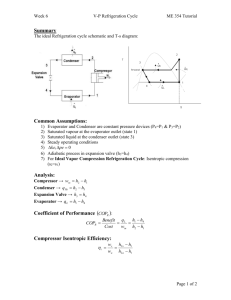
the evaporator inlet, the refrigeration load, the COP of the... 11-20
advertisement
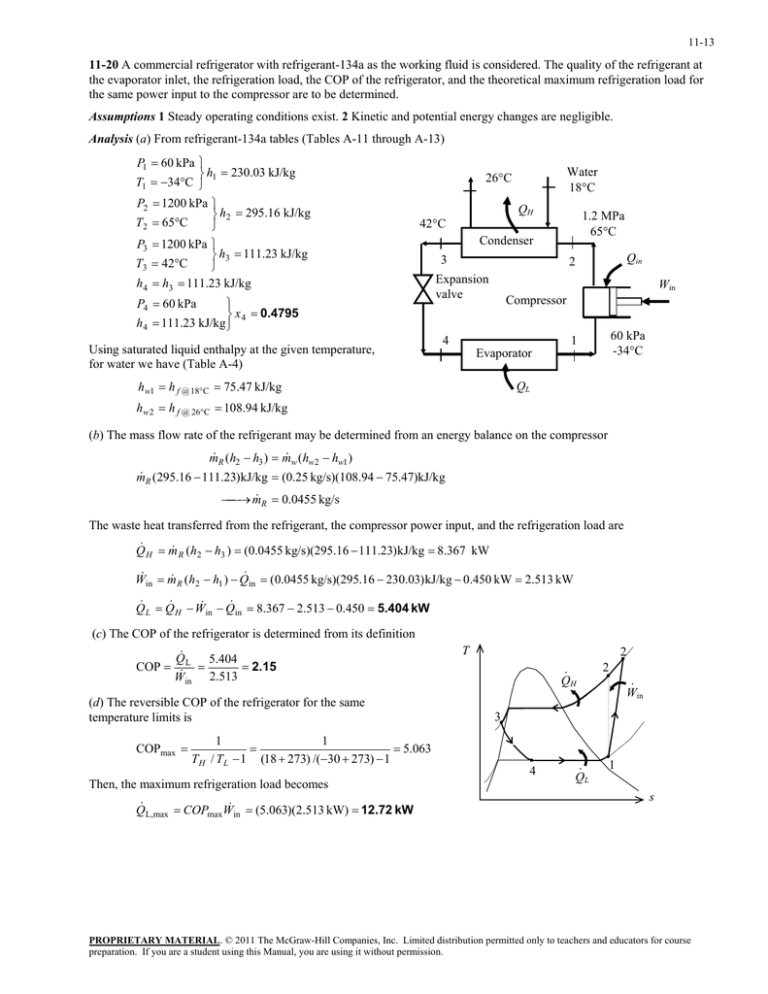
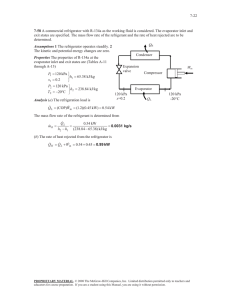
11-13 11-20 A commercial refrigerator with refrigerant-134a as the working fluid is considered. The quality of the refrigerant at the evaporator inlet, the refrigeration load, the COP of the refrigerator, and the theoretical maximum refrigeration load for the same power input to the compressor are to be determined. Assumptions 1 Steady operating conditions exist. 2 Kinetic and potential energy changes are negligible. Analysis (a) From refrigerant-134a tables (Tables A-11 through A-13) P1 60 kPa h1 230.03 kJ/kg T1 34C P2 1200 kPa h2 295.16 kJ/kg T2 65C P3 1200 kPa h3 111.23 kJ/kg T3 42C h4 h3 111.23 kJ/kg Water 18C 26C QH 1.2 MPa 65C 42C P4 60 kPa x 4 0.4795 h4 111.23 kJ/kg Using saturated liquid enthalpy at the given temperature, for water we have (Table A-4) Condenser 3 Qin 2 Expansion valve Win Compressor 4 60 kPa -34C 1 Evaporator hw1 h f @ 18C 75.47 kJ/kg QL hw 2 h f @ 26C 108.94 kJ/kg (b) The mass flow rate of the refrigerant may be determined from an energy balance on the compressor m R (h2 h3 ) m w (hw2 hw1 ) m R (295.16 111.23)kJ/kg (0.25 kg/s)(108.94 75.47)kJ/kg m R 0.0455 kg/s The waste heat transferred from the refrigerant, the compressor power input, and the refrigeration load are Q H m R (h2 h3 ) (0.0455 kg/s)(295.16 111.23)kJ/kg 8.367 kW W in m R ( h2 h1 ) Q in (0.0455 kg/s)(295.16 230.03) kJ/kg 0.450 kW 2.513 kW Q L Q H W in Q in 8.367 2.513 0.450 5.404 kW (c) The COP of the refrigerator is determined from its definition COP Q L 5.404 2.15 W in 2.513 (d) The reversible COP of the refrigerator for the same temperature limits is COPmax 1 1 5.063 T H / T L 1 (18 273) /(30 273) 1 Then, the maximum refrigeration load becomes Q L,max COPmaxW in (5.063)(2.513 kW) 12.72 kW T 2 · QH 2 · Win 3 4 · QL 1 s PROPRIETARY MATERIAL. © 2011 The McGraw-Hill Companies, Inc. Limited distribution permitted only to teachers and educators for course preparation. If you are a student using this Manual, you are using it without permission.