SCH 3U Name: Unit 4: Solutions Date: Factors That Affect Solubility
advertisement

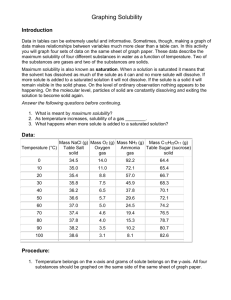
SCH 3U Name: __________________________ Unit 4: Solutions Date: __________________________ Factors That Affect Solubility Part 2 Another Way to Classify Solutions: Unsaturated, Saturated and Supersaturated Solutions Term Definition Solubility The solubility of a substance is defined as the maximum amount of solute that will dissolve in a given quantity of solvent at a specific temperature. A solute is generally considered “soluble” if more than 1 g of it will dissolve in 100 mL of the solvent. A solute is generally described as insoluble or sparingly or slightly soluble if it has a solubility between 0.1 g and 1 g per 100 mL of solvent. A solute is described as insoluble if less than 0.1 g of solute will dissolve in 100 mL of solvent. Note that this does not necessarily mean that none of the solute will dissolve in the solvent, just that not much of it does (at the temperature quoted). Unsaturated solution An unsaturated solution is one which could dissolve more solute in the solvent at a given temperature. Saturated solution A saturated solution contains all of the solute it can dissolve at the given temperature. To ensure a solution is saturated, people often mix the solution up so that some undissolved solid is visible at the bottom of the container. This ensures that the solution above the leftover solid has taken in all of the solute it can at the specified temperature. Supersaturated solution A supersaturated solution contains more dissolved solute than a saturated solution at the same temperature. For example, an aqueous solution might be heated so that more solute can be dissolved in the water than could be at room temperature. Then, if the solution is cooled slowly, often the solute will not come out of solution. This results in more solute being in solution once room temperature is again reached that would’ve been if the heating and solute loading step had not occurred. Temperature and Solubility a) The effect of temperature on the solubility of solids Refer to Figure 2 on page 316. 1) You are looking at solubility curves for several different ionic solids. A solubility curve shows the relationship between the solubility of a solute and the temperature of the solvent. What is the general trend in the solubility curve for most of the ionic solids shown? 2) Why is it that the solubility of a compound should always be specified at a certain temperature? 3) There are some Practice problems underneath Figure 2 on page 316 that continue on to page 317. Answer #1a, b, d and #2. 4) Is a solution of 50 g of KNO3(s) in 100 g of water at 40oC saturated, unsaturated or supersaturated? Explain. 5) Determine if each of the following solutions is unsaturated, saturated or supersaturated. i) 15 g of KClO3(aq) in 100 g of water at 40oC ii) 10 g of KClO3(aq) in 50 g of water at 65oC iii) 8 g of KClO3(aq) in 50 g of water at 50oC b) The effect of temperature on the solubility of liquids and gases Practice 1. Consider the solubitities of potassium chlorate, KClO3(s), methanol, CH3OH(l) and nitrogen gas, N2(g). Which substance’s solubility in water is LEAST affected by temperature? Explain. 2. Why are many species of fish less likely to be found in warm water than in cooler water? Pressure and Solubility Practice 1. A scuba diver experiencing the bends as a result of nitrogen bubbles in the blood may be treated in a recompression changer. The air pressure in this sealed chamber can be increased to many times the atmospheric pressure. Explain how a recompression chamber can be used to remove nitrogen bubbles safely. Factors that Affect the Rate of Dissolving Practice 1. Suppose you are dissolving a solid in a liquid. a) List 3 factors that affect the rate of dissolving. b) Identify a factor that affects both solubility and rate of dissolving.