Test yourself: 1
advertisement

BB051B Applied Bioinformatics
Name:
Please answer all these questions and give the handout to the assistants!
Test yourself: 1
This page contains a randomly selected series of exam questions that we have used
in the past in real exams, and that you should be able to answer by now.
Exam questions used in the past:
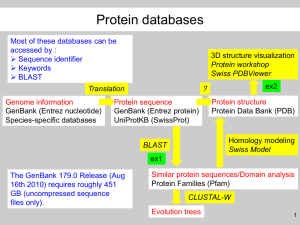
1. A colleague has sequenced a novel gene. He comes to you and asks for help.
What do you do first and why?
2. What are the major strengths and the major weaknesses of the SwissProt
database?
3. What is the role of the accession code in sequence databases?
4. What are the major advantages and disadvantages of WWW based servers?
1
5. What is the difference between a SNP and a mutation mentioned in OMIM?
6. What are BLAST and MRS abbreviations of, and what kind of searches can be
done with these two programs?
7. What is a Dayhoff matrix?
8. If a match from a BLAST database search is reported to have an E-value of
0.0, should it be considered highly insignificant or highly significant?
9. What is the role of the accession-code in databases?
10. Give 4 examples of protein features that can be found in the feature section of
a SwissProt file.
2
11. Sequence alignment is an important tool? Write down three scientifically
relevant biological questions that can be answered with/using sequence
alignment(s). (So, don't ask trivial things like "What does the alignment look
of...").
12. In January 2009, I ran BLAST on a bacterial protease against SwissProt. I
found a hit. In August 2012, I did the same run, and got the same hit, but this
time the E-score of this same hit was worse than in January. Why?
13. Below you see a part of a BLAST output. Explain:
1. Query
2. Subject
3. The occurrence of the region with the XXXXX
4. How to circumvent XXXX in the output
5. The –
6. The ...
Query: 361 ILNPYIINVTLAAKMCSQVLCQEQGVCIRKNWNSSDYLHLNPDNFAIQLEKGGKFTVRGK 420
ILNPYI.NV LAA.MCSQVLCQ.QGVCIRKNWNS...LHLNPDNFAI
GK.TVRGK
Sbjct: 361 ILNPYILNVALAARMCSQVLCQQQGVCIRKNWNSTNFLHLNPDNFAILS--RGKYTVRGK 418
Query: 421 PTLEDLEQFSEKFYCSCYSTLSCKEXXXXXXXXXXXXCIADGVCIDAFLKPPMETEEPQI 480
PTLED.EQF.EKF.CSCY TLSCKE
CIADGVCIDA.LKP METEEPQI
Sbjct: 418 PTLEDIEQFTEKFFCSCYGTLSCKEKADVADVDAVDVCIADGVCIDAYLKPTMETEEPEI 478
*END
3
Another question that often comes back is to tell me what A-K are in the BLAST
output below:
4
Test yourself: 2
This page contains a randomly selected series of exam questions that we have used
in the past in real exams, and that you should be able to answer by now.
Exam questions used in the past:
1. Which residues are hydrophobic?
2. Which residues are hydrophilic?
3. What is special about proline?
4. What is special about histidine?
5. Why is Proline often observed at the surface of a protein?
6. Why is Tryptophan the most conserved residue?
5
7. Align without gaps and explain your reasoning:
LLLDAWLLL with LLLDWLLL
8. Given a pattern definition from the PROSITE database: -G-[AS]-x-{RHK}-x3-CDoes the following sequence fulfill the pattern?
Explain why (not).
GASRLKSC
9. Why are Asp, Asn and Ser more often than other residues found in beta turns?
10. Mention two types of mutations (i.e. Phe -> Asp or Trp -> Ala) that generally
make proteins more stable (perhaps this question can only be answered in two weeks...)
11. Why is Leucine not a good active site residue?
6
12. Why is Cysteine seldom seen at the surface of a protein?
13. Why is Proline often observed at the surface of a protein?
14. What is helix capping?
15. Which residues are good for a helix? And why?
16. Why is the capping effect by charged amino acids stronger at the N-terminal
end than at the C-terminal end of a helix?
7
17. At which end of a helix can you find prolines, and why?
8