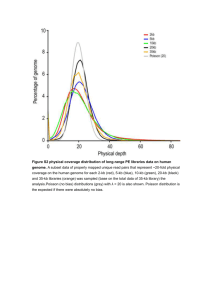
Response letter We would like to thank the Reviewers for having
advertisement

Response letter We would like to thank the Reviewers for having taken in reviewing our manuscript so thoughtfully. We have carefully taken the comments into consideration in preparing our revision and we hope that the paper is now suitable for publication in your journal. The English style of the paper has been revised carefully. Responses to the Reviewers are reported as follows: -----------------------------------------------------------------------------------------------------------------------Referee 1: Major Compulsory Revisions 1. Some additional descriptions of methods could be helpful to the general reader. a. SIR and SMR seem to be calculated until 2007. It is not clear what is the contribution of patients enrolled after 2007. Were they included in the analyses? The contribution of patients enrolled/active after 2009 was not included in the analyses of SIR. This is explained in Methods [line 138]. b. In Italy, a large number of Cancer Registries are active with relevant heterogeneity among areas. It is not clear why authors chose 5 registries. Does it depend on the area of residence of enrolled patients? Authors should clarify their assumption. We have clarified these points in Methods [lines 151-156]. 2. a. b. c. d. A few errors in Table 2 should be corrected Lip has a code C00. Is it included or not? The order (by ICD or frequency) of presentation is unclear. Cervix uteri was not included in AIDS-defining cancers before 1993 , please add a footnote. Anus (C21) was associated to HIV in all studies. I suggest to show results separately for this neoplasm. e. Skin cancer (non-melanoma) is not currently reported by cancer registries. Authors should clarify how they obtained IR for SIR calculation. f. The footnote OTHER CANCER reports the code C03-C06, which is also included in the C01C10 group shown in the main Table. Please check. We apologize for these errors. We have checked the Table 2 and corrected the errors. Anus (C21) is reported together with Colon or Rectum by cancer registries. The SIRs were ordered by ICD. A footnote about Cervix uteri was added and the footnote OTHER CANCER was modified. 3. The results presented in Tables 4 and 5 should be clarified. Authors should select the main variables needed to compare and summarize the most relevant estimated risks. These two Tables should be presented as an Appendix, if necessary. I suggest to use consistently 1 or 2 decimal points. Finally, p-values provide the same information of CI and could be omitted here. In order to make results clearer and to evaluate possible bias, the following changes have been made: a) the analysis of two different multivariate Poisson regression models has been deleted, and only one multivariate Poisson regression model was fitted including all covariates, e.i. socio-demographic, calendar and time-period, and clinical variables (Table 4). Cancer occurrence, AIDS events, CD4 cell count and antiretroviral therapy were included as time-dependent covariates, whereas gender, age at enrolment, hepatitis C virus co-infection and year of enrolment were included as fixed covariates. The selection of variables for the most parsimonious model was performed using a backward stepwise procedure, with p=0.20 for retaining each variable in the model. b) the Poisson regression models performed for the three patient-categories (ADC, NADC and free-cancer) is not included in this version. c) we have clarified Table 4, so we would like to keep both p-value and CIs in it. In order to evaluate the effect of antiretroviral therapy on risk of death, we have also fitted a marginal structural model, with cancer occurrence, AIDS events, and CD4 cell count as time-dependent covariates. 4. A number of previous studies, both population-based (Serraino Cancer 1997,Serraino JAIDS 2009, Zucchetto CID 2010) and clinical-based (Dorrucci JAIDS 1995, Guaraldi JAIDS2014), have addressed the topic in Italy. Authors should discuss similarities and differences with results obtained by comparable studies in the same area. Thank you for the articles suggested. We agree that these studies have provided interesting data on cancer epidemiology in HIV-positive subjects in Italy and most of them have been considered in our paper and added as references. However, these studies were not comparable properly with our as regards NADC and ADC incidence, because of selection criteria, as they included only newly seroconverted, naïve or AIDS patients. 5. Discussion could be shortened. Discussion has been shortened. Minor Essential Revisions 1. Please, add page numbers to the manuscript. We have added page numbers to the manuscript. 2. Page 4. Please specify how fitted values in Figure 1 were calculated. We have specified in Methods how fitted values in Figure 1 were calculated [lines 164167] 3. Consistently throughout the manuscript (Abstract, Table 3, text ), please state that analyses were stratified in <1998 and >=1998 (not before or after 1998). The expression“<1998 and >=1998” was changed into “before or after 1998” in the whole manuscript and a definition of “before or after 1998” was added in Methods [lines 143145]. ---------------------------------------------------------------------------------------------------------------------------------------------Referee 2: Major Compulsory Revisions 1. In the methods MASTER cohort included about 24,500 HIV-infected adults starting from 1986 but in the analysis the final number of patients included was 16,268 (66.4%) but not justification of the excluded patients or their characteristics were reported. Two MASTER centers have been excluded because did not codify cancer cases. We have explained this point in Methods [lines 99-100] . 2. In the methods a definition of lost to follow-up was enunciated but it is not clear if those lost to follow-up still contribute to the time at risk until last observation. Patients lost to follow-up have been included, and they contribute to time at risk until last observation. 3. A quantification of losses to follow-up should be included in order to elucidate its impact on the final cohort. A quantification of losses to follow-up has been included in Results [lines 191-192]. 4. Moreover the contribution in terms of time-at-risk given by patients entering in the cohort in the last years (>2008) to the final follow-up has to be considered not so relevant. SIR seems not to be calculated after 2007 and SMR not calculated after 2010, but still the period 2006-2012 is still used as reference category in the Poisson regression analysis. Especially when considering lately developing cancer such as solid cancers in the NADC group analysis, using this class as reference category could introduce a bias. In this revised version of the article, SIRs were calculated until 2009 and SMRs until 2012. The SMR analysis is an external comparison between the overall mortality in our cohort and that in the general population. On the contrary the Poisson analysis is an internal comparison which evaluates the factors associated with mortality in HIV-infected patients. Therefore we used the 2006-2012 period as the reference for assessing the association of year of enrolment with death using a Poisson model in the internal comparison, whereas the comparison with the general population (external comparison) was referred to the whole observational period, up to 2012. 5. In the methods, it is not clear if time-at-risk was ended at cancer diagnosis for those with a cancer diagnosis, and how time-at-risk was calculated when performing site-specific SIR estimations. We apologize for this, the statistical analysis in Methods has been clarified [lines 137-139]. 6. The choice of comparing cancer incidence using only 5 cancer registries should be clarified as well as the limitation to incidence date up to 2007 since data from all Italian cancer registries are available up to 2009 for more than 25 population based registries either using official data form IARC (http://ci5.iarc.fr/Default.aspx) than more updated data from Italian Cancer Registries (http://itacan.ispo.toscana.it/italian/itacan.htm). The better criteria to be used to compare incidence rates should be based on area of residence of the patients (if this information is accessible) using the available data (considering of course that not all registries have contributed to the whole period) and not using only 5 registries, in this way accounting to the geographical differences shown in cancer incidences in Italy by cancer registries. We have clarified these points in Methods [lines 151-156]. 7. In Table 2 classification of cancer cases according to ICD-10 should be checked since, some types is not clear if they are included and in which group, e.g lip cancer (C00) in the first group of NADCs and C03-C07 also included in the other cancers group. We apologize for these errors and we thank the reviewer for his note. The Table 2 has been modified. 8. In the Poisson analysis is not clear why a single multivariate model was not used, in order to better sustain considerations given in the discussion such as the increased risk in the earlier years of cohort-entry justified by the presence of a larger proportion of IDUs in these yours: this approach could allow to quantify the independent contribution of the single variable considered. In the choice of variables to be included in the final model a more relaxed approach should be considered, including those with a p-value lower than 0.10 or 0.20 eventually performing a backward elimination procedure to define the final model. The analysis of two different multivariate Poisson regression models has been deleted, and only one multivariate Poisson regression model was fitted including all covariates, e.i. socio-demographic, calendar and time-period, and clinical variables (Table 4). The selection of variables for the most parsimonious model was performed using a backward stepwise procedure, with p=0.20 for retaining each variable in the model. [lines 181-183]. 9. The use of time-dependent variables such as cancer occurrence, previous AIDS event or CD4+ nadir as time-independent characteristics should be cautionally performed and discussed. Cancer occurrence, previous AIDS event, count of CD4 and antiretroviral therapy were included as time-dependent covariates, whereas gender, age at enrolment, hepatitis C virus co-infection and year of enrolment were included as fixed covariates [lines 170-174]. 10. When considering previous AIDS event it is not clear when analyzing mortality in patients who developed a NADC, how ADC eventually occurring were considered. The use of era of antiretroviral therapy instead of regimen actually performed from the patient it is not clear, since using this approach the conclusion that cART initiation should be initiated independently to CD4+ cell counts is not clearly consistent. In the mortality analysis, ADC were not included in AIDS events (see Methods lines 172173). Cancer occurrence was included in the model as time dependent variable. Therefore a patient developing NADC and ADC in separate years (none developed both of them in the same year) was considered in both categories of exposure, with each other adjusted risk estimates. We agree that our conclusions on the effect of cART were not supported adequately by the data and therefore we have deleted them. 11. In the discussion, that should be shortened, no mention and comparison to what was found in similar studies on cancer incidence and mortality performed in Italy on HIV/AIDS patients. Discussion has been shortened and a comparison with similar studies was added. 12. SIR and SMR should not be compared directly and/or considerations should be smoothed accordingly. We have changed the discussion accordingly. 13. The contribution of cross-checks with mortality registries should be quantified. A cross-check with mortality registries has been performed in one third of patients included in this study [lines 128-129]. Minor Essential Revisions 1. The use of before and after should be uniformed through the text since when considering cART era before 1998 is intended up to 1997 (<1998) while when using birth’s year before 1970 actually includes this last year (<=1970). The expression“<1998 and >=1998” was changed into “before or after 1998” in the whole manuscript and a definition of “before or after 1998” was added in Methods [lines 143144]. 2. The ICD classification is indicated for the 9th revision as International Classification of Diseases while for the 10th revision as the International Statistical Classification of Disease, please uniform. We have changed “International Statistical Classification of Disease” into “International Classification of Diseases” [lines 124-125]. 3. In Table 2, the list of cancer types does not seems to follow-up a progressive order according to ICD-10 neither a ordered according to absolute numbers or estimated SIRs. The SIRs were ordered according to ICD-10. 4. In Table 5 the relative risk was indicated as RR in the univariate analysis and IRR in the multivariate analysis. We have deleted the Table 5 and related analysis. -----------------------------------------------------------------------------------------------------------------------Referee 3: 1. Methods section needs to be largely revised. Master cohort officially started later (late nineties) than 1986 and it should be clearly stated that data before the creation were retrospectively collected from participating centres. We have added the following sentence in Methods: “The Italian MASTER cohort is a hospital-based multicenter, open, dynamic cohort established in the mid-1990s, with retrospective patients’ enrolment from 1986 to 1997 and prospective recruitment subsequently.” [lines 96-98]. 2. The authors report that about 24.500 are included in the Master cohort but then only around 16.200 were included in this study. More details should be given about this issue. It seems that the major reason was related to missing CD4. The authors should also better clarify about patients “lost to follow-up”. Were they excluded from the analysis or were they considered at risk until the date of lost to follow-up? Two MASTER centers have been excluded because they did not codify cancer cases. We explained this point in Methods [lines 99-100]. A quantification of losses to follow-up has been included in Results [lines 191-192]. Patients lost to follow-up have been included contribute to time at risk until last observation. 3. The authors do not clarify if patients having already a cancer when enrolled were excluded when calculating the incidence rate. The patients with cancer diagnosis before enrolment in our cohort were excluded from analysis. We have added this point in Methods [lines 107-108]. 4. The descriptive data of people included in this analysis (table) show a very unusual birth-year distribution. In fact, more than 70% of people resulted to be born before 1950. This is strongly in contrast with the known epidemiology of HIV infection in Italy in which more than 80% of cases diagnosed with HIV were born after 1950. Further, the birth’s year distribution reported in table 1 is in contrast with the age-category distribution (this is instead sound!). We thank the reviewer for having noted this mistake, now birth’s year is not included. 5. Also the risk factor distribution does not seem to reflect the epidemiology of HIV in Italy, at least for the most recent years. We have checked the distribution of risk factors and confirm our data. There may be several explanations of differences in risk factor distribution in our cohort as respect to the other Italian studies. The MASTER cohort is the only Italian cohort that enrolls both treatment-naïve and treatment-experienced HIV infected patients, independently of HIV stage, whereas the other Italian cohorts used various selection criteria, including newly seroconverted, naïve or AIDS patients only. 6. The most important issue is related to the results reported in table 4 and table 5. First, these tables are also too filled in and difficult to read. Second, the analyses here reported do not include an important covariate that is the calendar period (e.g, divided in 5 years period or at least roughly divided by pre-HAART era and HAART era). Third, the stratification (table 4) in two multiple poisson regression models (roughly defined as time-period and clinical covariates) does not seem to provide clear information to better understand which are the most important characteristics associated with the outcome. I feel that the last model should include all covariates (socio-demographic, calendar and time-period, and clinical). The analysis of two different multivariate Poisson regression models has been deleted, and only one multivariate Poisson regression model was fitted including all covariates, e.i. socio-demographic, calendar and time-period, and clinical variables (Table 4). Cancer occurrence, AIDS events, CD4 cell count and antiretroviral therapy were included as time-dependent covariates, whereas gender, age at enrolment, hepatitis C virus coinfection and year of enrolment were included as fixed covariates. In order to evaluate the effect of antiretroviral therapy on risk of death, we have also fitted a marginal structural model, with cancer occurrence, AIDS events, and CD4 cell count as time-dependent covariates. 7. Regarding table 5 it is not clear to me if the risk-time for those with cancer started when they developed cancer or when they entered in the cohort. If the analyses considered as time zero the date of cohort entry this is conceptually wrong. We agree with this point, the Table 5 was deleted.