Soln2 - Temperature Dependence of CO2 in Water
advertisement

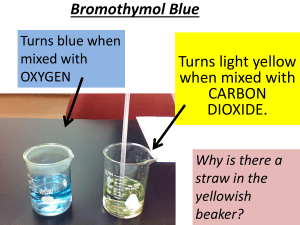
Solution Unit Name: ___________________________ Page 1 of 4 Temperature Dependence of CO2 in Water BACKGROUND: Champagne, soda pop, beer, natural soda water are all examples of carbon dioxide dissolved in water. As drinkers of these beverages probably know, the amount of gas changes as the liquid warms; the drink goes “flat.” The more CO2 dissolved in water makes the water more acidic. Ammonia will neutralize the acid. Ocean water is the largest reservoir of carbon dioxide in the world. The gas gets dissolved in it partly by the action of waves. Partly by the fact that rain drops also get carbon dioxide in them. Purpose: To study the affect of temperature on the solubility of carbon dioxide. MATERIALS: balance 1 Alka Seltzer tablet 3 beakers 250-mL bromothymol blue solution (BTB) (Acid/base indicator) 250 mL of room temperature water household ammonia (in dropper bottle) 3 test tubes (Medium size) ice cubes graduated cylinder hotplate PROCEDURE (ANSWER THE QUESTIONS IN THE PROCEDURE AS YOU GO) Fill in the prediction about the solubility of carbon dioxide in water. Carbon dioxide will be _____________ soluble in ______________ water. 1. Get one Alka Seltzer table, break into pieces and weigh a piece until you have exactly 1 gram of material. Do this again; so that you have 2 1-gram samples. The accuracy of your experiment will depend on how carefully you measure your quantities of Alka Seltzer. 2. Label 3 beakers; A, B, and C. Mark the 3 test vials in the same way. Use the graduated cylinder to measure exactly 100 mL of water for each of the beakers; in A put hot water that you have warmed in the microwave to about 60 OC, in B put 100 mL cold water from the refrigerator, and in C put 100 mL room temperature water to act as a control. In each of the 3 beakers put exactly the same number of drops of bromothymol blue (BTB) so that the same rich color is observed in all the containers. (20 drops of BTB is advisable). 3. Write down your prediction of what will happen when your samples of Alka Seltzer are added to the hot and iced water containers. Do you expect there to be a difference? If so, what will that difference be like? _____________________________________________________________________________ 4. Put 1 gram of Alka Seltzer in the hot water in the beaker and 1 gram in the cold water in the beaker. Observe the color changes and note the rates of the reactions. Write down your observations. __________ _____________________________________________________________________________________ 5. Compare the results with your predictions. Make a new prediction. Do you think there will a difference in acidity between containers A and B? If so, which will be more acidic? ___________________________ _____________________________________________________________________________________ 6. When all bubbling has stopped pour off 20 mL from each container into the 3 test tubes. Add household ammonia, drop by drop, to test tubes A and B only. Keep a record of the number of drops needed to change the liquids in A and B to the same color as that in your control test tube C. Swirl the test tubes Solution Unit Name: ___________________________ Page 2 of 4 gently between drops to mix the ammonia into solution Note: Wait at least a full minute after your liquid turns blue before you conclude that it matches the color of your control The color may fade. 7. Answer the questions above, develop a data table, and answer the questions online. OBSERVATIONS AND QUESTIONS: Record your observations and compare the number of drops of ammonia that it took to neutralize both hot and cold water. Copy the questions and save them using the file name your first name last initial-the title of the lab. Place the cursor at the end of the question and type your answers. Save the Answers and email the answers to me at deb50duncan@gmail.com : 1. Why was it necessary to be accurate in measuring the 2 Alka Seltzer portions? 2. Why was it important that the same amount of water and the same amount of BTB be in each of the containers? 3. What was the function of the control? 4. What does the number of drops of ammonia indicate? 5. How were your predictions confirmed or denied by the results? (to answer you have to state your predictions.) CONCLUSION AND CONNECTIONS: 1. How do you explain the difference in the amounts of ammonia used in the test vials? (If you haven’t done acid/bases ask me.) 2. What can you conclude concerning the water temperature difference and the solubility of carbon dioxide? 3. On the basis of what you have learned during this experiment answer the following: If the Earth’s climate warms because of additional carbon dioxide in the atmosphere and then the oceans of the world warm, what will happen to the ability of the oceans to absorb carbon dioxide? Will it increase, decrease, or remain the same? Why? Solution Unit Name: ___________________________ Page 3 of 4 4. If the amount of carbon dioxide in the atmosphere determines the amount of heating of the Earth by the greenhouse effect, how do you think the warming of ocean water will affect the global climate? 5. Write a paragraph or two that includes you conclusions and discuss the implications of what you observed on the future of our planet (Global Warming). Where are we heading in the future? Information: Ocean feedback effects Ocean CO2 chemistry - If a gas can dissolve in water, the way carbon dioxide does, the gas-liquid surface becomes a place of considerable molecular activity. Moving gas molecules dive into the surface of the liquid. Wind can stir up the water surface and mix the gas into the liquid. Within the water the water molecules move about and knock the gas molecules around. Some of the gas molecules are continually being pushed out at the surface. There is movement of the gas molecules both in and out of the liquid. At a specific temperature of air and water equilibrium is achieved. If the temperature changes, equilibrium is upset and a new equilibrium will be established at the new temperature. The ocean holds about 60 times as much carbon dioxide as the atmosphere does. Because of this fact, the condition under which the ocean water absorbs carbon dioxide controls the amount of carbon dioxide in the atmosphere. When the temperature of the ocean increases, the volume of carbon dioxide in the ocean will decrease. Solution Unit Name: ___________________________ Page 4 of 4