13. - iannonechem.com

Application of
Electrochemical Cells
Lesson 12
Banana Watch
Application of Electrochemical Cells
1.
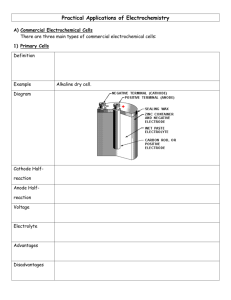
Zn/C or LeClanche Cell
Anode:
Anode Reaction:
Cathode:
Cathode Reaction:
Electrolyte:
Inexpensive
Zn
Zn
→ Zn 2+ + 2e -
C
Mn 4+ +1e → Mn +3
NH
4
Cl and MnO
2
Not rechargeable Short life
Application of Electrochemical Cell
2.
The Alkaline Cell
Anode:
Anode Reaction:
Cathode:
Cathode Reaction:
Electrolyte:
More expensive
Zn
Zn
→ Zn 2+
C
Mn 4+ +1e → Mn 3+
KOH and MnO
2
Not rechargeable
+ 2e -
Longer life
Application of Electrochemical Cells
3.
The Lead Acid Battery (Automobile)
Anode:
Anode Reaction:
Pb
Pb → Pb 2+ + 2e -
Cathode: PbO
2
Cathode Reaction: PbO
2
+ HSO
4
-
Electrolyte: H
2
SO
4
+ 3H + + 2e → PbSO
4
+ 2H
2
Rechargeable Long life Large current
Application of Electrochemical Cells
4.
The Fuel Cell
Overall Reaction:
Expensive
H
2
+ ½O
2
→ H
2
O + energy
Requires fuel Environmentally friendly
Nickel Cadmium
Rechargable
Cordless Phones
Nickel Metal Hydride
Rechargable
Lithium
Rechargable
Cameras
Laptops
Corrosion of Iron
Corrosion is oxidation : Fe
(s)
→
Fe 2+ + 2e -
Rust is initially Fe(OH)
2 which dries to become Fe
2
O
3
.
There are three requirements for the corrosion of iron.
Iron
Water
Oxygen
Corrosion is spontaneous or an electrochemical cell .
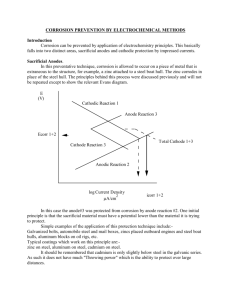
Corrosion of Iron
The anode reaction is the oxidation of Fe
The cathode reaction is the reduction of O
2 and H
2
O outer circle of drop
Cations to cathode and anions to anode
Electrons flow from the anode to the cathode
Anode Fe
(s)
→ Fe 2+ + 2e -
1
Cathode
/
2
O
2
+ H
2
O + 2e → 2OH -
Water Drop
Fe
OH -
Fe(OH)
2(s) e -
Fe 2+
Fe
Fe 2+ e -
Fe(OH)
2(s)
OH -
Fe
Rustlow solubility
Iron Surface
Methods of Preventing Corrosion
Protective Coatings
Paint
Grease
Electroplating
Plastic
Cathodic Protection
Remember corrosion is oxidation
Make iron the cathode of a cell, which is the site of reduction , oxidation cannot occur.
Fe Nail in Water
Attach a piece of Zn or Mg All lower than Fe
.
-
Cathode or Reduction
Zn Anode
-
-
-
-
-
-
Zn → Zn 2+ + 2e -
Sacrificial anode
Electrons flow from anode to cathode to protect the Fe
Mg bracelets on a ship
Cathodic Protection
Remember corrosion is oxidation Make iron the cathode of a cell, which is the site of reduction , oxidation cannot occur.
+
e -
-
Cathode No oxidation!
Scrap iron anode Fe
(s)
-
→ Fe 2+ + 2e -
-
50 km Fe pipe How do we protect it from corrosion?
Coat with plastic
Make it the cathode or negative side of an electrolytic cell
Make Fe the negative side of an electrolytic cell
Cathodic Protection- red and H
2 bubbles
Unprotected nail- blue indicating Fe 2+
Fe and Cu- not a good idea
Add a piece of Zn to the Fe
Cathodic Protection- red and H
2 bubbles
Solution is NaCl, K
3
Fe(CN)
6
, and phenolphathalein. Corrosion of Fe will show as blue- reaction with K
3
Fe(CN)
6
. Cathodic protection will reduce water and show as bubbles and pink.
BC Fast Ferry
The Aluminum hull is protected by an electrolytic cell
When it was first put to sea this system was not in operation.
The Paint peeled off requiring a new multimillion dollar paint job.