L5-virulence
advertisement

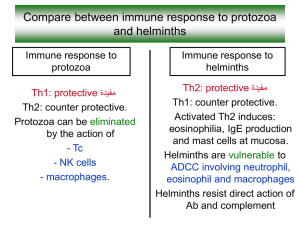
Lecture 5 – Host-Parasite Coevolution II: Virulence, resistance & Tolerance I. Variation in host damage Observation: Variation in a microbe’s ability to damage a host exists. Microbes can be mutualistic, commensal and parasitic, even within the same genera or species. For example, Escherichia coli exhibit a large range of host interactions from mutualistic (producing vitamin K) to parasitic (producing death). Question: What causes this variation? A. Host damage is caused by a parasite’s pathogenicity. Pathogenicity is the parasite’s ability or potential to cause host damage (e.g. the mechanism of damage). B. The extent of host damage (in terms of fitness) induced by parasite pathogenicity is termed virulence (e.g. the extent of damage) II. Pathogenesis – the generation of suffering A. Impairing host capabilities 1. Direct interference - some parasites directly interfere with organ function. For example, some trematodes infect fish gills, reducing gas exchange efficiency. Also, some parasites may hide in immune privileged sites (e.g. the eye or testes) causing organ damage that does not necessarily benefit the parasite (e.g. blindness or sterility) B. Destruction of tissues – Intended damage to host systems that benefit parasite transmission. For example, host castration. C. Virulence factors – any parasite derived factor that allows the parasite to enter the host, persist, multiply and/or transmit. 1. Adhesion factors (adhesins) – commonly found in bacteria. Molecules (e.g. polysaccharides) that promote adhesion to internal tissues (e.g. epithelial cells). EXAMPLES: - Fimbriae – bristle-like hairs that coat bacterial cell wall that facilitate adherence to other bacteria or host cells. - Pili – Long appendage that allows bacteria to engage in DNA transfer (i.e. conjugation) 2.Colonization factors – allows pathogen to survive in hostile host environment EXAMPLE: Helicobacter pylori produce urease (the colonization factor) that neutralizes the gastric acid in the stomach allowing them to establish. This ultimately leads to peptic ulcers. 3. Invasion factors – Molecules that facilitate entry into host cells or movement throughout the host. EXAMPLE: malaria induces uptake into host cells (endocytosis) by binding with host cell surface receptors. EXAMPLE: Leishmania degrades the extra-cellular matrix to facilitate movement throughout the host’s body. 4. Immune evasion factors – immune evasion factors – molecules that inhibit host immune defense (see lecture 4) Lecture 5 – Host-Parasite Coevolution II: Virulence, resistance & Tolerance EXAMPLE: Ebola virus produces numerous factors that interfere with the host’s immune response, such as phagocyte apoptosis, interferon inhibition and antigen overload. 5. Toxins – parasite compounds that damage the host. Often act like enzymes (e.g. proteases) A. Endotoxins – compounds associated with the parasite itself (e.g. bacterial LPS). Endotoxins are among the most potent toxins for mammals (e.g. 107 times more potent than strychnine or 105 times more potent than snake venom. Abundant LPS can lead to septic shock. B. Exotoxins – proteins released from parasites; can destroy host tissues or interfere with normal cell function far from the point of release. Can be categorized by target tissues: 1. Cytotoxins – kill cells 2. neurotoxins – interfere with nervous system (e.g. tetrodotoxin) 3. Enterotoxins – affect GI tract epithelia 4. Cardio toxins – affect heart D. Immunopathology – tissue damage caused by a (sometimes inappropriate) host immune response. EXAMPLE: Septic shock – Massive amounts of LPS can cause a massive inflammatory response. This causes vasodilation allowing fluid to leave the vessels and enter the tissues. EXAMPLE: gamete damage – Free radicals are excellent immune effectors produced by various host cells. However, free radicals are indiscriminant and cause host cell damage, especially gametes. II. Virulence A. Measures of virulence 1. Numerous measures including rate of host mortality, degree of host morbidity, rate of cell lysis (if a bacteriophage) and the loss in host fitness (offspring production). B. Evolution of virulence 1. Non-adaptive virulence – virulence and parasite transmission rate (i.e. parasite fitness) are not correlated a. coincidental virulence – parasite accidentally finds itself in a host with whom it has no co-evolutionary history. Result may be a highly virulent host response EXAMPLE: Rabies and ebola b. Short-sighted virulence – virulence is an inadvertent consequence of mutation and selection within the host; enhanced by parasite competition within the host. This may be due to the parasite invading tissues that offer no advantage in parasite transmission. EXAMPLE: Polio 2. Adaptive virulence Lecture 5 – Host-Parasite Coevolution II: Virulence, resistance & Tolerance a. Avirulent hypothesis – parasite and host co-evolution will eventually result in benign relationship (a.k.a. conventional wisdom hypothesis) - Logic: based on too much host damage by parasite would lead to host loss, thereby harming the parasite. Thus, parasite produces more porpagules by ensuring host survival. - Assumption: virulence and transmission rate negatively correlated. - Problem: does not account for within host competition among parasite strains. b. Trade-off hypothesis – virulence evolves optimally within the constraint of a virulence-host life span trade-off. 1. Assumptions Assumption 1: relationship between parasite virulence and transmission is positive and decelerating. Assumption 2: Deceleration due to a cost to virulence in terms of host survival or use as a host. 2. Basic reproductive number (R0) – the average number of new infections (secondary cases) caused by the original infection (primary case) in a wholly susceptible population. It measures the maximum reproductive potential of a parasite between one generation and the next for a given host population in a given environment. If R0 > 1, parasite will spread in population (leads to epidemic) If R0 is < 1, parasite will decline in population Depends on: - Transmission rate – probability that parasite infects next host upon encounter - Host population size in a wholly susceptible population - Background host mortality - Host recovery rate - Parasite virulence 3. Most likely the correct model of virulence evolution