on streptozotocin induced diabetic nephropathy. Four
advertisement

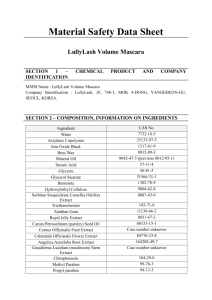
SUPPLEMENTARY MATERIAL Amelioration of diabetic nephropathy by orange peel extract in rats Parkar Nishad1,2 and Addepalli Veeranjaneyulu.2 1 Dr. Bhanuben Nanavati College of Pharmacy, Vile Parle West, Mumbai 400056 1,2 Department of Pharmacology, SPP School of Pharmacy and Technology Management, NMIMS University, Vile Parle (W), Mumbai, 400 056, India Abstract: The present study was aimed to evaluate the effect alcoholic orange peel extract (OPE) on streptozotocin induced diabetic nephropathy. Four weeks after the induction of diabetes, treatment with OPE (100 and 200mg/kg) was further given for four weeks. Treatment with OPE 200 improved renal functions and significantly prevented the raise in creatinine, urea and blood urea nitrogen (BUN) levels. Histological analysis of kidneys revealed that Tubulointerstitial Fibrosis Index was significantly decreased in OPE 200 group. The results indicated prevention of diabetic nephropathy in rats by OPE treatment and suggests OPE as a potential treatment option. Keywords: – Diabetic Nephropathy; Orange peel extract, Extracellular matrix. Experimental Collection of Plant material and preparation of OPE Fresh oranges were purchased from the local market of Mumbai, Maharashtra, India. Peels of the fresh fruits were separated and shade dried. The dried orange peels were sorted and the good quality peels were subjected to size reduction by pulverisation. The powdered peels were extracted using methanol as a solvent in a soxhlet apparatus and completed with six cycles. The extract obtained was dried on a water bath followed by vacuum oven at 40 0C until completely dried. The dried extract was refrigerated until used. Phytochemical screening of the extract for flavonoids The dried orange peels were exhaustively extracted using methanol in a soxhlet apparatus. The fresh extract was subjected to preliminary qualitative phytochemical screening to confirm the presence of flavonoids. Standard tests for flavonoids (Shinoda test, lead acetate test and sodium hydroxide test) mentioned in literature were performed on the extract (Harborne JB., 1998). Total Phenolic Content The total phenolic content (TPC) of the extract was determined spectrophotometrically by Folin Ciocalteu’s reagent using gallic acid as standard. (Claudia Anesini, et.al 2008). The concentration of polyphenols in samples was derived from a standard curve of gallic acid ranging from 10-50 µg/mL. 1.0 mL of sample extract was transferred in triplicate to separate tubes containing 5.0 mL of a 1/10 dilution of Folin-Ciocalteu’s reagent in water. Then, 4.0 mL of sodium carbonate solution (7.5% w/v) was added. The tubes were vortexed and allowed to stand at room temperature for 60 mins. The absorbance of the resultant color developed was measured at 765 nm against reagent blank. The TPC was expressed as gallic acid equivalents (GAE) in mg/g of the orange peel extract (n = 3). Total Flavonoid Content The total flavonoid content of orange peel extract was determined using AlCl 3 method. (Brighente et.al.2007) Briefly, 0.5 ml of 2% aluminium chloride (AlCl3) in methanol was mixed with the same volume of methanolic solution of orange peel extract. The mixture was vortexed and kept at room temperature for 1 h. The absorbance of the samples was measured at 415 nm using a spectrophotometer versus reagent blank sample. Total flavonoids content was expressed as quercetin equivalents (mg QE/g dry extract). The assay was performed in triplicate and the mean values are reported. Drugs and chemicals Streptozotocin (STZ) was purchased from Sigma (St. Louis, USA). Minocycline was obtained as a gift sample from US Vitamins (Mumbai, India). All the diagnostic kits were purchased from Erba Diagnostics, Mumbai, India. All the other reagents used were of analytical grade purchased from Merck (Mumbai, India). Animals Female Wistar rats 10-12 wks (200-240 g) were used for the study. Animals were purchased from Haffkine Institute, Parel (Mumbai, India). The animals were housed in a clean environment at a temperature of 25±10C, relative humidity 45-55% under a 12 hr light/ dark cycle with food and water available ad libitum. All procedures met the guidelines of Institutional Animal Ethics Committee (IEC) and were approved by Committee for the Purpose of Control and Supervision of Experiments on Animals (CPCSEA). (Approval no. CPCSEA/IAEC/SPTM/P-21/2011) Preparation of the drug solution Orange peel extract (OPE) and minocycline were suspended in 0.5% CMC solution before use. STZ was dissolved in freshly prepared ice cold citrate buffer (pH 4.5). Induction and assessment of diabetes Female Wistar rats were fasted over night (12h) and diabetes was induced by a single dose of STZ (50mg/kg, i.p. 1ml/kg body wt.). For biochemical estimations, blood was collected by retro orbital bleeding under mild anesthesia in accordance with National Cancer Institute guideline for Rodent Blood Collection (2012). Further, during the experiment, blood was withdrawn from alternate eyes to reduce the damage and allowed timely recovery of the eye as suggested in the guidelines (NCI Guideline, 2012). The plasma glucose levels were monitored after 48 h of STZ injection using a glucose oxidase-peroxidase kit and weekly during the experiment. The animals with plasma glucose level of >350mg/dl were used for the study. Experimental design The animals were divided into 5 groups of 6 animals each as follows: Group 1 – Diabetic Vehicle control (DW + 0.5 % CMC), Group 2 - treated with OPE (100 mg/kg p.o.), Group 3 – treated with OPE (200mg/kg p.o.), Group 4 – treated with standard Minocycline (50 mg/kg p.o.). Age matched normal animals in Group 5 were untreated. Four weeks after induction of diabetes, the animals were further treated for the next four weeks daily p.o. Assessment of renal function At the end of eight weeks study protocol, the animals of each group were housed individually in metabolic cages for a period of 24 hr. Urine samples were collected under a layer of toluene. Blood was collected by retro orbital puncture, serum separated and stored at -800C for further analysis. The renal function was assessed by measuring serum and urine levels of creatinine, urea and albumin. Creatinine clearance was calculated as an index of glomerular filteration rate (GFR) .Creatinine clearance was calculated using the formula: [CreatinineUrine/CreatinineSerum]* [Vol. Urine (ml)/ time (hrs)*60] Systemic Blood Pressure After 8 weeks of treatment, rats were anesthetized with pentobarbital sodium and body temperature was maintained at 37 ± 1°C. Systemic blood pressure was monitored via a catheter inserted into the femoral artery. Blood pressure was recorded digitally through a data acquisition system (Iworx systems USA). Mean arterial pressure (MAP) was calculated using the following formula: MAP = diastolic pressure + 1/3 (systolic pressure - diastolic pressure). Renal Hypertrophy, histopathology The animals were sacrificed under anaesthesia and left kidney was excised from each of the sacrificed rats. The kidney was decapsulated and rinsed in saline, blotted dry on a tissue paper and weighed. The kidney/bodyweight ratio was calculated. Data was expressed as relative organ weight of one kidney to 100g of total body weight. Kidney samples were then transferred to 10% v/v formaldehyde in separate containers and subjected to histopathological analysis using H &E and Masson’s Trichrome staining using standard procedures. Stained sections were observed under light microscope. Tubulointerstitial Fibrosis Index Excess MMP-2 and MMP-9 activity resulted in remodelling of ECM. Tubulointerstitial fibrosis index was measured to evaluated severity of injury to renal tissue (Christine Maric, 2004). On 5 cortical and 5 outer medullary fields it was scored semi quantitatively. Severity of tubulointerstitial fibrosis was defined as tubular atrophy or dilatation, presence of inflammatory cells, deposition of ECM, and interstitial cell proliferation. The degree of tubulointerstitial fibrosis was graded on a scale of 0 to 4: grade 0, affected area 0% (normal); grade 1, affected area less than 10%; grade 2, affected area 10 to 25%; grade 3, affected area 25 to 75%; grade 4, affected area greater than 75%. Estimation of tubulointerstitial fibrosis was performed with the observer masked to the treatment groups. Statistical analysis Data was expressed as mean ± SD. The data was statistically analysed using Graph Pad Prism software (Graph Pad Inc., San Diego, CA, USA). For parametric data ANOVA (One way analysis of variance) was used for multiple comparisons. In case ANOVA showed significant differences, post hoc analysis was performed using Dunnet test. For non-parametric measures, Kruskal-Wallis test followed by Dunn’s post-hoc test were used. p < 0.05 was considered statistically significant. References Brighente, M. Dias, L.G. Verdi, and M.G. Pizzolatti. (2007) Antioxidant Activity and Total Phenolic Content of Some Brazilian Species. Pharmaceutical Biology, Vol. 45, No. 2, pp. 156–161. Christine Maric, Kathryn Sandberg, and Carmen Hinojosa-Laborde (2004) Glomerulosclerosis and Tubulointerstitial Fibrosis are attenuated with 17β-Estradiol in the Aging Dahl Salt Sensitive Rat. J Am Soc Nephrol 15: 1546–1556. Claudia Anesini, Graciela E. Ferraro, and Rosana Filip (2008) Total Polyphenol Content and Antioxidant Capacity of Commercially Available Tea (Camellia sinensis) in Argentina. J. Agric. Food Chem., 56, 9225–9229. Harborne JB., (1998) Phytochemical Methods: A Guide to Modern Techniques of Plant Analysis. (3rd edition) Chapman and Hall Co., New York, pp.1-302. National Cancer Institute Guideline for Rodent Blood Collection (2012). Available from http://ncifrederick.cancer.gov/Lasp/acuc/Frederick/Media/Documents/ACUC20.pdf Figure S1. Effect of 4-week treatment with OPE on A. Renal Tubulointerstitial Fibrosis Index and B. Renal histopathology with H & E Staining. The values are given as mean ± SD. *p < 0.05 when compared with vehicle treated diabetic group. A. B. A C B D E A –NORMO, B – DB + 0.5% CMC, C – OPE – 100, D – OPE 200, E- MINO