Antimicrobials 101
advertisement

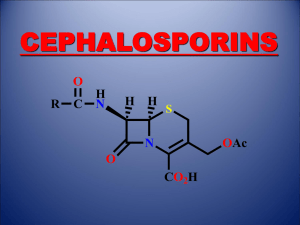
Antibiotics 101 Puja Van Epps 1/20/14 Beta-lactams Core PCN structure Core Cephalosporin structure Beta-lactams Beta-lactamases are enzymes produced by some bacteria that provide resistance against beta lactams through hydrolysis of the β-lactam ring Natural Penicillins Bicillin L-A (Penicillin G benzathine) – IM only Penicillin G (IV) Penicillin V = PO Natural Penicillins- Spectra Groups Gram Positive Gram Negative Anaerobes Other Important Organisms Group A/B/C/G strep S. pneumoniae* viridans streptococci gr.*, Strep milleri* Enterococcus (feacalis>faecium) Neisseria meningitidis* Pasteuralla multocida Haemophilus ducreyi Actinomyces Clostridial sp. Peptostreptococcus Fusobacterium Treponema pallidum Natural Penicillins Bicillin: Primary, secondary, latent and late latent syphillis PCN G: Neurosyphillis; systemic infection due to susceptible bacteria (Streptococci) PCN V: Group A strep pharyngitis Anti-staphylococcal Penicillins Nafcillin, oxacillin, methicillin, dicloxacillin (PO) Penicillinase is a specific type of β-lactamase, showing specificity for Penicillins First β-lactamase to be identified; PCN R in S. aureus Major Uses: Methicillin-susceptible S. aureus or Coagulase Negative Staph; PCN-susceptible strains of Streptococci No gram negative activity Aminopenicillins Ampicillin/amoxicillin; Augmentin (AmoxClav); Unasyn (Amp-Sulbactam) Amp/amox – Great for susceptible streps and enterococcus; very limited GN activity; cover anaerobes Addition of Clavulanate or Sulbactam enhances Gram negative activity No activity against MSSA without the betalactamase inhibitor. Aminopenicillins Important holes in coverage Pseudomonas sp. Atypical gram negatives – mycoplasma pneumoniae, chlamydia pneumoniae, legionella sp. Enterobacter sp. If susceptible Ampicillin is the DOC for Enterococcus and Listeria Anti-Pseudomonal Penicillins Ticarcillin, Ticar-Clav, Piperacillin, Pip-Tazo Generally good gram positive, gram negative and anaerobic coverage Ticarcillin and Piperacillin without their beta-lactamase inhibitor DO NOT cover MSSA Important holes in coverage: MRSA (ESBL+, KPC+, or other resistant GN) Stenotrophomonas maltophilia – Ticar-Clav is second line, Pip/Tazo does not cover. Cephalosporins 5 generations, increasing gram negative coverage with each generation First Generation Cephalosporins Cefadroxil, Cephalexin (PO) Cefazolin (IV) Gram Positive Group A, B, C, G Strep Strep pneumo Viridans strep MSSA Gram Negative E. coli, Klebsiella sp., Proteus mirabilis Anaerobes No activity First Generation Cephalosporins Important holes in coverage – MRSA, Enterococcus, Pseudomonas, anerobes Second Generation Cephalosporins Cefuroxime (IV, PO), Cefotetan (IV), Cefoxitin (IV) In addition to the coverage of 1st generation - H. influenzae, M. catarrhalis, Neisseria sp., and anearobic coverage (variable) Important holes in coverage: - MRSA, Enterococcus, Pseudomonas Third Generation Cephalosporins Ceftriaxone, Cefotaxime, Ceftazadime (IV) Cefixime, Cefdinir (PO) In general less active against gram-positive aerobes than previous generations, but have greater activity against gramnegatives Cefotaxime and Ceftriaxone have the best gram + coverage in the group Only Ceftazadime covers Pseudomonas Third Generation Cephalosporins Major holes in coverage – - Enterococcus, MRSA, Pseudomonas (except Ceftazidime), +/- Acinetobacter, Listeria Ceftazidime crosses BBB, Ceftriaxone in inflamed meninges Fourth Generation Cephalosporins Cefepime (IV) gram-positives: similar to first generation gram-negatives: broad, including Pseudomonas Major holes: MRSA, poor anaerobic coverage, listeria Crosses BBB Fifth Generation Cephalosporin Ceftaroline (IV) Major advantage: - MRSA Major holes in coverage: - Pseudomonas, enterococcus and anaerobes CAP, SSTI Cephalosporin Review Antipseudomonal – Ceftazadime and Cefepime Anti-MRSA – Ceftaroline Anti-Enterococcal – None (Ceftaroline has in-vitro activity against E. faecalis) Enterobacter sp. can develop resistance to cephalosporins during treatment, therefore not the treatment of choice Carbapenems Ertapenem, Doripenem, Imipenem, Meropenem Broadest spectrum of activity Have activity against gram-positive and gramnegative aerobes and anaerobes Bacteria not covered by carbapenems include MRSA, VRE, MR coagulase-negative staph Additional ertapenem exceptions: Pseudomonas, Acinetobacter, Enterococcus Carbapenems Major holes in coverage: - Atypicals (Legionella, Mycoplasma) , MRSA, VRE, Stenotrophomonas maltophilia, KPC+ Ertapenem does not cover: - Pseudomonas, Acinetobacter, Enterococcus Monobactam Aztreonam: binds preferentially to PBP 3 of gram-negative aerobes No gram positive or anaerobic activity Major uses – Hospital acquired infections in patients with anaphylaxis to any beta lactams (does not have cross reactivity) Important gram neg holes: Acinetobacter, ESBL+, KPC+ Fluoroquinolones Ciprofloxacin, Levofloxacin, Moxifloxacin Broad spectrum of activity, excellent bioavailability, tissue penetration Cipro has poor gram + coverage Disadvantages: resistance, expense, C diff Advantages: Atypical coverage, Antipseudomonal (Cipro, Levo) Aminoglycosides Gentamicin, Tobramycin, Amikacin inhibit protein synthesis by irreversibly binding to 30S ribosome, bactericidal For gram + use in combination with cell wall agents Broad spectrum gram neg coverage including Pseudomonas and Acinetobacter Also have mycobacterial coverage Aminoglycosides – adverse effects Nephrotoxicity – Nonoligouric renal failure from damage to the proximal tubules – Underlying CKD, Age, other nephrotixins, duration, high troughs Ototoxicity – 8th cranial nerve damage - vestibular and auditory toxicity; irreversible – Related to duration of therapy (>2wks) Macrolides Clarithromycin, Erythromycin, Azithromycin Inhibit protein synthesis by reversibly binding to the 50s ribosomal unit Macrolides Gram-Positive Aerobes – Clarithro>Erythro>Azithro Gram-Negative Aerobes – Azithro>Clarithro>Erythro No activity against any Enterobacteriaceae or Pseudomonas Anaerobes – activity against upper airway anaerobes Atypical Bacteria – Excellent Also cover – Mycobacterium avium complex, Campylobacter, Borrelia, Bordetella, Brucella. Anti-MRSA drugs Vancomycin Inhibits synthesis and assembly of the second stage of peptidoglycan polymers Gram-positive bacteria: excellent coverage Major uses: MRSA, MSSA (in PCN all), PCN R streptococci No activity against gram-negatives or anaerobes If MIC to Vancomycin in MRSA is ≥ 2, Do not use Vancomycin Red-Man Syndrome – flushing, pruritus, rash – related to rate of infusion – resolves spontaneously – may lengthen infusion NOT AN ALLERGY Daptomycin Lipopeptide; binds to components of the cell membrane and causes rapid depolarization, inhibiting intracellular synthesis of DNA, RNA, and protein Major uses - SAB, Right-sided IE caused by S. aureus, VRE Indicated for SSTI, R sided IE Do not use for lung infections including MRSA PNA – pulmonary surfactant inhibits Daptomycin Linezolid Binds to the 50S ribosomal subunit near the surface interface of 30S subunit – causes inhibition of 70S initiation complex which inhibits protein synthesis Active against wide range of Gram + bacteria, limited to no Gram negative or anearobic activity Major uses – MRSA, VRE. Major problem thrombocytopenia with prolonged use (>2wks), bacteriostatic (cidal against Enterococcus) Tigecyline Binds to the 30S ribosomal subunit of susceptible bacteria, inhibiting protein synthesis. Broad spectrum of activity including – - MRSA, VRE, gram negatives (including resistant GN) Major holesThe 3 P’s – Pseudomonas, Proteus and doesn’t get in the urine Indicated for complicated SSTI, intra-abdominal infections, CAP Major problems: GI issues, and shown to have increased mortality in serious infections – monotherapy only as a last resort. Clindamycin Inhibits protein synthesis by binding exclusively to the 50S ribosomal subunit Major uses - MRSA (some isolates), anaerobic coverage Clindamycin A positive D test indicates the presence of macrolide-inducible resistance to clindamycin produced by an inducible methylase that alters the common ribosomal binding site for macrolides, clindamycin Tetracylines Doxycyline, Minocyline Good gram pos, neg and anaerobic coverage Major uses MRSA, anti-malarial prophylaxis, rickettsial infections, Borrelia burgdorferi Trimethoprim, TMX-Sulfa Inhibit various steps within the folic acid biosynthetic pathway Good gram pos and gram neg coverage (CA-MRSA) Important uses: Pneumocystis, Stenotrophomonas maltophilia, Nocardia Major holes Pseudomonas, anaerobes