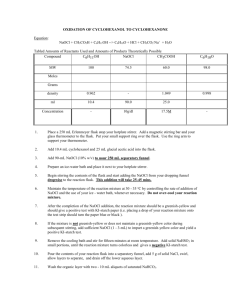
Concentration: 0.5% and 5.25%
advertisement
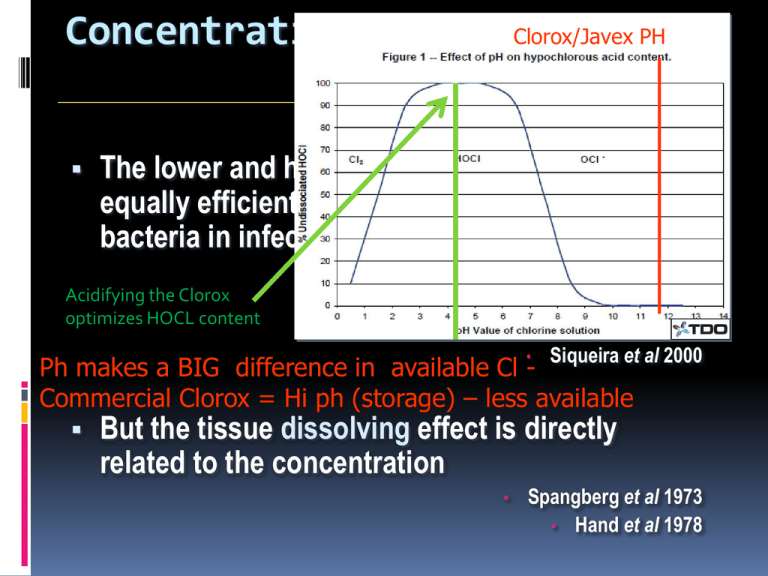
Concentration: 0.5% and 5.25% Clorox/Javex PH The lower and higher concentrations are equally efficient in reducing the number of bacteria in infected RC Spangberg et al 1973 • Cveck et al 1976b Why is this? • Bystrom and Sundqvist 1985 • Siqueira et al 2000 • Acidifying the Clorox optimizes HOCL content Ph makes a BIG difference in available Cl Commercial Clorox = Hi ph (storage) – less available But the tissue dissolving effect is directly related to the concentration • Spangberg et al 1973 • Hand et al 1978 How Do We compensate ? 1. Use regular NaOCl for C&S - solvency 2. Use “modified” formula for final bacteriocidal rinse. Modified NaOCl 1. Lowers the Ph – increases available Cl- (HOCL) 2. Lowers TOXICITY Formula 50 ml distilled water 0.5 ml NaOCl (Clorox) 0.5 ml Acetic acid (White Vinegar) Mixed fresh for every treatment - 2 hr shelf life