Chapter 14 free response 2ClO2 (aq) + 2OH-1 → ClO3
advertisement

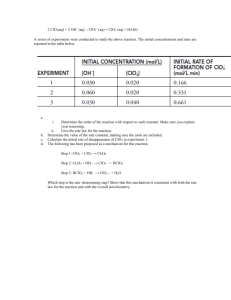
Chapter 14 free response 2ClO2 (aq) + 2OH-1 → ClO3-1 (aq) + ClO2-1 + H2O (l) A series of experiments were conducted to study the above reaction. The initial concentrations and rates are in the following table. Experiment [OH-1] 1 0.030 0.020 0.166 2 0.060 0.020 0.331 3 0.030 0.040 0.661 [ClO2] initial rate of formation of ClO3-1 1. Determine the order with respect to each reactant and justify your answer. 2.. give the rate law for the reaction 3. Determine the value of k. 4. Calculate the initial rate of disappearance of OH-1 in the second experiment. 5. The following is a proposed mechanism for this reaction: Step 1 : ClO2 + ClO2 → Cl2O4 Step 2: Cl2O4 + OH-1 → ClO3-1 + HClO2 Step 3: HClO2 + OH-1 → ClO2-1 + H2O Which step is the rate determining step? Show that this mechanism is consistent with both the rate law for the reaction and the overall stoichiometry. 2. For the reaction of (CH3)3CBr with OH− (CH3)3CBr + OH−(CH3)3COH + Br− The following data were obtained in the laboratory. TIME (s) [(CH3)3CBr] 0.0 0.100 30.0 0.074 60.0 0.055 90.0 0.041 (a) Determine the order of this reaction. Sketch your graph. (b) Determine the value of the rate constant and include proper units. (c) At what time is the concentration of (CH3)3CBr equal to [0.086]? Justify your answer. 4.