CHE 101: Fundamentals of Inorganic Chemistry
advertisement

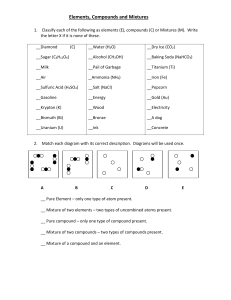
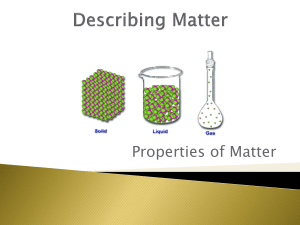
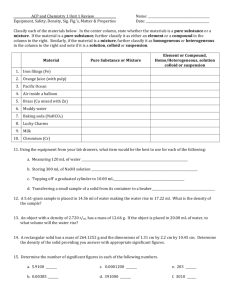
CHEM 121: Introduction to Chemistry Classify to the best of your ability each of the following substances as a mixture or a pure substance. If a mixture, is it homogeneous or heterogeneous? If a pure substance, is it a compound or an element? mercury in a thermometer tea a classroom full of students ethanol baking soda carbon monoxide aluminum foil a new penny a flower neon gas A clear liquid reacts with a blue liquid to form a white solid (that always melts at 880oC) and a colorless gas. Both liquids and the solid may be broken down into something simpler by chemical means; the gas cannot. Classify each of the following as a compound, element, or mixture--or state "not enough information to determine". blue liquid clear liquid + blue liquid white solid colorless gas Is this a chemical change or a physical change?