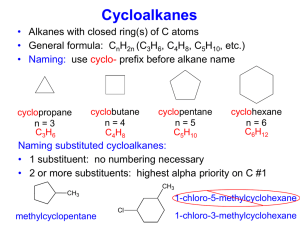
A. CH 3 CH 2 C
advertisement

C-H bonds in methane Figure 22.2: (a) Lewis stucture of ethane (C2H6). (b) molecular structure of ethane HYBRIDIZATION? Name calling H H C H H methane Name calling H H H C C H H H ethane Name calling H H H H C C C H H H H propane Name calling H H H H H C C C C H H H H H butane Name calling H H H H H H C C C C C H H H H H H pentane Name calling H H H H H H H C C C C C C H H H H H H H hexane Name calling H H H H H H H H C C C C C C C H H H H H H H H heptane Name calling H H H H H H H H H C C C C C C C C H H H H H H H H H octane Name calling • • • • • • • • CH4 C2H6 C3H8 C4H10 C5H12 C6H14 C7H16 C8H18 methane ethane propane butane pentane hexane heptane octane Figure 22.3: Structures of (a) propane (b) butane H H H H C C C H H H H H H C H HYBRIDIZATION? H C H H C H H C H H Figure 22.3: Structures of (a) propane (b) butane H H H H C C C H H H H H H C H H C H H C H H C H H Figure 22.3: Structures of (a) propane (b) butane C C C C C C C Figure 22.3: Structures of (a) propane (b) butane C C C C CH CH3 CH3 CH H H H H H C C C C H H H Reactions of Alkanes Combustion alkane + O2 CO2 + H2O + heat Learning Check Alk3 Complete and balance the reaction for the complete combustion of C7H16 Solution Alk3 Step 1 C7H16 + O2 CO2 + H2O Step 2 C7H16 + O2 7 CO2 + 8 H2O Step 3 C7H16 + 11 O2 7 CO2 + 8 H2O Combustion In the Cell Metabolic oxidation is combustion C6H12 O6 + 6O2 6CO2 + 6H2O + heat glucose • How does this reaction occur in living organisms? Aerobic Oxidation Occurs in a Mitochondrion Located within a Cell Branched Alkanes Structural Formulas Structural Isomers Alkyl Groups Branches on carbon chains H H H C H H H H C C H H methane H ethane Alkyl Groups Branches on carbon chains H H H C H H H C C H H CH3 methyl CH3CH2 ethyl Branched Alkanes CH3 CH3CHCH3 methyl groups CH3 CH3 CH3CHCH2CHCH3 Naming Branched Alkanes CH3 methyl branch CH3CH2CH2CHCH2CH3 6 5 4 3 2 1 Count Naming Branched Alkanes CH3 methyl branch CH3CH2CH2CHCH2CH3 6 5 4 3 2 1 Count 3-Methylhexane on third C CH3 six carbon chain group Naming Summary 1. Count the C’s in the longest chain 2. Name each attached group 3 Count the longest carbon chain to give the first attached group the smallest number 4. Name and locate each group Learning Check Alk4 A. CH3 CH3 CHCH2CH CH3 B. CH3 CH3 CH3 CH3CH2CHCH2CH CH3 CH2CH3 Solution Alk4 A. CH3 CH3 CH3CHCH2CHCH3 B. CH3 2,4-dimethylpentane CH3 CH3CH2CHCH2CHCH2CH3 CH3 3,3,5-trimethylheptane Learning Check Alk5 Write a condensed structure for A. 3,4-dimethylheptane B. 2,2-dimethyloctane Solution Alk5 A. 3,4-dimethylheptane CH3 CH3CH2CHCHCH2CH2CH3 CH3 B. 2,2-dimethyloctane CH3 CH3CCH2CH2CH2CH2CH2CH3 CH3 Isomers Same molecular formula Same number and types of atoms Different arrangement of atoms Butane structures n-butane methylpropane Learning Check Alk6 Write 3 isomers of C5H12 and name each. Solution Alk6 CH3CH2CH2CH2CH3 pentane or n-pentane CH3 CH3CHCH2CH3 2-methylbutane CH3 CH3CCH3 CH3 2,2-dimethylpropane Cyclic Alkanes Cyclopropane structure Cyclohexane structure Figure 22.11: The structure of benzene Naming Cycloalkanes with Side Groups Number of side groups Naming One Side group name goes in front of the cycloalkane name. Two Number the ring in the direction that gives the lowest numbers to the side groups. Cycloalkanes with Side Groups CH3 methylcyclopentane CH3 CH3 1,2-dimethylcyclopentane CH3 CH3 1,2,4-trimethylcyclohexane CH3 Learning Check Alk8 Name the following cyclic alkanes CH 3 CH 3 CH 3 CH 3 CH3 Solution Alk8 CH3 1,2-dimethylcyclobutane CH3 CH3 methylcyclopentane CH3 1,3-dimethylcyclohexane CH3 Saturated and Unsaturated Compounds Saturated compounds (alkanes) have the maximum number of hydrogen atoms attached to each carbon atom Unsaturated compounds have fewer hydrogen atoms attached to the carbon chain than alkanes Unsaturated compounds contain double or triple bonds Alkenes Carbon-carbon double bonds Names end in -ene H2C=CH2 ethene (ethylene) H2C=CH-CH3 propene (propylene) cyclohexene Alkynes Carbon-carbon triple bonds Names end in -yne HCCH ethyne(acetylene) HCC-CH3 propyne Naming Alkenes and Alkynes When the carbon chain has 4 or more C atoms, number the chain to give the lowest number to the double or triple bond. 1 2 3 4 CH2=CHCH2CH3 1-butene CH3CH=CHCH3 2-butene CH3C CCH3 2-butyne Learning Check HA3 Write the IUPAC name for each of the following unsaturated compounds: A. CH3CH2CCCH3 CH3 CH3 B. CH3C=CHCH3 C. Solutions HA3 Write the IUPAC name for each of the following unsaturated compounds: A. CH3CH2CH=CHCH3 2-pentyne CH3 CH3 B. CH3C=CHCH3 2-methyl-2-butene C. 3-methylcyclopentene Calling names • ALKANES • ALKENES • ALKYNES • CYCLO• ALKYL- Hydrogenation Adds a hydrogen atom to each carbon atom of a double bond H H + H–C=C–H H H Ni H–C–C–H H H H H ethene ethane Products of Hydrogenation Adding H2 to vegetable oils produces compounds with higher melting points Margarines Soft margarines Shortenings (solid) Figure 22.11: The structure of benzene Figure 22.12: Some selected substituted benzenes and their names Compounds containing aromatic rings are often used in dyes, such as these for sale in a market in Nepal Source: Getty Images Bonding in ethane Bonding in ethylene Bonding in acytylene Cis and Trans Isomers Double bond is fixed Cis/trans Isomers are possible CH3 CH3 CH = CH cis CH3 CH = CH trans CH3 Vision begins with a light induced cis-trans isomerization reaction of retinal isomers • Structural – chain butane methyl propane • Structural - position 2methylhexane 3methylhexane • Structural – function • Stereo - geometrical • Stereo - optical cis trans STEREO-ISOMERS Non-Superimposable Mirror Images COOH H HO CH 3 COOH HO C H COOH H CH3 C CH3 lactic acid OH Electromagnetic Radiation (b) Figure 20.13: Unpolarized light consists of waves vibrating in many different planes Figure 20.14: Rotation of the plane of polarized light by an optically active substance. Optical Isomer and Interaction with Light Enatiomers rotate the plane of polarized light. Dextrorotatory- “d” isomer Complex which rotates plane of polarized light to the right. Levorotatory- “l” isomer Complex which rotates plane of polarized light to the left. Chiral molecules are optically active because effect on light Polarizing sun glasses reduce glare of polarized reflections from surfaces