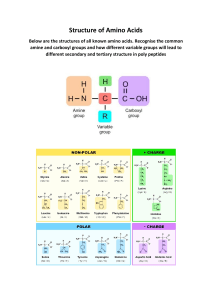
Biochemistry: Proteins STUDY WELL! Definition: PROTEINS § General definition: are naturally occurring, unbranched polymer in which the monomer units are amino acids. § Structural definition: Is a peptide in which at least 40 amino acid residues are present. Examples of PROTEINS Ø Ø Ø Ø Ø HORMONES = messengers ENZYMES = speed up reactions CELL RECEPTORS = “antinnae” ANTIBODIES = fight foreign invaders MEMBRANE CHANNELS = allowing specific molecules to enter or leave a cell QUESTION What are the fundamental components that make up proteins? AMINO ACID: the building block of proteins • -amino acid- is an amino acid in which the amino group and the carboxyl group are attached to the -carbon atom. • Except: Proline • has a structural feature not found in any other standard amino acid. • side chain- propyl group is bonded to both the a-carbon atom and the amino nitrogen atom,giving a cyclic side chain CLASSIFICATION OF AMINO ACIDS ACCORDING TO SIDE CHAIN POLARITY CLASSIFICATION OF AMINO ACIDS Nonpolar amino acid • is an amino acid that contains one amino group, one carboxyl group, and a nonpolar side chain. G,A,V,L,I,P,F,M,W Polar neutral • is an amino acid that contains one amino group, one carboxyl group, and a side chain that is polar but neutral. S,C,N,T,Q,Y Polar basic • is an amino acid that contains two amino groups and one carboxyl group, the second amino group being part of the side chain. H,K,R Polar acidic • is an amino acid that contains one amino group and two carboxyl groups, the second carboxyl group being part of the side chain. D,E NAMING AMINO ACIDS GIV-LAPMWF CNS-TQY NON POLAR AMINO ACID POLAR NEUTRAL HKR POLAR BASIC DE POLAR ACIDIC CLASSIFICATION OF AMINO ACIDS : AMINO ACIDS (9) HYDROPHOBIC SIDE CHAIN FUNCTIONAL GROUP G H group A Aliphatic R group (methyl) V Aliphatic R group (isopropyl) I Aliphatic R group (isobutyl group) L P Aliphatic R group (additional methylene group) Aliphatic R group (propyl) F Aromatic Ring (Phenyl group/ alanine w/ phenyl substituent on the - carbon) M Sulfur group (thioether) W Aromatic Ring (indole ring -5 membered nitrogen ring fused w/ benzene ring) CLASSIFICATION OF AMINO ACIDS : AMIN O ACIDS (6) Uncharged, Non-Ionic Side Chains C due to SH or Thiol group S T N Q Y FUNCTIONAL GROUP due to OH group (Hydroxyl group) due to OH group (Hydroxyl group) due to Amide groups (amide w/ one methylene/ amide of aspartic acid) due to Amide groups (amide w/ 2 methylene) due to OH group (Aromatic Hydroxyl group) CLASSIFICATION OF AMINO ACIDS : AMIN O ACIDS (3) Positively charged K (-amino group/ butyl amine/ alanine w/ propylamine substituent on the -carbon) H R FUNCTIONAL GROUP (imidazole ring/ 5 membered heterocyclic ring) (guanidino group) CLASSIFICATION OF AMINO ACIDS : AMINO ACIDS (2) Negatively charged D (4C atom/ one methylene) E FUNCTIONAL GROUP 5C atom/ two methylene) IDENTIFY THE AMINO ACID: I IDENTIFY THE AMINO ACID: T IDENTIFY THE AMINO ACID: D IDENTIFY THE AMINO ACID: W IDENTIFY THE AMINO ACID: F Classification of amino acids ESSENTIAL AMINO ACIDS ▹ is a standard amino acid needed for protein synthesis that must be obtained from dietary sources because the human body cannot synthesize it in adequate amounts from other substances. ▹ R*,H,M,I,L,K,W,T,F,V NON-ESSENTIAL AMINO ACIDS ▹ Synthesized by the body ▹ C,A,N,D,E,Y,S, Q,G,P, USES, FUNCTIONS AND DEFICIENCY OF PROTEINS KWASHIORKOR MARASMUS Inability to grow or gain weight Weight loss Edema or swelling of the hands and feet Dehydration Stomach bulging Stomach shrinkage occurs in people who have a severe protein deficiency. Children who develop kwashiorkor are often older than children who develop marasmus. occurs more often in young children and babies. It leads to dehydration and weight loss. Starvation is a form of this disorder USES, FUNCTIONS AND DEFICIENCY OF AMINO ACIDS AMINO ACIDS USES/ FUNCTIONS DEFICIENCY Trp § Necessary for the synthesis of neurotransmitter serotonin (5hydroxytryptamine). A natural relaxant, helps alleviate insomnia by inducing normal sleep; reduces anxiety and depression. § Can be metabolized to niacin if needed § Used to synthesized melatonin (5-methoxy-N-acetyltryptamine) Pellagra, Tyr § Precursor of dopamine, norepinephrine, epinephrine § Promotes healthy thyroid functioning Hypothyroidism Val, I l e , Leu § enhance energy, increase endurance, and aid in muscle tissue recovery and repair. § lowers elevated blood sugar levels and increases growth hormone production. MSUD (maple syrup urine disease) Lys § precursor for L-carathine which is essential for healthy nervous system function. § for adequate absorption of calcium and bone development in children R e t a r d e d growth USES, FUNCTIONS AND DEFICIENCY OF AMINO ACIDS AMINO ACIDS USES/ FUNCTIONS Met § Is antioxidant. It helps in breakdown of fats and aids in reducing muscle degeneration. Helps lower cholesterol levels by increasing the liver's production of lecithin; reduces liver fat and protects the kidneys. § principle supplier of sulfur, which inactivates free radicals. Is a natural chelating agent for heavy metals and helps detoxify the body of these metals. § Adequate methionine prevents disorders of the hair, skin and nails; Phe § Beneficial for healthy nervous system. It may be useful against depression and suppressing appetite. § Used to produce dopamine and norepinephrine, chemicals that promote alertness, elevate mood, decrease pain, aid in memory and learning, and reduce hunger and appetite. § D-phenylalanine - natural form. May improve rigidity, walking disabilities, speech difficulties and depression associated with Parkinson’s disease. § L-phenylalanine can be converted into L-tyrosine, which is in turn converted into L-DOPA. L-DOPA is a precursor for dopamine, norepinephrine (noradrenalin), and epinephrine (adrenaline) § DL-phenylalanine DEFICIENCY L i v e Deterioration r USES, FUNCTIONS AND DEFICIENCY OF AMINO ACIDS AMINO ACIDS Ala Cys Gln Gly His Thr USES/ FUNCTIONS § Removes toxic substances released from breakdown of muscle protein during intensive exercise § antioxidant (free radical scavenger), and has synergetic effect when taken with other antioxidants such as vitamin E and selenium. § Promotes healthy brain function. It is also necessary for the synthesis of RNA and DNA molecules. § Component of skin and is beneficial for wound healing. It acts as neurotransmitter § Important for the synthesis of red and white blood cells. § It is a precursor for histamine which is good for sexual arousal. Improve blood flow. § It helps promote equilibrium in the central nervous system—aids in balancing state of emotion. USES, FUNCTIONS AND DEFICIENCY OF AMINO ACIDS AMINO ACIDS Asp Pro Arg USES/ FUNCTIONS § Enhances stamina, aids in removal of toxins and ammonia from the body, and beneficial in the synthesis of proteins involved in the immune system. § plays role in intracellular signaling. § plays role in blood vessel relaxation, stimulating and maintaining erection in men, production of ejaculate, and removal of excess ammonia from the body. GLUCOGENIC- glycine, alanine, serine, aspartic acid, asparagine, glutamic acid, glutamine, proline, valine, methionine, cysteine, histidine and arginine KETOGENIC-lysine and leucine BOTH-tryptophan, phenylalanine, tyrosine, isoleucine and threonine Acid-base properties Zwitterion = means double ion Is a molecule that has a positive charge on one atom and a negative charge on another atom but which has no net charge. § Basic solution - NH3+ of the zwitterion loses a proton and negatively charged species is formed. § Acidic solution - the zwitterion accepts a proton (H+) to form a positively charged ion. Guidelines for amino acid form as function of solution pH follows: § Low pH: § High pH: All acid groups are protonated (-COOH). All amino groups are protonated (NH3 +) All acid groups are deprotonated (COO-). All amino groups are deprotonated (-NH2). § Neutral pH: All acid groups are deprotonated (COO-). All amino groups are protonated (NH3+). ISOELECTRIC POINT § the pH at which an amino acid exists primarily in its zwitterion form. At the isoelectric point, almost all amino acid molecules in a solution (more than 99%) are present in their zwitterion form. PEPTIDE FORMATION Peptide bond § a covalent bond (amide bond) between the carboxyl group of one amino acid and the amino group of another amino acid. is an unbranched chain of amino acids, each joined to the next by a peptide bond. TYPES OF PEPTIDE: § dipeptide – a compound containing two amino acids. § Tripeptide – three amino acids joined together in a chain. § Oligopeptide – refer to peptides with 10 to 20 amino acid residues. § Polypeptide – long unbranched chain of amino acids, each joined to the next by a peptide bond. R H2N C H C O N O C R C H N R C C O N – terminal end (left) C – terminal end (right) Backbone (peptide bond & a-carbon –CH group) R- substituent PEPTIDE BOND Peptide Nomenclature IUPAC Rule: 1. The C-terminal amino acid residue keeps its full amino acid name. 2. All of the other amino acid residues have names that end in –yl. The –yl suffix replaces the –ine or ic acid ending of the amino acid name, except for tryptophan (tryptophyl), cysteine (cysteinyl), glutamine (glutaminyl), and asparagine (asparaginyl). 3. The amino acid naming sequence begins at the N-terminal amino acid residue. Biochemically important small peptides • Hormones both produced by the pituitary gland: 1. Oxytocin ▹ regulates uterine contraction and lactation, plays a role in stimulating the flow of milk in a nursing mother. 2.Vasopressin (ADH) ▹ regulates the excretion of water by the kidneys; ▹ Enhances reabsorption of free water ▹ also affects blood pressure. Biochemically important small peptides ▹ Neurotransmitters: Enkephalins – pain killers (pentapeptide):neurotransmitters or neuromodulators at many locations in the brain and spinal cord and are involv ed with pain perception, movement, mood, behavior, and neuroendocrine regulation; they are also found in nerve plexuses and exocrine glands of the gastrointestinal tract. ■Met-enkephalins = Tyr – Gly – Gly – Phe – Met ■Leu- enkephalins = Tyr – Gly – Gly – Phe – Leu Biochemically important small peptides ▹ Antioxidants: Glutathione - regulator of oxidationreduction reaction - antioxidant, protecting cellular contents from oxidizing agents such as peroxides and superoxides (highly reactive forms of oxygen often generated within the cell in response to bacterial invasion) with unusual features. Glutathione - (Glu – Cys - Gly) (tripeptide) Unusual features. The amino acid Glu, an acidic amino acid, is bonded to Cys, through the sidechain carboxyl group rather than through its acarbon carboxyl group. Bonding in proteins 1. Peptide bond – the strongest bond 2. Disulfide bond - is a covalent bond between two sulfur. Results from the oxidation of the –SH (sulfhydryl) groups of two cysteine molecules to form “cystine” 3. Hydrogen Bond - result from the attraction of electronegative atoms in the protein molecule. Weaker than the peptide and disulfide bonds. H-bond: can occur between amino acids with polar R groups. functional groups: -OH -NH2 -COOH -CONH2 1. Ionic bond or salt bridges - formed between groups which are positively and negatively charge. 2. Hydrophobic bond - formed by amino acids like leucine, valine, phenylalanine, tryptophan, and proline which adhere each other forming a “micelle” and which do not mix well with water - result when two nonpolar side chains are close to each other. CLASSIFICATION OF PROTEINS 47 CLASSIFICATION OF PROTEINS Based on the: I. number of peptide chain II. chemical composition III. level of structural organization IV. gross structure V. Function 48 I. Based on the number of peptide chain 1. monomeric protein - is a protein in which only one peptide chain is present. ▹ Example: myoglobin 2. multimeric protein - is a protein in which more than one peptide chain is present. ▹ The peptide chains present in multimeric proteins are called protein subunits. ▹ Example: insulin 49 II. Based on chemical composition I. A. Simple protein is a protein in which only amino acid residues are present. 1. 2. Albumins a. b. c. a. b. Examples: Serum albumin (blood) Lactalbumin (milk) Ovalbumin (eggwhite) Properties: Soluble in water and dilute neutral salt solution Coagulated by heat and precipitated by full saturation with (NH4)2SO4 but not w/ NaCl except with the presence of acid. 50 II. Based on chemical composition B. Globulins 1. Examples: a. Ovoglobulin (eggwhite) b. Edestin (hempseed) c. Legumin (peas) d. Myosinogen (muscles) e. Serum globulin (blood) 2. Properties: a. Soluble in neutral dilute salt solutions but not in water. (Neutral salts refers refer to salts of strong acids and bases as NaCl, MgSO4 and (NH4)2SO4.) b. Coagulated by heat and can be precipitated from their solutions by half saturation with (NH4)2SO4 and complete saturation with NaCl. 51 II. Based on chemical composition C. Glutelins 1. Examples: a. Glutenin (wheat) b. Oryzenin (rice) 2. Properties: a. Soluble in dilute acids and alkalies but insoluble in neutral solvents. C. Prolamines 1. Examples: a. Gliadin (wheat) b. Zein (corn) c. Hordein (barley) 2. Properties: a. Insoluble in ordinary solvent (water, dilute salt solutions, dilute acid and alkalies) but soluble in 70% alcohol at about neutral point. b. Not coagulable by heat. 52 II. Based on chemical composition E. Histones 1. Examples: a. Globin (hemoglobin) b. Thymus histones c. Scobrone of Mackerel 2. Properties: a. Soluble in water, dilute acids and alkalies but not in ammonia. b. Not readily coagulated by heat c. Strongly basic and occur in the tissues in the form of salt combinations with acid substances like the heme of the hemoglobin E. Protamines 1. 2. Examples: a. Salmin (salmon sperm) Properties: a. Contain smaller number of amino acids b. S o l u b l e i n w a t e r a n d dilute acids and alkalies c. Not coagulated by heat 53 II. Based on chemical composition G. Scleroproteins (Albuminoids) 1. Examples: a. Keratin (epidermal tissues) b. Elastins (Ligaments) c. Collagen (hides, bones and cartillage) 2. Properties: a. Insoluble in water and neutral solvents 54 II. Based on chemical composition II. Conjugated protein is a protein that has one or more non-amino acid entities present in its structure in addition to one or more peptide chains. • prosthetic group is a non-amino acid group present in a conjugated protein. These non-amino acid components, which may be organic or inorganic. 55 II. Based on chemical composition Nucleoprotein A. Examples: 1. a. b. c. d. e. Ribosomes (site for protein synthesis in cells) Viruses (self-replicating, infectious complex) Chromatin Products from glandular tissues Germ of grains Properties: 2. a. b. c. d. Prosthetic group: Nucleic acids Combination of histones and protamins with nucleic acids Soluble in dilute solutions of NaCl Precipitated by acidification 56 Based on chemical composition B. Glycoproteins (more proteins less carbohydrates) Examples: 1. a. b. c. d. e. f. Mucin aka mucoproteins – more carbs less proteins (saliva, mucous secretion of the nose) Tendomucoid (tendons) Osseomucoid (bones) gamma globulin (antibody) Interferon (antiviral protection) Simple proteins like globulins and albumins Properties: 2. a. b. c. Prosthetic group: carbohydrates Use: Utilize for lubricating purposes in view of their slimy nature Use: Help in protecting the membranes of the GIT against digestion since they are not digested by the enzymes of GIT. C. Phosphoproteins 1. 2. a. b. a. Examples: Casein (milk) Vitellin (egg yolk) Properties: Prosthetic group: H3PO4 57 Based on chemical composition D. Chromoproteins/ Hemoproteins 1. Examples: a. b. c. d. 2. Hemoglobin (blood carrier of O2 in blood ) Myoglobin (oxygen binder in muscles) Cytochromes Rhodopsin Properties: a. Prosthetic group: hematin or heme unit E. Lipoproteins 1. a. b. c. d. 2. a. b. Examples: Lecithin Cephalin low-density lipoprotein (LDL) – lipid carrier high-density lipoprotein (HDL) – lipid carrier Properties: Prosthetic group: Lipids/ fatty substance Occurrence: Blood serum, brain tissues, cell nuclei, egg yolk and milk 58 Based on chemical composition D. Metalloproteins 1. a. b. 2. a. Examples: iron–ferritin (storage complex for iron) zinc–alcohol dehydrogenase (enzyme in alcohol oxidation) Properties: Prosthetic group: metal ion 59 II. Based on chemical composition iii.Derived proteins. These include substances formed from simple conjugated proteins. A. 1. Primary Protein derivatives – proteins which have undergone slight intramolecular rearrangement through the hydrolytic action of certain physical and chemical agents. - synonymous w/ denatured proteins a. b. Proteans Examples: § § § Myosan from myosin Edestan from edestin Properties Are insoluble substances resulting from the preliminary actions of water, dilute acids or enzymes 60 Based on chemical composition 2. Metaproteins a. Examples: § § b. Acid metaproteins (acid albuminate) A l k a l i metaproteins (alkali albuminate) Properties § § § Are product of further hydrolysis Soluble in weak acids and alkalies neutral salt solution Insoluble in neutral salt solutions 3. Coagulated proteins a. b. Examples: § § § Cooked egg albumin Cooked meat Properties Insoluble products res u l t in g f ro m eit her the action of heat, alcohol, ultraviolet rays o r e v e n s i m p l e mechanical shaking 61 Based on chemical composition B. Secondary Protein derivatives Product of more extensive hydrolysis § Mixtures of fragments of original proteins varying in composition and size § Exhibit certain common properties such as solubility in water and non-coagulability by heat. § Primary proteoses 1. a. Properties § § Soluble in water, precipitated by conc.HNO 3 and by half saturation with (NH4)2SO4 or ZnSO4. Not coagulated by heat 2. Secondary proteoses a. Properties § Precipitated only by complete saturation with (NH 4 ) 2 SO 4 but nit with picric acid and HNO3 . 62 Based on chemical composition 3. Peptones a. Properties § § § Soluble in water Not coagulated by heat Not precipitated by saturation with (NH4)2SO4 but by certain alkaloidal reagents, such as,phosphotingstic and tannic acids. 4. Peptides § § § Are combinations of two or more amino acids, the carboxyl group of one being united with the amino group of the other. Present properties like peptones Ex. di, tri,tera,penta etc. 63 III. Level of structural organization 64 III. Level of structural organization 65 III. Level of structural organization: PRIMARY § Sequence of amino acids in a protein – that is, the order in which the amino acids are connected to each other § Also involves the order of attachment of the amino acids to each other through covalent* peptide bonds. § These bonds are formed between the carboxyl group of one amino acid with the amino group of another. “peptide bond planarity” is the zigzag arrangement 66 Level of structural organization: PRIMARY MYOGLOBIN The primary structure of human myoglobin. This diagram gives only the sequence of the amino acids present and conveys no information about the actual three dimensional shape of the protein. 67 Level of structural organization: PRIMARY Primary Structure of Bovine Insulin (51 AA) First protein to be fully sequenced (by Fred Sanger in 1953). For this, he won his first Nobel Prize (his second was for the Sanger deoxy method of DNA sequencing 68 III. Level of structural organization: SECONDARY § Is the arrangement in space adopted by the backbone portion of a protein. § I s a r e s u l t o f hy drogen b o nd i ng between carbonyl oxygen atom of a peptide linkage and the hydrogen atom of an amino group of another peptide linkage farther along the backbone. Common types of secondary structure 1. 2. Alpha helix (-helix) Beta pleated sheet (-pleated sheet). 69 Level of structural organization: SECONDARY ALPHA HELIX § is a protein secondary structure in which a single protein chain adopts a shape that resembles a coiled spring (helix), with the coil configuration maintained by hydrogen bonds. 70 Level of structural organization: SECONDARY § is a protein secondary structure in which two fully extended protein chain segments in the same or different molecules are held together by hydrogen bonds. § Hydrogen bonds form between oxygen and hydrogen peptide linkage atoms § different parts of a single chain that folds back on itself (intrachain bonds) § Or between atom s in different peptide chains in those proteins that contain more than one chain (interchain bonds). 71 Level of structural organization: SECONDARY l l l Core of many proteins is the sheet Form rigid structures with the H-bond Can be of 2 types l l Anti-parallel – run in an opposite direction of its neighbor (A) Parallel – run in the same direction with longer looping sections between them (B) 72 Level of structural organization: TERTIARY § The overall three-dimensional shape of a protein that results from the interactions between amino acids side chains (R groups) that are widely separated from each other within a peptide chain. Four types of stabilizing interactions contribute to the tertiary structure of a protein: 1. covalent disulfide bonds 2. electrostatic attractions (salt bridges) 3. Hydrogen bonds 4. hydrophobic attractions 73 Level of structural organization: TERTIARY 74 Level of structural organization: QUATERNARY § Is the organization among the various peptide chains in a multimeric protein. § The three-dimensional shape of a protein consisting of two or more independent peptide chains, which results from noncovalent interactions between R groups § For example, the hum a n hem oglobin molecule is a tetramer made up of two alpha and two beta polypeptide chains. § This is also when the protein associates with non-proteic groups. For example, carbohydrates can be added to form a glycoprotein. Bonds: §Electrostatic §H-bond §Hydrophobic 75 IV. GROSS STRUCTURE: ▹ FIBROUS ▹ GLOBULAR ▹ MEMBRANEOUS 76 GROSS STRUCTURE: Fibrous Protein whose molecules have an elongated shape with one dimension much longer than the others. Properties: §Tend to have simple, regular and linear structures. §Largely insoluble in ordinary aqueous media §Molecular weights are high §Functions is for structural and support Fibrous Proteins (insoluble) Occurrence and Functions Keratins found in wool, feathers, hooves, silk, and fingernails Collagen f o u n d i n t e n d o n s , s bone, and other connective tissue Elastins found in blood vessels and ligaments Myosins found in muscle tissue Fibrin found in blood clots 77 GROSS STRUCTURE: § Protein whose molecules have peptide chains that are folded into spherical or globular shapes. § The folding in such that most of amino acids with hydrophobic side chains (nonpolar R groups) are in the interior of the molecules and most of the hydrophilic side chains (polar) are on the outside of the molecule. Properties: § Soluble in aqeous media § Have been crystallized and have definite molecular weights § Can be denatured Globular Proteins (soluble) Occurrence and Functions Insulin regulatory hormone for controlling glucose metabolism Myoglobin involved in oxygen storage in muscles Hemoglob involved in oxygen in transport in blood Transferri n involved in iron transport in blood Immuno globulins involved in immune system responses 78 GROSS STRUCTURE: FIBROUS water-insoluble GLOBULAR Water-soluble usually have a single type several types of secondary of secondary structure structure. generally have structural functions that provide support and external protection, involved in metabolic c h e m i s t r y, p e r f o r m i n g functions such as catalysis, transport, and regulation. Lesser in kind of proteins Greater in kind of proteins M o s t a b u n d a n t i n t h e Less human body Greater mass composition Lesser mass composition 79 GROSS STRUCTURE: MEMBRANEOUS § is a protein that is found associated with a membrane system of a cell. § Soluble in aqueous media § Have been crystallized and have definite molecular weights § Can be denatured 80 V. BASED ON FUNCTIONS CATALYTIC Proteins with the role of biochemical catalyst are called enzymes. • Enzymes participate in almost all of the metabolic reactions that occur in cells. The chemistry of human genetics, is very dependent on the presence of enzymes. DEFENSE also called immunoglobulins or antibodies w/c are central functioning of the body’s immune system • They bind to foreign substances, such as bacteria and viruses, to help combat invasion of the body by foreign particles. - proteins bind to particular small biomolecules and TRANSPORT transport them to other locations in the body and then release the small molecules as needed at the destination location. • Examples: • hemoglobin - carries oxygen from the lungs to other organs and tissues. • Transferrin - which carries iron from the liver to the bone marrow. • High- and low-density lipoproteins - are carriers of cholesterol in the bloodstream 81 BASED ON FUNCTIONS transmit signals to coordinate biochemical processes between different cells, tissues, and organs MESSENGER • insulin • glucagon • Human growth hormone CONTRACTILE are necessary for all forms of movement • Actin and myosin • Sperm can “swim” because of long flagella made up of contractile proteins. confer stiffness and rigidity to otherwise fluid-like biochemical STRUCTURAL systems • Collagen - component of cartilage, • Keratin - gives mechanical strength as well as protective covering to hair, fingernails, feathers, hooves, and some animal shells. 82 BASED ON FUNCTIONS STORAGE These proteins bind (and store) small molecules for future use • During degradation of hemoglobin the iron atoms present are released and become part of ferritin, an iron-storage protein, which saves the iron for use in the biosynthesis of new hemoglobin molecules. • Myoglobin - an oxygen-storage protein present in muscle; the oxygen so stored is a reserve oxygen source for working muscle. REGULATORY often found “embedded” in the exterior surface of cell membranes. • Act as sites at which messenger molecules, including messenger proteins such as insulin, can bind and thereby initiate the effect that the messenger “carries.” • Are often the molecules that bind to enzymes (catalytic proteins), thereby turning them “on” and “off” and thus controlling enzymatic action. TRANSMEMBRANE help control the movement of small molecules and ions through the cell membrane 83 BASED ON FUNCTIONS NUTRIENT These proteins are particularly important in the early stages of life, from embryo to infant. • Casein - milk • Ovalbumin - found in egg white (>50% present) BUFFER These proteins are part of the system by which the acid-base balance within body fluids is maintained • Hemoglobin has a buffering role in addition to being an oxygen carrier. • Transmembrane proteins regulate the movement of ions in and out of cells, ensuring that ion concentrations are those needed for correct acidity/alkalinity. FLUID BALANCE These proteins help maintain fluid balance between blood and surrounding tissue • Albumin and globulin • Found in the capillary beds of the circulatory system. When increased blood pressure generated by a pumping heart forces water and nutrients out of the capillaries, these proteins remain behind (since they are too big to cross cellular membranes). As their concentration increases (due to less fluid being present), osmotic pressure “forces” draw water back into the capillaries, which is necessary for fluid balance to be maintained. DO NOT SHARE THIS FILE! GRAPHICS YOU CAN USE (LOGOS) SEALS COMPONENTS OF ACADEMIC SYSTEM (ICONS) Active Learning Student Success Program IndustryRelevant Curriculum Success Ladder LOCATIONS SCHOOL BUILDINGS COLOR PALETTES PHINMA Education/PTCPHINMA Ed Secondary Colors RGB: 32, 44, 97 CMYK: 100, 93, 30, 25 RGB: 25, 111, 56 CMYK: 87, 27, 95, 14 Araullo University CMYK: 100, 93, 30, 25 CMYK: 16, 25, 100, 0 RGB: 46, 103, 167 CMYK: 85, 50, 0, 0 RGB: 66, 139, 51 CMYK: 70, 10, 100, 12 RGB: 255, 212, 0 CMYK: 0, 10, 100, 0 University of Pangasinan CMYK: 75, 50, 100, 40 CMYK: 0, 15, 100, 40 CMYK: 90, 30, 100, 16 CMYK: 75, 50, 100, 40 CMYK: 0, 25, 100, 0 RGB: 28, 28, 26 CMYK: 0, 0, 0, 100 Southwestern University RGB: 144, 7, 16 RGB: 35, 31, 32 St. Jude College Cagayan de Oro CollegeUniversity of Iloilo CMYK: 0, 9, 89, 18 RGB: 114, 114, 115 CMYK: 0, 0, 0, 60 CMYK: 0, 0, 100, 0 RGB: 36, 124, 61 CMYK: 217, 163, 40