Interest and
Outside
Activity
Reporting
Form
Released July 16, 2012
Overview
• Interest and Outside Activity Reporting Form
• Routing
• Changes to Public Health Service conflict of
interest regulation
• Sample Interest and Outside Activity
Reporting Form
New form incorporates
• Reporting of outside professional activities
– Conflict of Commitment
– Reviewed by respective Vice Chancellor’s
office (see contact list in folders)
• Additional Public Health Service disclosure
and training requirements
Who uses this form?
• Sponsored research investigators
• Full time university employees and faculty
engaged in outside employment or other
professional activities (except office/service
personnel)
When should the form be
completed?
• Before engaging in outside professional
activities; OR
• Before receiving funding for sponsored
research; OR
• Before submitting an application for research
sponsored by the Public Health Service
How often do I need to update it?
• Annually
• Within 30 days of any changes in financial
interests
• Before engaging in a new outside
professional activity
What financial interests should I
disclose?
DO DISCLOSE
• Interests related to your
institutional
responsibilities, including:
– Income from outside UNL
– Equity
– Intellectual property rights
and interests
• International payments
• If PHS-funded: some
sponsored or reimbursed
travel
DO NOT DISCLOSE
• Payments from UNL
• Income from seminars,
lectures, or teaching
engagements sponsored
by:
– U.S. Fed, State, or local
government
– U.S. institution of higher
education or affiliated
hospital, medical center, or
research institute
Where does the form go?
• Routed to:
– Your department chair/director
– The appropriate dean
• Administrative Review:
– Vice Chancellor reviewer (NEW)
– Research Compliance Services
These individuals may request revisions or
acknowledge the form.
And then?
APPROVAL
• You will receive an
approval notification
email
FURTHER REVIEW
• Conflict of Interest in
Research Committee
• Board of Regents
• You’re done for one year
or until your
circumstances change!
Conflict of Interest Regulations Changes for
PUBLIC HEALTH SERVICE
GRANTS
PHS Investigators
Investigator
• Investigator definition
– The project director or principal
Investigator and any other person,
regardless of title or position, who is
responsible for the design, conduct, or
reporting of research funded by the
PHS, or proposed for such funding, which
may include, for example, collaborators or
consultants
PHS Investigators
Timeframe
• All investigators complete IOARF by the time
of submitting proposals
PHS Investigators
Conflict of Interest Training
•
•
•
•
•
Online
Provided by National Institutes of Health
Maximum one hour
Completion certificate when finished
Upload that to your IOARF
Valid for four years
PHS Investigators
Sponsored or Reimbursed Travel
• When to disclose
– No later than 30 days after the travel
– Encouraged to disclose any upcoming
sponsored/reimbursed travel you know
about during annual disclosure
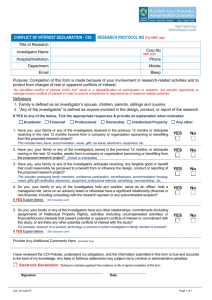
What does the form look like?
• Let’s take a look:
– nugrant.unl.edu
Questions?
Maria Funk
Conflict of Interest Coordinator
Office of Research Responsibility
209 Alexander West
University of Nebraska – Lincoln
(402)472-1837
mfunk2@unl.edu
©2007 The Board of Regents of the University of Nebraska. All rights reserved.