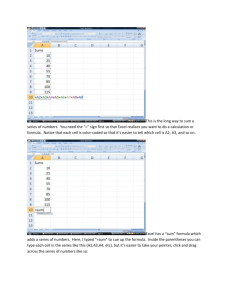
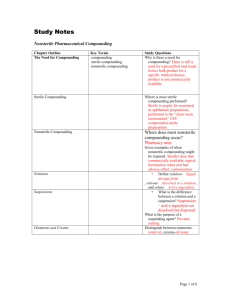
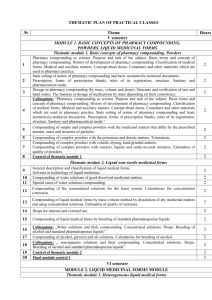
20 Paul David Way Stoughton, MA 02072 Phone & Fax (781) 297
advertisement

500 West Cummings Park, Suite 3475 Woburn, MA 01801 781-933-1107 Fax 781-933-1109 20 Paul David Way Stoughton, MA 02072 Phone & Fax (781) 297-0965 November 17, 2015 Honorable Jason M. Lewis, Senate Chair Joint Committee on Public Health State House Room 511B Boston, MA 02133 Honorable Kate Hogan, House Chair Joint Committee on Public Health State House, Room 130 Boston, MA 02133 Re: Testimony in support of H.2061: An Act Relative to Expanding Veterinary Treatment Practices Dear Chairman Lewis, Chairwoman Hogan, and honored members of the committee: The Massachusetts Pharmacists Association (MPhA), representing the interests of pharmacists from all practice settings, and the Massachusetts Independent Pharmacists Association, representing the interests of Independent Pharmacies in the Commonwealth offer our support along with recommendations to amend H.2061. The Associations agree with the premise and purpose of this bill: Veterinarians are in need of certain compounded medications for “office use” to treat animals with emergent conditions; and amendments to provisions created by Chapter 159 of the Acts of 2014 are required to permit a compounding pharmacy to engage in non-prescription specific, anticipatory compounding if it is to be the source of these medications. The associations request that the Committee consider the following recommendations prior to taking action on this bill: 1. As an alternative to a change in the definition of compounding, the Massachusetts Board of Registration in Pharmacy (MA BORP) could be authorized to grant waivers to sterile and complex non-sterile compounding pharmacies licensed by the state of Massachusetts to supply licensed veterinarians with approved emergent compounded medications for office use. 2. The Boards of Pharmacy and Veterinary Medicine should be required to promulgate joint regulations to enact this law that include the following: a. A process for the creation of a list of compounded medications necessary to address emergent situations and a process by which this list can be amended. b. A requirement that veterinarians follow Board of Registration in Pharmacy guidelines for drug storage including temperature and humidity monitoring to protect pharmacies from unwarranted claims of improperly compounded medications. These veterinarians shall be subject to inspection by the MA BORP for appropriate drug storage only. c. A requirement that the Veterinary practice be required to provide the pharmacy with timely and accurate records accounting for all compounded medications received. Our interpretation of federal policy suggests that compounding animal drugs for office use does not run afoul of federal laws or regulation, nor would it require registration with the Food and Drug Administration (FDA) as required by M.G.L. subsection (c) of section 39F of Chapter 112. This subsection states: An entity that intends to compound and distribute a sterile drug preparation or a complex non-sterile drug preparation to pharmacies, wholesalers or prescribers within or outside of the commonwealth: (i) in anticipation of a prescription… shall adhere to the most current standards established under cGMP when engaging in any form of compounding. Such pharmacies shall obtain and hold a manufacturer’s license appropriate to this practice, from the federal Food and Drug Administration, before engaging in any sterile compounding or complex non-sterile compounding. The FDA, which has long held that non-prescription anticipatory compounding to be illegal in the absence of a manufacturer’s license, now provides an alternative to a manufacturer’s license for compounding pharmacies engaged in this activity. Under the recently enacted section 503B of the federal Food, Drug, and Cosmetic (FDC) Act, the FDA calls for these pharmacies to voluntarily register as “Outsourcing Facilities”. However, an FDA guidance Q&A states that a facility that engages in this activity only for animal drugs should not register as an Outsourcing Facility because the drugs it produces will not be eligible for the statutory exemptions described in section 503B of the FDC Act. In essence, the FDA has chosen to put the onus of regulation and oversight for compounding for office use for animal drugs solely on the states. Therefore, in the interest of providing veterinarians with medications needed to properly treat animals in emergent conditions, we ask the Committee to amend and move this important bill forward with a favorable recommendation. Sincerely, David Johnson Executive Vice President, MPhA Todd Brown, Executive Director, MIPA