PPT
advertisement

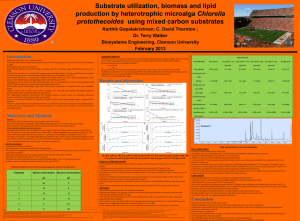
Elucidating the xylose metabolising properties of Scheffersomyces stipitis using a genome scale metabolic model Kevin Correia, Goutham Vemuri, Radhakrishnan Mahadevan Pathway Tools Conference, Menlo Park, CA March 6th, 2013 Overview • Challenges with lignocellulose fermentation • Xylose metabolism in yeast • Nature’s lignocellulose metabolizer: Scheffersomyces stipitis • S. stipitis genome-scale metabolic models • Exploring xylitol production mechanisms • Conclusions and Future Work Lignocellulose fermentation • Lignocellulose feedstocks offer a sustainable source of biomass for biofuels and biochemicals • They contain a variety of fermentable sugars: glucose, xylose, arabinose • Xylose content can range from 10-30% of the biomass in softwood, hardwood, and herbaceous agriculture residue Pentose fermentation in S. cerevisiae • Wild-type Saccharomyces cerevisiae cannot efficiently ferment pentose sugars: xylose and arabinose • S. cerevisiae has been genetically engineered to metabolise xylose via: – Yeast oxidoreductive pathway (Scheffersomyces stipitis) – Bacterial isomerase pathway (Thermus thermophilus) • Xylitol accumulates as a by-product with the yeast pathway due to a cofactor imbalance Pentose fermentation in yeasts • Two enzymes exists in yeast for xylose reductase: – NADPH-dependent xylose reductase (Candida utilus) – NADPH/NADH-dependent xylose reductase (Scheffersomyces stipitis, Pachysolen tannophilus) • C. utilus has excessive xylitol production • P. tannophilus produces 13-30% xylitol under oxygen limiting conditions • This suggests other redox balancing mechanisms exist in S. stipitis Scheffersomyces stipitis • Found in the gut of wood digesting beetles • Can ferment all major components of lignocellulos biomass: glucose, mannose, xylose, arabinose, rhamnose, cellobiose • 48% ethanol yield from xylose • Little to no xylitol production Scheffersomyces stipitis: genome scale metabolic models • iBB814: Balaji Balagurunathan et al. Reconstruction and analysis of a genome-scale metabolic model for Scheffersomyces stipitis. Microbial Cell Factories 2012, 11:27 • iSS884: Caspeta et al. Genome-scale metabolic reconstructions of Pichia stipitis and Pichia pastoris and in silico evaluation of their potentials. BMC Systems Biology 2012, 6:24 • iTL885: Liu et al. A constraint-based model of Scheffersomyces stipitis for improved ethanol production. Biotechnology for Biofuels 2012, 5:7 iBB814: Xylose reductase study iBB814: Xylose reductase study In silico production of xylitol • Balaguruthan, Caspeta and Liu show that xylitol is not a fermentation byproduct in their models, but fail to explore metabolic mechanisms • Simulations in our study show that arabinitol is a byproduct during ethanol fermentation, and xylitol if FVA is used Jeppsson et al. Appl Environ Microbiol. 1995 July; 61(7): 2596–2600. Study objectives • Develop a comprehensive S. stipitis model by reviewing the published models and literature • Run batch and chemostat experiments to fine-tune model parameters; analyse gene expression • Evaluate metabolic mechanisms that lead to xylitol production • Overlay chemostat and gene expression data over the metabolic model to gain insight into regulation in S. stipitis metabolism Proposed mechanisms leading to reduced xylitol production 1. 2. 3. 4. Xylose reductase cofactor specificity Crabtree effect and robustness analysis Alternative oxidase Suboptimal growth Xylitol yield sensitivity to aeration and XR cofactor specificity • Xylitol yield and the Crabtree effect • Crabtree positive yeasts have high uptake rates of substrate and low uptake of oxygen • Crabtree negative yeasts have lower substrate uptake rates and metabolism is sensitive to oxygen uptake Redox balancing with alternative oxidase Joseph-Horne et al. Biochim Biophys Acta. 2001 Apr 2;1504(2-3):17995. Xylitol yield sensitivity to AOX Ethanol yield sensitivity to AOX Xylitol yield and suboptimal growth • An alternative mechanism to account for low xylitol yields in S. stipitis is suboptimal growth • S. stiptis often has lower growth rate in microaerobic conditions when grown on xylose, relative to glucose Succinate bypass • Jeffries (2009) proposed a succinate bypass that allows S. stipitis to convert NADH to NADPH NADPH production envelope • Simulations show that the bypass leads to suboptimal growth compared to NADH kinase Conclusions • We compiled a comprehensive S. stipitis genome scale model from published and unpublished models • We evaluated metabolic mechanisms leading to xylitol production – Cofactor specificity, suboptimal growth and oxygen sensitive metabolism have a greater sensitivity to xylitol yield than alternative oxidase Next steps • Integrate chemostat results, metabolic model, and gene expression • Perform additional experiments in different conditions to explore xylitol production in S. stipitis Acknowledgments • Dr. Radhakrishnan Mahadeven, University of Toronto • Peter Y. Li, University of Toronto • Dr. Goutham Vemuri, BioAmber • Xin Wen, University of Guelph • Dr. Hung Lee, University of Guelph • Bioconversion Network