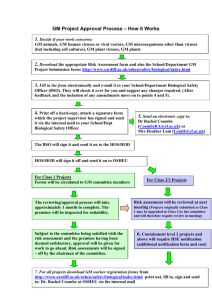
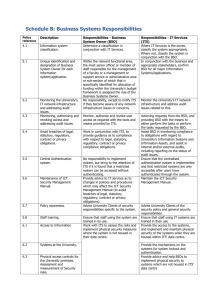
Form 5 Releasing Contained Laboratories
advertisement

Biosafety Unit, CRISP, FHML, MUMC+ Releasing form Contained Laboratories - Date and time maintenance activities: - Laboratory roomnumber : - Containment Level* : - Type apparatus, /leiding etc that needs maintenance: - Defect: : Clarification: The space in which the work should be carried out and the accessible parts of the device are cleaned and disinfected in accordance with the requirements as stated in the instruction disinfection. The disinfection is carried out with .......................................................................................... (disinfectant) Therefore, no microbiological risks are present during the repair. If there are doubts, consult the BSO for measures to be taken. ** Name : Position* : Department : Signature : Date and time of release : Name BSO (in case of ML-II, BSL2 or DM-II/DM-III) : Signature : * At Containment level ML-I, the Deppartmental Expert or Labmanager signs; From Containment level II/2 both BSO and Departmental expert od labmanager have to sign. ** There is still a contamination: …………………………