA_m -> NADP + 2 H_m + 4 NADH_m + 2 GLU_m + 2 CO2 + 2 AcCoA_m 41 PHE + NADPH + H + O2 -> TYR + NADP 42 TYR + AKG + 2 O2 + CoA + H2O <-> GLU + CO2 + 2 H + FUM_m + AcCoA + Acetate 43 VAL_m + AKG_m + CoA_m + 3 NAD_m + FAD_m + 3 H2O + ATP_m -> 2 H_m + GLU_m + SUCCoA_m + CO2 + 3 NADH_m + FADH2_m + ADP_m + Pi_m 44 ILE_m + AKG_m + 2 NAD_m + 2 CoA_m + FAD_m + H2O + HCO3_m + ATP_m -> GLU_m + SUCCoA_m + AcCoA_m + 2 NADH_m + CO2 + FADH2_m + ADP_m + Pi_m 45 LEU_m + AKG_m + NAD_m + 2 CoA_m + ATP_m + HCO3_m + H2O -> GLU_m + NADH_m + CO2 + H_m + Pi_m + ADP_m + Acetate + 2 AcCoA_m 46 CYS + AKG + SO3 + O2 + H2O -> GLU + PYR + S2O3 + 2 H 47 MET + 2 ATP + H2O + SER + CoA + NAD + HCO3 -> NH4 + CYS + CO2 + NADH + SUCCoA_m + ADP + Pi Transport 48 CIT_m + MAL <-> CIT + MAL_m 49 PRO <-> PRO_m 50 ARG <-> ARG_m PEP Regeneration 51 OAA_m + ATP_m -> ADP_m + PEP_m + CO2 52 PEP <-> PEP_m Oxidative Phosphorylation 53 NADH_m + H_m + 0.5 O2 + 2.5 ADP_m + 2.5 Pi_m -> NAD_m + H2O + 2.5 ATP_m 54 FADH2_m + 0.5 O2 + 1.5 ADP_m + 1.5 Pi_m -> FAD_m + H2O + 1.5 ATP_m 4 Nucleotide Synthesis 55 R5P + ATP -> PRPP + AMP 56 PRPP + 2 GLN + GLY + ASP + 4 ATP + CO2 -> IMP + 2 GLU + FUM + 4 ADP + 2 H2O 57 IMP + ASP + 2 ATP + GTP -> dATP + FUM + 2 ADP + GDP 58 IMP + GLN + 3 ATP + NAD + 2 H2O -> dGTP + GLU + 2 ADP + AMP + NADH 59 HCO3 + NH4 + ASP + PRPP + 3 ATP + NAD -> dTTP + 3 ADP + CO2 + NADH 60 dTTP + GLN + ATP -> dCTP + GLU + ADP 5 Uptake or Production of Substrates (Measured Fluxes) 61 GLC6P + ADP + H -> GLC_ex + ATP 62 PYR + NADH + 2 H -> LAC_ex + NAD + H 63 GLN <-> GLN_ex 64 GLU <-> GLU_ex 65 ALA -> ALA_ex 66 SER -> SER_ex 67 GLY -> GLY_ex 68 ASP -> ASP_ex 69 ARG_m -> ARG_ex 70 HIS_m -> HIS_ex 71 CYS -> CYS_ex 72 THR -> THR_ex 73 ILE_m -> ILE_ex 74 VAL_m -> VAL_ex 75 PHE -> PHE_ex 76 TYR -> TYR_ex 77 LYS_m -> LYS_ex 78 LEU_m -> LEU_ex 79 PRO -> PRO_ex 80 MET -> MET_ex 81 ASN -> ASN_ex 0.0148 dATP + 0.0099 dCTP + 0.0099 dGTP + 0.0148 dTTP + 0.07 ATP + 0.033 dATP + 0.0551 dCTP + 0.0624 dGTP + 0.033 dTTP + 0.07 ATP + 0.6 ALA + 0.377 ARG_m + 0.359 ASP + 0.288 ASN + 0.145 CYS + 0.322 GLN + 0.386 GLU + 0.538 GLY + 0.143 HIS_m + 0.324 ILE_m + 0.564 LEU_m + 0.57 LYS_m + 0.138 MET + 0.219 PHE + 0.313 PRO + 0.43 SER + 0.386 THR + 0.044 TRP + 0.182 TYR + 0.416 VAL_m + 29.04 ATP + 0.279 GLC6P + 1.3148 ATP + 0.109 GAP + 1.9184 AcCoA + 1.70476 ATP + 3.40952 NADH + 3.51852 H + 82 0.109 NADH + 0.008 GAP + 0.0704 AcCoA + 0.06256 ATP + 0.12512 NADH + 0.13312 H + 0.008 NADH + 0.324 AcCoA + 0.324 ATP + 0.288 NADH + 0.288 H -> Biomass + 0.07 ADP + 0.07 Pi + 0.07 ADP + 0.07 Pi + 29.04 ADP + 29.04 Pi + 1.3148 ADP + 1.3148 Pi + 1.9184 CoA + 1.70476 ADP + 1.70476 Pi + 3.40952 NAD + 1.49112 H2O + 0.109 NAD + 0.0704 CoA + 0.06256 ADP + 0.06256 Pi + 0.12512 NAD + 0.05472 H2O + 0.008 NAD + 0.162 CoA + 0.324 ADP + 0.324 Pi + 0.288 NAD + 0.162 CO2 0.5768 ALA + 0.411919 ASN + 0.330176 ASP + 0.35901 GLN + 0.464684 GLU + 0.683611 GLY + 0.669275 PRO + 1.010565 SER + 0.198476 ARG_m + 0.232693 CYS + 0.204171 83 HIS_m + 0.30046 ILE_m + 0.574408 LEU_m + 0.54615 LYS_m + 0.114338 MET + 0.366898 PHE + 0.979228 THR + 0.19333 TRP + 0.251256 TYR + 0.796933 VAL_m + 39.89 ATP -> MAB + 39.89 ADP + 39.89 Pi 6 Supplementary Figure 1. Flux balance model scheme. PEP regeneration pathway was only active at pH 7.8 and therefore the fluxes OAAmPEPm and PEPmPEP were constrained under all other conditions. 7 Supplementary Table 3. Measured specific consumption and production rates of amino acids given in nmol/106 cells/h, and biomass and mAb given in µg/106 cells/h. pH 7.2 Compound Glc Lac Gln Glu Ala Ser Gly Asp Arg His Cys Thr Ile Val Phe Tyr Lys Leu Pro Met Asn Biomass mAb WD 1.0 WD 1.5 -271.2 (±79.0) -162.1 (±27.0) 266.7 (±144.4) 35.3 (±23.8) -39.2 (±7.6) -75 (±17.1) 4.1 (±1.4) 8.1 (±4.7) 6.9 (±1.8) 13.2 (±0. 8) -12.3 (±3.8) -18.7 (±5.3) -0.9 (±1.1) 0.4 (±2.3) -0.5 (±0.1) -0.7 (±0.1) -4.1 (±1.9) -5.7 (±2.6) -1.9 (±0.8) -2.8 (±1.1) -1.3 (±0.5) -2.4 (±1.8) -4.9 (±1.4) -7.6 (±0.1) -9.3 (±4.1) -10.3 (±2.9) -7.8 (±2.6) -9.3 (±1.5) -2.8 (±1.1) -3.7 (±1.5) -2.4 (±0.9) -4.2 (±1.2) -5.2 (±0.7) -8.7 (±1.2) -9.2 (±3.8) -11.8 (±4.0) -4.5 (±2.0) -4.3 (±2.3) -2.6 (±1.1) -3.1 (±1.5) -3.5 (±0.9) -4.3 (±1.4) 10.7 (±2.3) 11.4 (±1.9) 1.2 (±0.1) 1.3 (±0.3) pH 6.8 pH 7.8 WD 1.9 WD 1.9 WD 1.9 -87.7 (±19.1) 6.7 (±10.7) -7.7 (±2.6) 3.3 (±0.8) 4.4 (±1.8) -3.8 (±0.9) -0.4 (±0.5) -0.5 (±0.1) -1.5 (±0.3) -0.5 (±0.3) -0.6 (±0.2) -1.6 (±0.5) -5.8 (±1.7) -3.9 (±0.9) -0.9 (±0.3) -0.7 (±0.1) -1.6 (±0.4) -5.4 (±0.7) -2.5 (±1.3) -0.9 (±0.2) -1.2 (±0.1) 6.3 (±0.9) 0.6 (±0.1) -31.5 (±16.2) -57.6 (±17.4) -12.8 (±5.1) 1.4 (±0.7) 5.7 (±2.2) -0.4 (±0.1) -1.2 (±0.7) -0.3 (±0.0) -0.8 (±0.5) -0.5 (±0.5) -1.4 (±0.3) -1.5 (±1.0) -3.1 (±1.1) -2.7 (±1.8) -0.8 (±0.6) -1.5 (±0.7) -1.0 (±0.5) -3.6 (±1.7) -1.1 (±0.2) -0.5 (±0.3) -1.1 (±0.5) 3.3 (±1.2) 0.9 (±0.2) -196.2 (±1.4) 408.7 (±51.1) -12.2 (±6.8) 6.6 (±0.5) 6.6 (±3.3) -5.2 (±0.3) 5.8 (±0.2) -0.7 (±0.1) -1.0 (±0.3) -0.8 (±0.2) -2.0 (±0.1) -0.5 (±0.1) -5.3 (±0.4) -4.4 (±0.5) -0.7 (±0.1) -0.9 (±0.1) -1.7 (±0.6) -4.6 (±0.3) -2.6 (±0.9) -0.8 (±0.1) -1.2 (±0.0) 0.9 (±0.9) 0.5 (±0.1) 8 Biomass composition The biomass composition of murine hybridoma cells averaged in previous work by Altamirano et al. (2001), Selvarasu et al. (2010) and Xie and Wang (1994) was obtained using the relative compositions from Sheikh et al. (2005) (Supplementary Table 4). The mole of each cellular component can be calculated from the biomass weight. Dry cell weight of CRL 1606 cells was considered as 250 pg/cell.5 Furthermore, diphosphatidyglycerol was not taken into consideration because of its negligible proportion in cellular composition. Therefore, protein, DNA/RNA, carbohydrate, cholesterol, and phospholipids (phosphoglycerides and sphingomyelin) were included in the equations of biomass formation. In addition to biomass synthesis, monoclonal antibody (mAb) synthesis was also considered in our network by utilizing the amino acids composition of IgG1 from Quek et al. (2010) (Supplementary Table 5). The theoretical amount of 4.3 ATP per mol amino acid is taken into account for protein synthesis. Monte Carlo error treatment method The reported 95% confidence interval was compared to the confidence obtained by a Monte Carlo error treatment method, where the error of the measured fluxes was assumed normally distributed and 10% of the measured flux, except for biomass and antibody (12%).7 From the resulting distributions 10 000 samples were randomly picked and used for FBA. The fluxes calculated from FBA also followed normal distribution except some fluxes having the constraint of an irreversible reaction. In those cases either the upper or lower bound was set to zero based on their distribution results and constraints. 9 Supplementary Table 4: Averaged dry cell composition (protein, carbohydrates, nucleotides, lipids) of murine hybridoma cells derived from literature 4. Metabolite nmol /μg DW 0.6 ALA ARG ASP ASN CYS GLN GLU GLY HIS ILE Protein LEU LYS MET PHE PRO SER THR TRP TYR VAL 0.377 0.359 0.288 0.145 0.322 0.386 0.538 0.143 0.324 0.564 0.57 0.138 0.219 0.313 0.43 0.386 0.044 0.182 0.416 Metabolite Carbohydrates Glycogen Nucleotides Lipids dAMP dCMP dGMP dTMP AMP CMP GMP UMP Cholesterol Phosphatidylcholine Phosphatidylethanolamine Phosphatidylinostitol Phosphatidylserine Phosphatidylglycerol Diphosphatidylglycerol Sphingomyelin nmol/μg DW 0.279 0.0148 0.0099 0.0099 0.0148 0.033 0.0551 0.0624 0.033 0.018 0.069 0.026 0.01 0.003 0.001 0.003 0.008 10 Supplementary Table 5: Amino acid composition of IgG1 derived from an average IgG1 protein sequence 6. molar mass-H2O g/mol AA ALA 71.08 ASN 114.1 ASP 115.09 GLN 128.13 GLU 129.12 GLY 57.05 PRO 97.12 SER 87.08 ARG 156.19 CYS 103.14 HIS 137.14 ILE 113.16 LEU 113.16 LYS 128.17 MET 131.19 PHE 147.18 THR 101.1 TRP 186.21 TYR 163.18 VAL 99.13 sum (nmol AA/μg mAB) mass fraction g AA/g mAB 0.041 0.047 0.038 0.046 0.06 0.039 0.065 0.088 0.031 0.024 0.028 0.034 0.065 0.07 0.015 0.054 0.099 0.036 0.041 0.079 nmol AA/μg mAB 0.577 0.412 0.33 0.359 0.465 0.684 0.669 1.0106 0.198 0.233 0.204 0.3 0.574 0.546 0.114 0.367 0.979 0.193 0.251 0.797 9.264 11 Supplementary Table 6: Comparison of experimentally determined flux values from a 13-C tracer study8 and calculated fluxes using maximization of ATP as the objective function. The metabolic network built for FBA is based on the literature8 together with oxidative phosphorylation pathway (FADH2 -> 1.5 ATP and NADHm/NADH -> 2.5 ATP). Pentose phosphate pathway was excluded from the model resulting in 40 intracellular metabolites and 63 reactions. 19 extracellular fluxes were used for input in the model and 39 intracellular fluxes described commonly in both the literature and model were compared by the goodness-of-fit. Chisquare test was performed to obtain the minimized variance weighted sum of squared residuals8,9. Reactions G6P <-> F6P F6P -> DHAP + GAP DHAP <-> GAP GAP <-> [3PG] + NADH + ATP [3PG] <-> PEP PEP + ATP -> PYR PYR + NADH <-> LAC PYR <-> PYRm PYRm -> AcCoAm + CO2 + NADHm AcCoAm + OAAm -> CITm CITm <-> AKGm + CO2 + NADHm AKGm -> SUCm + CO2 + NADHm SUCm <-> FUMm FUMm <-> MALm + FADH2 MALm <-> OAAm + NADHm MALm <-> PYRm + CO2 + NADHm PYRm + CO2 + ATP -> OAAm MALm <-> MAL MAL <-> OAA + NADH CITm <-> CIT CIT + ATP -> AcCoA + OAA AcCoA + 0.899 ATP -> FA FA <-> FAm FAm -> AcCoAm + 0.899 ATP ALA <-> PYR SER -> GLY + Cl SER -> PYR AKGm <-> GLU GLU -> PRO GLN -> GLU ASP <-> OAAm 13 Calculated C 95% fluxes experimental confidence (max ATP) values interval 204.9 203.2 168.4 236.4 204.9 203.2 168.6 237.2 204.9 202.8 168.2 237.2 409.8 406 338.1 475 409.8 406 338.1 475 409.8 406 338.1 475 289.8 289.8 240.4 347.5 113.5 109.7 36.9 182.1 142.04 138.3 67.9 207.2 158.2 154.4 81.7 248.2 158.2 154.4 80.6 238.4 182.3 178.6 106.3 259.3 189.1 185.3 111.8 270.1 191.2 187.4 113.8 266.5 162.6 133.9 74.3 213.1 28.6 53.5 32.5 84.8 0 24.9 7.1 56.5 -0.0338 0 -77 0 -0.0338 0 -77 0 0.0338 0 0 77 0.0338 0 0 77 1.6353 1.6 0.9 82.1 0 0 0 41.1 0 0 0 41.1 -6.6 -6.6 -7.3 -6.1 5.1 5.2 4.5 6.1 0.04 0 0 0.8 -24.1 -24.2 -30.5 -16.9 2.1 2.1 1.9 2.4 33.1 33.8 26.9 40.5 -4.5 -4.4 -5.1 -4.1 12 ASP -> ASN 2.2 THR -> AcCoA + GLY 1.6 MET + CO2 -> SUCm + CO2 + Cl + NADHm 0.7 VAL + CO2 -> SUCm + 2 CO2 + 3 NADHm + 2.2 FADH2 ILE + CO2 -> SUCm + AcCoAm + CO2 + 2 4.0 NADHm + FADH2 PHE -> FUMm + 2 AcCoAm + CO2 1.3 TYR -> FUMm + 2 AcCoAm + CO2 0.8 LEU + CO2 -> 3 AcCoAm + CO2 + NADHm + 2.7 FADH2 χ20.025 = 0.48 < h= 8.6 < χ20.975 = 9.5 (DF=4) 2.2 1.6 1.6 2 0.9 0.9 2.5 2.1 2.1 2.1 1 2.8 4 2.7 4.8 1.2 0.8 0.6 0.4 1.6 1.1 2.7 1.1 3.6 13 Supplementary Figure 2. Glutamine profile during standard batch cultivation. Glutamine depletion was measured after 2.1 days of batch culture. 14 Supplementary Figure 3. Intracellular fluxes calculated by flux balance analysis at three different time points (WD 1.0, WD 1.5, WD 1.9) for standard pH 7.2 culture. Numbers correspond to reactions specified below. 15 Supplementary Figure 4. Comparison between intracellular fluxes at culture pH 7.2 and pH 6.8 calculated by flux balance analysis at WD 1.9. Numbers correspond to reactions specified below. 16 Supplementary Figure 5. Comparison between intracellular fluxes at culture pH 7.2 and pH 7.8 calculated by flux balance analysis at WD 1.9. Numbers correspond to reactions specified below. Reactions 48-49 included to metabolic network as additional constraints according to gene expression data. 17 References 1. Altamirano C, Illanes A, Casablancas A, Gámez X, Cairó JJ, Gòdia C. Analysis of CHO cells metabolic redistribution in a glutamate-based defined medium in continuous culture. Biotechnology Progress. 2001;17(6):1032–41. 2. Selvarasu S, Karimi IA, Ghim G-H, Lee D-Y. Genome-scale modeling and in silico analysis of mouse cell metabolic network. Molecular Biosystems. 2010;6(1):152–61. 3. Xie L, Wang DI. Stoichiometric analysis of animal cell growth and its application in medium design. Biotechnology and Bioengineering. 1994;43(11):1164–74. 4. Sheikh K, Förster J, Nielsen LK. Modeling hybridoma cell metabolism using a generic genome-scale metabolic model of Mus musculus. Biotechnology Progress. 2005;21(1):112–21. 5. Zupke C, Stephanopoulos G. Intracellular flux analysis in hybridomas using mass balances and in vitro (13)C NMR. Biotechnology and Bioengineering. 1995;45(4):292–303. 6. Quek L-E, Dietmair S, Krömer JO, Nielsen LK. Metabolic flux analysis in mammalian cell culture. Metabolic Engineering. 2010;12(2):161–71. 7. Goudar CT, Biener R, Konstantinov KB, Piret JM. Error propagation from prime variables into specific rates and metabolic fluxes for mammalian cells in perfusion culture. Biotechnology Progress. 2009;25(4):986–98. 8. Ahn WS, Antoniewicz MR. Metabolic flux analysis of CHO cells at growth and non-growth phases using isotopic tracers and mass spectrometry. Metabolic Engineering. 2011;13(5):598– 609. 8. Pardelha F, Albuquerque MGE, Reis M a M, Dias JML, Oliveira R. Flux balance analysis of mixed microbial cultures: application to the production of polyhydroxyalkanoates from complex mixtures of volatile fatty acids. J Biotechnol. 2012;162(2-3):336-45. 9. Al-Rubeai M, Fussenegger M. Systems Biology. (Al-Rubeai M, Fussenegger M, eds.). Dordrecht: Springer Netherlands; 2007. 18