Chapter 12 - Richsingiser.com
advertisement

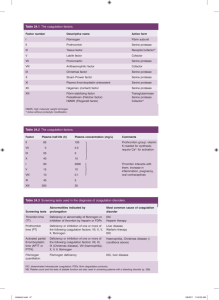
Fundamentals of Biochemistry Third Edition Donald Voet • Judith G. Voet • Charlotte W. Pratt Chapter 11 Enzymatic Catalysis Copyright © 2008 by John Wiley & Sons, Inc. Section 4 – Lysozyme NAG = N-acetylglucosamine NAM = N-acetylmuramic acid Non-polar environment Glu – pKR = much higher than 4.07 Asp – normal pKR = 3.09 Transition state analog Covalent catalysis Section 5 – Serine Proteases • Highly reactive serine • Many digestive enzymes use this mechanism • Inhibiting serine proteases can be very toxic Identifying the active site residues Serine was identified by chemical labeling His was identified by affinity labeling Trypsin with leupeptin inhibitor Chymotrypsin Elastase also have similar Structures Asp is very important Catalytic triad Asp, His, Ser Substrate Specificity Figure 11-30a Figure 11-30b Zymogens – inactive enzyme precursors Fundamentals of Biochemistry Third Edition Donald Voet • Judith G. Voet • Charlotte W. Pratt Chapter 12 Enzyme Kinetics, Inhibition, and Control Copyright © 2008 by John Wiley & Sons, Inc. Section 3 – Control of Enzyme Activity Allosteric regulation T-state R-state Figure 12-13 Covalent Modifications Phosphorylation is most common Ubiquitination important for protein degradation Glycogen Phosphorylase Penicillin • Penicillin – 1928 Alexander Fleming • Disrupts synthesis of peptidoglycans by transpeptidase – Used for bacterial cell walls • Lactam ring is highly reactive Types of Penicillin First, injection only Acid stable, orally Acid stable, orally Most widely used Mechanism of Penicillin Bacterial Evolution Bacterial now produce an enzyme lactamase Inactivates penicillin Human response Create inhibitor of β-lactamase Clavulanic acid Augmentin – combination of both amoxicillin and clavulanic acid New bateria resistant to both The battle continues….. HIV • Retrovirus – RNA genome • Make a polyprotein consisting of 3 proteins – Reverse transcriptase, integrase, protease – Drugs for all enzymes – Drugs for attachment Protease mechanisms • • • • Serine proteases (chymotrypsin) Cysteine proteases Aspartyl proteases (HIV protease) Metalloproteases HIV protease mechanism HIV protease inhibitors OH – mimics stable intermediate, benzene ring mimics hydrophobic aa Other HIV drug targets • Integrase inhibitors – FDA approved in 2007 (raltegravir) – Inhibits strand transfer of viral DNA into host genome • Reverse Transcriptase Inhibitors – AZT 1987 – Nucleoside inhibitors (terminate DNA chain) – Non-nucleoside inhibitors (irreveribily bind to RT) • Coming soon… – Viral fusion, maturation HIV T-cell – green HIV – red