CHEMISTRY TWO Module Alfa PROCESS STANDARDS. (from
advertisement

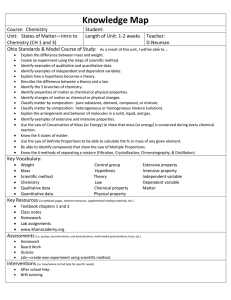
CHEMISTRY TWO Module Alfa PROCESS STANDARDS. (from Priority Academic Student Skills) 1.2. Use appropriate tools with measuring objects and/or events. accuracy and precision when 1.3. Use appropriate SI units when measuring objects and/or events. 2.1. Using observable properties, place an object or event into a classification system. 2.2. Identify the properties by which a classification system is based. 7.6. Communicate results in a variety of ways. CONTENT STANDARDS. (from Priority Academic Student Skills) 1.4. A compound is formed when two or more kinds of atoms bind together chemically. 1.4.a. Atoms interact with one another by transferring or sharing valence electrons. 1.4.b. Valence electrons reactivity of the element. govern the chemical properties and 1.4.c. Each compound has unique chemical and physical properties. 3.1. Matter can be found in four phases. Phase change occurs when heat energy is absorbed or released from the system. OBJECTIVES. This module deals with chemistry on the macroscopic level. Discussions will include the scientific method, matter, and the units of measurement. A1. Identify safe and unsafe laboratory practices. A2. Demonstrate knowledge of safety equipment. A3. Identify sections of a Material Safety Data Sheet (MSDS). A4. Name and describe the three basic steps of the study of chemistry. A5. Describe the steps of the scientific method. A6. Differentiate among hypotheses, theories, and laws. A7. Differentiate between physical and chemical properties. A8. Describe methods for separating mixtures. A9. Describe how elements combine to form compounds. A10. Describe matter. the interconversion among three states of A11. Identify and properly use SI units for quantities. A12. Demonstrate use of scientific notation. VOCABULARY. accuracy chemistry density extensive property homogeneous mixture intensive property Kelvin liter mass microscopic property physical property qualitative scientific method substance volume IN THE TEXT. LABORATORY. chemical property compound element heterogeneous mixture hypothesis International System of Units law macroscopic property matter mixture precision quantitative significant figures theory weight Chapter 1 (pages 2-39), Glossary (pages G1 – G9) Review of Laboratory Methods ASSIGNMENTS. Averaging Grades Syllabus Return Module Alfa Vocabulary Know Your Book MSDS worksheet Module Alfa Test ********